Abstract
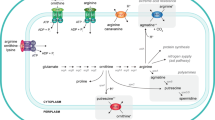
Cystathionine γ-lyase (CSE), one of three enzymes in the trans-sulfuration pathway, is responsible for the production of endogenous hydrogen sulfide (H2S) using l-cysteine or l-homocysteine as a substrate. The regulatory mechanism of CSE by exogenous H2S remains unknown. The transcription and expression of the CSE gene regulated by exogenous H2S at approximately physiologic concentrations was investigated using luciferase assay, Western blotting, and quantitative RT-PCR. The results revealed that exogenous H2S down-regulates the transcription and expression of CSE in mammalian cells at 10–80 μM. Exogenous H2S at 120 μM increases the transcription and expression of CSE significantly. At a concentration of exogenous H2S over 160 μM, the transcription and expression of CSE are inhibited completely. These findings suggest that CSE expression has not only a feedback to the enzyme itself at lower concentrations of exogenous H2S but also can be up-regulated at higher concentrations, and H2S may become toxic at higher levels.



Similar content being viewed by others
References
Bhatia M, Wong FL, Fu D, Lau HY, Moochhala SM, Moore PK (2005) Role of hydrogen sulfide in acute pancreatitis and associated lung injury. FASEB J 19:623–6255
Choudhary S, Rosenblatt KP, Fang L, Tian B, Wu ZH, Brasier AR (2011) High throughput short interfering RNA (siRNA) screening of the human kinome identifies novel kinases controlling the canonical nuclear factor-kappaB (NF-kappaB) activation pathway. J Biol Chem 286:37187–37195
Chunyu Z, Junbao D, Dingfang B, Hui Y, Xiuying T, Chaoshu T (2003) The regulatory effect of hydrogen sulfide on hypoxic pulmonary hypertension in rats. Biochem Biophys Res Commun 302:810–816
Diwakar L, Ravindranath V (2007) Inhibition of cystathionine-gamma-lyase leads to loss of glutathione and aggravation of mitochondrial dysfunction mediated by excitatory amino acid in the CNS. Neurochem Int 50:418–426
Elrod JW, Calvert JW, Morrison J, Doeller JE, Kraus DW, Tao L, Jiao X, Scalia R, Kiss L, Szabo C, Kimura H, Chow CW, Lefer DJ (2007) Hydrogen sulfide attenuates myocardial ischemia-reperfusion injury by preservation of mitochondrial function. Proc Natl Acad Sci USA 104:15560–51556
Enokido Y, Suzuki E, Iwasawa K, Namekata K, Okazawa H, Kimura H (2005) Cystathionine beta-synthase, a key enzyme for homocysteine metabolism, is preferentially expressed in the radial glia/astrocyte lineage of developing mouse CNS. FASEB J 19:1854–1856
Fang LP, Lin Q, Tang CS, Liu XM (2010) Hydrogen sulfide attenuates epithelial-mesenchymal transition of human alveolar epithelial cells. Pharmacol Res 61:298–305
Furne J, Saeed A, Levitt MD (2008) Whole tissue hydrogen sulfide concentration are orders of magnitude lower than presently accepted values. Am J Physiol Regul Integr Comp Physiol 295:R1479–R1485
Geng B, Du JB, Tang CS (2005) Application of sensitive sulphur electrode assay to measure and analyze cystathionine-gamma-lyase/hydrogen sulfide pathway of cardiovascular tissues, cells and plasma in rats. Beijing Da Xue Xue Bao 37:545–548
Hosoki R, Matsuki N, Kimura H (1997) The possible role of hydrogen sulfide as an endogenous smooth muscle relaxant in synergy with nitric oxide. Biochem Biophys Res Commun 237:527–531
