Abstract
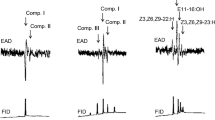
Males of Acanthoscelides obtectus (Coleoptera: Chrysomelidae, Bruchinae) emit methyl (E,R)-2,4,5-tetradecatrienoate that attracts females for mating. This study identified further roles for this compound in the sexual behavior of A. obtectus. Earlier observations revealed that males touched females with their antennae while tandem-running with them and initiated mounting and copulation, whereas they showed no such behavior toward other males. A series of subsequent laboratory choice tests were set up to establish if certain cuticular compounds aid contact sex recognition in A. obtectus. Males chose virgin females over other males. The activity toward females could be eliminated by rinsing with hexane, but was regained by application of female extract onto previously rinsed females. Gas chromatographic (GC) comparison of hexane extracts revealed the presence of two male-specific compounds, methyl (E,R)-2,4,5-tetradecatrienoate and octadecanal, which were absent from the behaviorally active female samples. Of the two compounds, methyl (E,R)-2,4,5-tetradecatrienoate was found to be responsible for the inhibition of male sexual behavior, similar to that observed with crude male extracts applied to virgin females. Furthermore, males preferred virgin over mated females. GC analyses revealed the presence of methyl (E,R)-2,4,5-tetradecatrienoate in mated females in amounts sufficient to curtail mating attempts. It appears that methyl (E,R)-2,4,5-tetradecatrienoate, besides being a male-produced sex pheromone, acts as a male-recognition signal in A. obtectus. Males also transfer it onto females during mating, resulting in mated females being avoided by courting males.


Similar content being viewed by others
References
Abate T, Ampofo JKO (1996) Insect pests of beans in Africa: their ecology and management. Annu Rev Entomol 41:45–73
Andersson J, Borg-Karlson A-K, Wiklund C (2000) Sexual cooperation and conflict in butterflies: a male-transferred anti-aphrodisiac reduces harassment of recently mated females. Proc R Soc Lond B 267:1271–1275
Annoscia D, Bruce T, Hooper T, Nazzi F, Pickett J (2010) Biological role of a compound produced by Acanthoscelides obtectus males. International Society of Chemical Ecology 26th Annual Meeting, Tours, France, 2010; P029, p 212
Brent CS, Byers JA (2011) Female attractiveness modulated by a male-derived anti-aphrodisiac pheromone in a plant bug. Anim Behav 82:937–943
Buckner JS (2010) Oxygenated derivatives of hydrocarbons. In: Blomquist GJ, Bagnères A-G (eds) Insect hydrocarbons. Biology, biochemistry, and chemical ecology. Cambridge University Press, Cambridge, pp 187–206
Carlson DA, Schlein Y (1991) Unusual polymethyl alkenes in tsetse flies acting as abstinon in Glossina morsitans. J Chem Ecol 17:267–284
Coates TW, Langley PA (1982) The causes of mating abstention in male tsetse flies Glossina morsitans. Physiol Entomol 7:235–242
Crudgington HS, Siva-Jothy MT (2000) Genital damage, kicking and early death: the battle of sexes takes a sinister turn in the bean weevil. Nature 407:855–856
Das AK, Huignard J, Barbier M, Quesneau-Thierry A (1980) Isolation of the two paragonial substances deposited into the spermatophores of Acanthoscelides obtectus (Coleoptera, Bruchidae). Cell Mol Life Sci 36:918–920
Griffith WP, Ley SV, Whitcombe GP, White AD (1987) Preparation and use of tetra-n-butylammonium per-ruthenate (TBAP reagent) and tetra-n-propylammonium per-ruthenate (TPAP reagent) as new catalytic oxidants for alcohols. J Chem Soc, Chem Commun. 1625–1627
Halstead DGH (1973) Preliminary biological studies on the pheromone produced by male Acanthoscelides obtectus (Say) (Coleoptera, Bruchidae). J Stored Prod Res 9:109–117
Hope JA, Horler DF, Rowlands DG (1967) A possible pheromone of the bruchid, Acanthoscelides obtectus (Say). J Stored Prod Res 3:387–388
Horler DF (1970) (−)-Methyl n-tetradeca-trans-2,4,5-trienoate, an allenic ester produced by the male dried bean beetle, Acanthoscelides obtectus (Say). J Chem Soc C 859–862
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu Rev Entomol 50:371–393
Huignard J (1974) Influence de la copμlation sur la function reproductrice femelle Acanthoscelides obtectus (Insecte coléoptère). I. Copμlation et spermatophore. Ann Sci Nat Zool Paris 16:361–434
Imura O (1990) Life histories of stored product insects. In: Fujii K, Gatehouse AMR (eds) Bruchids and legumes: Economics, ecology and coevolution. Kluwer, pp 257–269
Johansson BG, Jones TM (2007) The role of chemical communication in mate choice. Biol Rev 82:265–289
