Abstract
Background/Objectives
Our aim was to validate recent epidemiologic trends and describe the distribution of TIGAR-O risk factors in chronic pancreatitis (CP) patients.
Methods
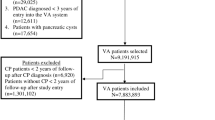
The NAPS-2 Continuation and Validation (NAPS2-CV) study prospectively enrolled 521 CP patients from 13 US centers from 2008 to 2012. CP was defined by definitive changes in imaging, endoscopy, or histology. Data were analyzed after stratification by demographic factors, physician-defined etiology, participating center, and TIGAR-O risk factors.
Results
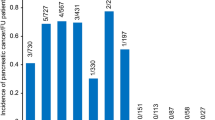
Demographics and physician-defined etiology in the NAPS2-CV study were similar to the original NAPS2 study. Mean age was 53 years (IQR 43, 62) with 55% males and 87% white. Overall, alcohol was the single most common etiology (46%) followed by idiopathic etiology (24%). Alcohol etiology was significantly more common in males, middle-aged (35–65 years), and non-whites. Females and elderly (≥65 years) were more likely to have idiopathic etiology, while younger patients (<35 years) to have genetic etiology. Variability in etiology was noted by participating centers (e.g., alcohol etiology ranged from 27 to 67% among centers enrolling ≥25 patients). Smoking was the most commonly identified (59%) risk factor followed by alcohol (53%), idiopathic (30%), obstructive (19%), and hyperlipidemia (13%). The presence of multiple TIGAR-O risk factors was common, with 1, 2, ≥3 risk factors observed in 27.6, 47.6, and 23.6% of the cohort, respectively.
Conclusion
Our data validate the current epidemiologic trends in CP. Alcohol remains the most common physician-defined etiology, while smoking was the most commonly identified TIGAR-O risk factor. Identification of multiple risk factors suggests CP to be a complex disease.

Similar content being viewed by others
References
Whitcomb DC, Yadav D, Adam S, et al. Multicenter approach to recurrent acute and chronic pancreatitis in the United States: the North American Pancreatitis Study 2 (NAPS2). Pancreatology. 2008;8:520–531.
Somogyi L, Martin SP, Venkatesan T, et al. Recurrent acute pancreatitis: an algorithmic approach to identification and elimination of inciting factors. Gastroenterology. 2001;120:708–717.
Steer ML, Waxman I, Freedman S. Chronic pancreatitis. N Engl J Med. 1995;332:1482–1490.
Forsmark CE. Management of chronic pancreatitis. Gastroenterology. 2013;144:1282–1291.e3.
Cote GA, Yadav D, Slivka A, et al. Alcohol and smoking as risk factors in an epidemiology study of patients with chronic pancreatitis. Clin Gastroenterol Hepatol. 2011;9:266–273. (quiz e27).
Yadav D, Slivka A, Sherman S, et al. Smoking is underrecognized as a risk factor for chronic pancreatitis. Pancreatology. 2010;10:713–719.
Frulloni L, Falconi M, Gabbrielli A, et al. Italian consensus guidelines for chronic pancreatitis. Dig Liver Dis. 2010;42:S381–S406.
Spanier B, Bruno MJ, Dijkgraaf MG. Incidence and mortality of acute and chronic pancreatitis in the Netherlands: a nationwide record-linked cohort study for the years 1995–2005. World J Gastroenterol. 2013;19:3018–3026.
Dominguez-Munoz JE, Lucendo A, Carballo LF, et al. A Spanish multicenter study to estimate the prevalence and incidence of chronic pancreatitis and its complications. Rev Esp Enferm Dig. 2014;106:239–245.
De-Las-Heras-Castano G. The study of chronic pancreatitis epidemiology—the big challenge. Rev Esp Enferm Dig. 2014;106:237–238.
Hirota M, Shimosegawa T, Masamune A, et al. The seventh nationwide epidemiological survey for chronic pancreatitis in Japan: clinical significance of smoking habit in Japanese patients. Pancreatology. 2014;14:490–496.
Levy P, Dominguez-Munoz E, Imrie C, et al. Epidemiology of chronic pancreatitis: burden of the disease and consequences. United Eur Gastroenterol J. 2014;2:345–354.