Ishii I, Akahoshi N, Yu XN, Kobayashi Y, Namekata K, Komaki G, Kimura H (2004) Murine cystathionine gamma-lyase: complete cDNA and genomic sequences, promoter activity, tissue distribution and developmental expression. Biochem J 381:113–123
Kaneko Y, Kimura Y, Kimura H, Niki I (2006) l-cysteine inhibits insulin release from the pancreatic beta-cell: possible involvement of metabolic production of hydrogen sulfide, a novel gasotransmitter. Diabetes 55:1391–1397
Kang K, Zhao M, Jiang H, Tan G, Pan S, Sun X (2009) Role of hydrogen sulfide in hepatic ischemia-reperfusion-induced injury in rats. Liver Transpl 15:1306–1314
Kimura H (2002) Hydrogen sulfide as a neuromodulator. Mol Neurobiol 26:13–19
Kimura H (2011) Hydrogen sulfide: its production and functions. Exp Physiol 96:833–835
Levitt MD, Abdel-Rehim MS, Furne J (2011) Free and acid-labile hydrogen sulfide concentration in mouse tissues: anomalously high free hydrogen sulfide in aortic tissue. Antioxid Redox Signal 15:373–378
Li L, Bhatia M, Zhu YZ, Zhu YC, Ramnath RD, Wang ZJ, Anuar FB, Whiteman M, Salto-Tellez M, Moore PK (2005) Hydrogen sulfide is a novel mediator of lipopolysaccharide-induced inflammation in the mouse. FASEB J 19:1196–1198
Madden JA, Ahlf SB, Dantuma MW, Olson KR, Roerig DL (2012) Precursors and inhibitors of hydrogen sulfide synthesis affect acute hypoxic pulmonary vasoconstriction in the intact lung. J Appl Physiol 112:411–418
Patel P, Vatish M, Heptinstall J, Wang R, Carson RJ (2009) The endogenous production of hydrogen sulphide in intrauterine tissues. Reprod Biol Endocrinol 7:10
Reiffenstein RJ, Hulbert WC, Roth SH (1992) Toxicology of hydrogen sulfide. Annu Rev Pharmacol Toxicol 32:109–134
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108
Shibuya N, Tanaka M, Yoshida M, Ogasawara Y, Togawa T, Ishii K, Kimura H (2009) 3-Mercaptopyruvate sulfurtransferase produces hydrogen sulfide and bound sulfane sulfur in the brain. Antioxid Redox Signal 11:703–714
Sun YG, Cao YX, Wang WW, Ma SF, Yao T, Zhu YC (2008) Hydrogen sulphide is an inhibitor of L-type calcium channels and mechanical contraction in rat cardiomyocytes. Cardiovasc Res 79:632–641
Telezhkin V, Brazier SP, Cayzac S, Muller CT, Riccardi D, Kemp PJ (2009) Hydrogen sulfide inhibits human BKCa channels. Adv Exp Med Biol 648:65–72
Vitvitsky V, Thomas M, Ghorpade A, Gendelman HE, Banerjee R (2006) A functional trans-sulfuration pathway in the brain links to glutathione homeostasis. J Biol Chem 281:35785–35793
Wagner F, Scheuerle A, Weber S, Stahl B, McCook O, Knoferl MW, Huber-Lang M, Seitz DH, Thomas J, Asfar P, Szabo C, Moller P, Gebhard F, Georgieff M, Calzia E, Radermacher P, Wagner K (2011) Cardiopulmonary, histologic, and inflammatory effects of intravenous Na2S after blunt chest trauma-induced lung contusion in mice. J Trauma 71:1659–1667
Wang R (2003) The gasotransmitter role of hydrogen sulfide. Antioxid Redox Signal 5:493–501
Wang R (2009) Hydrogen sulfide: a new EDRF. Kidney Int 76:700–704
Wang R (2010) Hydrogen sulfide: the third gasotransmitter in biology and medicine. Antioxid Redox Signal 12:1061–1064
Wang YF, Mainali P, Tang CS, Shi L, Zhang CY, Yan H, Liu XQ, Du JB (2008) Effects of nitric oxide and hydrogen sulfide on the relaxation of pulmonary arteries in rats. Chin Med J (Engl) 121:420–423