Kim G-H, Takabayashi J, Takahashi S, Tabata K (1993) Function of contact pheromone in the mating behavior of the Cryptomeria bark borer, Semanotus japonicus Lacordaire (Coleoptera: Cerambycidae). Appl Entomol Zool 28:525–535
Lange R, Reinhardt K, Michiels NK, Anthes N (2013) Functions, diversity, and evolution of traumatic mating. Biol Rev 88:585–601
Löfstedt C (1993) Moth pheromone genetics and evolution. Phil Trans R Soc Lond B 340:167–177
Maklakov AA, Kremer N, Arnqvist G (2006) Ageing and the evolution of female resistance to remating in seed beetles. Biol Lett 2:62–6
Maklakov AA, Kremer N, Arnqvist G (2007) The effects of age at mating on female life-history in a seed beetle. Behav Ecol 18:551–555
McCullagh, P, Nelder, JA (1989) Generalized linear models. Chapman & Hall/CRC Monographs on Statistics and Applied Probability, 37, 2nd ed, pp 511
Mori K (2012) Pheromone synthesis. Part 249: Syntheses of methyl (R, E)-2,4,5-tetradecatrienoate and methyl (2E,4Z)-2,4-decadienoate, the pheromone components of the male dried bean beetle, Acanthoscelides obtectus (Say). Tetrahedron 68:1936–1946
Nelson DR, Dillwith JW, Blomquist GJ (1981) Cuticμlar hydrocarbons of the house fly, Musca domestica. Insects Biochem 11:187–197
Nojima S, Shimomura K, Honda H, Yamamoto I, Ohsawa K (2007) Contact sex pheromone components of the cowpea weevil, Callosobruchus maculatus. J Chem Ecol 33:923–933
Parker GA, Pizzari T (2010) Sperm competition and ejaculate economics. Biol Rev 85:897–934
Pickett JA (1990) GC-MS in insect pheromone identification: three extreme case histories. In: McCaffery AR, Wilson ID (eds) Chromatography and isolation of insect hormones and pheromones. Plenum Press, pp 299–309
Prouvost O, Trabalon M, Papke M, Schulz S (1999) Contact sex signals on web and cuticle of Tegenaria atrica (Araneae, Agelenidae). Arch Insect Biochem Physiol 40:194–202
Savković U, Vučković I, Stojković B (2012) The growth on different stored legume species affects the profiles of cuticμlar hydrocarbon (CHC) in Acanthoscelides obtectus (Say). J Stored Prod Res 50:66–72
Schlein Y, Galun R, Ben-Eliahu MN (1980) Abstinons. Male-produced deterrents of mating in flies. J Chem Ecol 7:285–290
Slatyer RA, Mautz BS, Backwell PRY, Jennions MD (2012) Estimating genetic benefits of polyandry from experimental studies: a meta-analysis. Biol Rev 87:1–33
Smadja C, Butlin RK (2009) On the scent of speciation: the chemosensory system and its role in premating isolation. Heredity 102:77–97
Southgate BJ (1979) Biology of the bruchidae. Annu Rev Entomol 24:449–473
Stojković B, Savković U, Đorđević M, Tucić N (2014) Host-shift effects on mating behavior and incipient pre-mating isolation in seed beetle. Behav Ecol 25:553–564
Tanaka K, Ohsawa K, Honda H, Yamamoto I (1981) Copμlation release pheromone, erectin, from the azuki bean weevil (Callosobruchus chinensis L.). J Pestic Sci 6:75–82
Thomas ML (2011) Detection of female mating status using chemical signals and cues. Biol Rev 86:1–13
Tran BMD, Credland PF (1995) Consequences of inbreeding for the cowpea seed beetle, Callosobruchus macμlatus (F.) (Coleoptera: Bruchidae). Biol J Linn Soc 56:483–503
Tucić N, Cliksman I, Śeślija D, Milanovic D, Mikμljanac D, Stoikovic O (1996) Laboratory evolution of longevity in the bean weevil (Acanthoscelides obtectus). J Evol Biol 52:1713–1725
Weddle CB, Steiger S, Hamaker CG, Ower GD, Mitchell C, Sakaluk SK, Hunt J (2013) Cuticular hydrocarbons as a basis for chemosensory self-referencing in crickets: a potentially universal mechanism facilitating polyandry in insects. Ecol Lett 16:346–353
Acknowledgments
The authors thank Professors Kenji Mori and Wittko Francke for providing enantiomerically pure samples of methyl (E,R)-2,4,5-tetradecatrienoate. The authors are grateful for the comments of Professor Tibor Jermy and Dr Árpád Szentesi, as well as two anonymous reviewers, on an earlier version of the manuscript. This work was in part supported by the Hungarian Scientific Research Fund (OTKA) (grant K81494), by the Research and Technology Innovation Fund (grant OMFB-00609/2010) and by the Royal Society Grant IE111142 to JV. Rothamsted receives grant-aided support from the Biotechnology and Biological Sciences Research Council (BBSRC) of the United Kingdom.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 98 kb)
Rights and permissions
About this article
Cite this article
Vuts, J., Powers, S.J., Caulfield, J.C. et al. Multiple Roles of a Male-Specific Compound in the Sexual Behavior of the Dried Bean Beetle, Acanthoscelides Obtectus . J Chem Ecol 41, 287–293 (2015). https://doi.org/10.1007/s10886-015-0560-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-015-0560-3