Etemad B, Whitcomb DC. Chronic pancreatitis: diagnosis, classification, and new genetic developments. Gastroenterology. 2001;120:682–707.
Aoun E, Chang CC, Greer JB, et al. Pathways to injury in chronic pancreatitis: decoding the role of the high-risk SPINK1 N34S haplotype using meta-analysis. PLoS One. 2008;3:e2003.
Stevens T, Conwell DL, Zuccaro G. Pathogenesis of chronic pancreatitis: an evidence-based review of past theories and recent developments. Am J Gastroenterol. 2004;99:2256–2270.
Greer JB, LaRusch J, Brand RE, et al. ABO blood group and chronic pancreatitis risk in the NAPS2 cohort. Pancreas. 2011;40:1188–1194.
Yadav D, Hawes RH, Brand RE, et al. Alcohol consumption, cigarette smoking, and the risk of recurrent acute and chronic pancreatitis. Arch Intern Med. 2009;169:1035–1045.
Burton F, Alkaade S, Collins D, et al. Use and perceived effectiveness of non-analgesic medical therapies for chronic pancreatitis in the United States. Aliment Pharmacol Ther. 2011;33:149–159.
Mullady DK, Yadav D, Amann ST, et al. Type of pain, pain-associated complications, quality of life, disability and resource utilisation in chronic pancreatitis: a prospective cohort study. Gut. 2011;60:77–84.
Amann ST, Yadav D, Barmada MM, et al. Physical and mental quality of life in chronic pancreatitis: a case-control study from the North American Pancreatitis Study 2 cohort. Pancreas. 2013;42:293–300.
Brand H, Diergaarde B, O’Connell MR, et al. Variation in the gamma-glutamyltransferase 1 gene and risk of chronic pancreatitis. Pancreas. 2013;42:836–840.
Melzack R. The McGill Pain Questionnaire: major properties and scoring methods. Pain. 1975;1:277–299.
Amtmann D, Cook KF, Jensen MP, et al. Development of a PROMIS item bank to measure pain interference. Pain. 2010;150:173–182.
Skinner HA, Sheu WJ. Reliability of alcohol use indices. The Lifetime Drinking History and the MAST. J Stud Alcohol. 1982;43:1157–1170.
LaRusch J, Whitcomb DC. Genetics of pancreatitis. Curr Opin Gastroenterol. 2011;27:467–474.
Apte MV, Wilson JS. Stellate cell activation in alcoholic pancreatitis. Pancreas. 2003;27:316–320.
Talukdar R, Tandon RK. Pancreatic stellate cells: new target in the treatment of chronic pancreatitis. J Gastroenterol Hepatol. 2008;23:34–41.
Christian JB, Bourgeois N, Snipes R, et al. Prevalence of severe (500 to 2,000 mg/dl) hypertriglyceridemia in United States adults. Am J Cardiol. 2011;107:891–897.
Kappelman MD, Rifas-Shiman SL, Kleinman K, et al. The prevalence and geographic distribution of Crohn’s disease and ulcerative colitis in the United States. Clin Gastroenterol Hepatol. 2007;5:1424–1429.
Funding
NIH (DCW—DK061451, U01DK108306), NIH (DY—DK077906, U01DK108306), and UL1 RR024153 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research. NIH (DC—U01DK108327) and NIH (CEF—U01DK108320).
Author information
Authors and Affiliations
Contributions
DLC, DY, and DCW were involved in study design. DLC, DY, PAB, BSS, SS, SA, TBG, MAA, CMW, MDL, TM, CEF, GAC, NMG, REB, AG, and AS were involved in acquisition of data. DLC and DY were involved in analysis and interpretation. DLC and DY were involved in drafting of manuscript. All authors were involved in critical review. YT, SRW, DLC, and DY were involved in statistical analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Conwell, D.L., Banks, P.A., Sandhu, B.S. et al. Validation of Demographics, Etiology, and Risk Factors for Chronic Pancreatitis in the USA: A Report of the North American Pancreas Study (NAPS) Group. Dig Dis Sci 62, 2133–2140 (2017). https://doi.org/10.1007/s10620-017-4621-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4621-z