Whitfield NL, Kreimier EL, Verdial FC, Skovgaard N, Olson KR (2008) Reappraisal of H2S/sulfide concentration in vertebrate blood and its potential significance in ischemic preconditioning and vascular signaling. Am J Physiol Regul Integr Comp Physiol 294:R1930–R1937
Wintner EA, Deckwerth TL, Langston W, Bengtsson A, Leviten D, Hill P, Insko MA, Dumpit R, Vanden Ekart E, Toombs CF, Szabo C (2010) A monobromobimane-based assay to measure the pharmacokinetic profile of reactive sulphide species in blood. Br J Pharmacol 160:941–957
Xu M, Wu YM, Li Q, Wang FW, He RR (2007) Electrophysiological effects of hydrogen sulfide on guinea pig papillary muscles in vitro. Sheng Li Xue Bao 59:215–220
Yan SK, Chang T, Wang H, Wu L, Wang R, Meng QH (2006) Effects of hydrogen sulfide on homocysteine-induced oxidative stress in vascular smooth muscle cells. Biochem Biophys Res Commun 351:485–491
Yang G, Cao K, Wu L, Wang R (2004) Cystathionine gamma-lyase overexpression inhibits cell proliferation via a H2S-dependent modulation of ERK1/2 phosphorylation and p21Cip/WAK−1. J Biol Chem 279:49199–49205
Yang W, Yang G, Jia X, Wu L, Wang R (2005) Activation of KATP channels by H2S in rat insulin-secreting cells and the underlying mechanisms. J Physiol 569:519–531
Yang G, Wu L, Wang R (2006) Pro-apoptotic effect of endogenous H2S on human aorta smooth muscle cells. FASEB J 20:553–555
Yang G, Wu L, Jiang B, Yang W, Qi J, Cao K, Meng Q, Mustafa AK, Mu W, Zhang S, Snyder SH, Wang R (2008) H2S as a physiologic vasorelaxant: hypertension in mice with deletion of cystathionine gamma-lyase. Science 322:587–590
Yang G, Wu L, Bryan S, Khaper N, Mani S, Wang R (2010) Cystathionine gamma-lyase deficiency and overproliferation of smooth muscle cells. Cardiovasc Res 86:487–495
Yin P, Zhao C, Li Z, Mei C, Yao W, Liu Y, Li N, Qi J, Wang L, Shi Y, Qiu S, Fan J, Zha X (2012) Sp1 is involved in regulation of cystathionine gamma-lyase gene expression and biological function by PI3 K/Akt pathway in human hepatocellular carcinoma cell lines. Cell Signal 24:1229–1240
Zhang J, Xie Y, Xu Y, Pan Y, Shao C (2011) Hydrogen sulfide contributes to hypoxia-induced radioresistance on hepatoma cells. J Radiat Res 52:622–628
Zhang L, Charron M, Wright WW, Chatterjee B, Song CS, Roy AK, Brown TR (2004) Nuclear factor-kappaB activates transcription of the androgen receptor gene in Sertoli cells isolated from testes of adult rats. Endocrinology 145:781–789
Zhao W, Wang R (2002) H2S-induced vasorelaxation and underlying cellular and molecular mechanisms. Am J Physiol Heart Circ Physiol 283:H474–H480
Zhao W, Zhang J, Lu Y, Wang R (2001) The vasorelaxant effect of H2S as a novel endogenous gaseous KATP channel opener. EMBO J 20:6008–6016
Zhu YZ, Wang ZJ, Ho P, Loke YY, Zhu YC, Huang SH, Tan CS, Whiteman M, Lu J, Moore PK (2007) Hydrogen sulfide and its possible roles in myocardial ischemia in experimental rats. J Appl Physiol 102:261–268
Acknowledgments
This study was supported by the National Basic Research Program of China (973 Program, No. 2010CB912604).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, M., Guo, Z. & Wang, S. The Effect of Certain Conditions in the Regulation of Cystathionine γ-Lyase by Exogenous Hydrogen Sulfide in Mammalian Cells. Biochem Genet 51, 503–513 (2013). https://doi.org/10.1007/s10528-013-9581-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-013-9581-1