Abstract
Background and Aims
Fusobacterium species are part of the gut microbiome in humans, but some species have been recognized as opportunistic pathogens implicated in inflammatory diseases including inflammatory bowel diseases. Here, we performed prevalence screening of Fusobacterium in ulcerative colitis (UC) in Japanese patients.
Methods
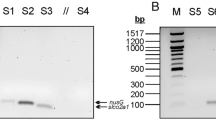
We examined Fusobacterium nucleatum (F. nucleatum) and whole Fusobacterium species (Pan-fusobacterium) by quantitative real-time PCR in 163 inflamed mucosae from 152 UC patients. Data were correlated with clinical subtypes of UC.
Results
In an initial prevalence screen, F. nucleatum and Pan-fusobacterium were detected in 6.3 % (4/64) and 53.1 % (34/64). For all 163 mucosae, the prevalence of Pan-fusobacterium was 54.6 % (89/163). Pan-fusobacterium status was concordant in inflamed and normal adjacent samples, and the matched cases during 1-year follow-up colonoscopy. The higher amount of Pan-fusobacterium was observed in chronic continuous type compared to one attack and relapse/remitting type (p = 0.039). The higher amount of Pan-fusobacterium was also associated with rather mild clinical course of disease, such as non-steroid dependency (p = 0.015), non-refractory phenotype (p = 0.013), and non-severe phenotype (p = 0.04). Based on the distribution of Pan-fusobacterium measurable cases, we identified 10 cases as having a high amount of Pan-fusobacterium (FB-high). The clinicopathological features of FB-high UC cases were also highlighted by chronic continuous type and mild phenotypes of disease.
Conclusion
Whole Fusobacterium species, but not F. nucleatum, are common in UC patients and have a role in persistence of colonic inflammation in UC. However, Fusobacterium infection is associated with rather mild clinical phenotypes of UC.



Similar content being viewed by others
References
Head KA, Jurenka JS. Inflammatory bowel disease part I: ulcerative colitis-pathophysiology and conventional and alternative treatment options. Altern Med Rev. 2003;8:247–283.
Lakatos L, Lakatos PL. Etiopathogenesis of inflammatory bowel diseases. Orv Hetil. 2003;144:1853–1860.
Sartor RB. Microbial influences in inflammatory bowel diseases. Gastroenterology. 2008;134:577–594.
Darfeuille-Michaud A, Neut C, Barnich N, et al. Presence of adherent Escherichia coli strains in ileal mucosa of patients with Crohn’s disease. Gastroenterology. 1998;115:1405–1413.
Petersen AM, Nielsen EM, Litrup E, Brynskov J, Mirsepasi H, Krogfelt KA. A phylogenetic group of Escherichia coli associated with active left-sided inflammatory bowel disease. BMC Microbiol. 2009;9:171.
Ohkusa T, Okayasu I, Ogihara T, Morita K, Ogawa M, Sato N. Induction of experimental ulcerative colitis by Fusobacterium varium isolated from colonic mucosa of patients with ulcerative colitis. Gut. 2003;52:79–83.
Signat B, Roques C, Poulet P, Duffaut D. Fusobacterium nucleatum in periodontal health and disease. Curr Issues Mol Biol. 2011;13:25–36.
Swidsinski A, Dörffel Y, Loening-Baucke V, et al. Acute appendicitis is characterised by local invasion with Fusobacterium nucleatum/necrophorum. Gut. 2011;60:34–40.
Moore WE, Moore LV. The bacteria of periodontal diseases. Periodontol. 1994;5:66–77.
Strauss J, Kaplan GG, Beck PL, et al. Invasive potential of gut mucosa-derived Fusobacterium nucleatum positively correlates with IBD status of the host. Inflamm Bowel Dis.. 2011;17:1971–1978.
Hayashi R, Tahara T, Shiroeda H, et al. (2012) Influence of IL17A polymorphisms (rs2275913 and rs3748067) on the susceptibility to ulcerative colitis. Clin Exp Med. 2013;13:239–244.
Okubo M, Tahara T, Shibata T, et al. Association study of common genetic variants in pre-microRNAs in patients with ulcerative colitis. J Clin Immunol. 2011;31:69–73.
Shiroeda H, Tahara T, Nakamura M, et al. Association between functional promoter polymorphisms of macrophage migration inhibitory factor (MIF) gene and ulcerative colitis in Japan. Cytokine. 2010;51:173–177.
Castellarin M, Warren RL, Freeman JD, et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res. 2012;22:299–306.
Boutaga K, van Winkelhoff AJ, Vandenbroucke-Grauls CM, Savelkoul PH. Periodontal pathogens: a quantitative comparison of anaerobic culture and real-time PCR. FEMS Immunol Med Microbiol. 2005;45:191–199.
Wilson GM, Flibotte S, Chopra V, Melnyk BL, Honer WG, Holt RA. DNA copy-number analysis in bipolar disorder and schizophrenia reveals aberrations in genes involved in glutamate signaling. Hum Mol Genet. 2006;15:743–749.
Rajilić-Stojanović M, Shanahan F, Guarner F, de Vos WM. Phylogenetic analysis of dysbiosis in ulcerative colitis during remission. Inflamm Bowel Dis. 2013;19:481–488.
Minami M, Ando T, Okamoto A, et al. Seroprevalence of Fusobacterium varium in ulcerative colitis patients in Japan. FEMS Immunol Med Microbiol. 2009;56:67–72.
Ekbom A, Helmick C, Zack M, Adami HO. Ulcerative colitis and colorectal cancer. A population-based study. N Engl J Med. 1990;323:1228–1233.
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–535.
Kostic AD, Gevers D, Pedamallu CS, et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res. 2012;22:292–298.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tahara, T., Shibata, T., Kawamura, T. et al. Fusobacterium Detected in Colonic Biopsy and Clinicopathological Features of Ulcerative Colitis in Japan. Dig Dis Sci 60, 205–210 (2015). https://doi.org/10.1007/s10620-014-3316-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3316-y