Abstract
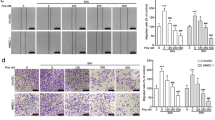
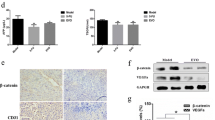
Small cell lung cancer (SCLC) demonstrates a strong etiological association with smoking. Although cigarette smoke is a mixture of about 4,000 compounds, nicotine is the addictive component of cigarette smoke. Several convergent studies have shown that nicotine promotes angiogenesis in lung cancers via the α7-nicotinic acetylcholine receptor (α7-nAChR) on endothelial cells. Therefore, we conjectured that α7-nAChR antagonists may attenuate nicotine-induced angiogenesis and be useful for the treatment of human SCLC. For the first time, our study explores the anti-angiogenic activity of MG624, a small-molecule α7-nAChR antagonist, in several experimental models of angiogenesis. We observed that MG624 potently suppressed the proliferation of primary human microvascular endothelial cells of the lung (HMEC-Ls). Furthermore, MG624 displayed robust anti-angiogenic activity in the Matrigel, rat aortic ring and rat retinal explant assays. The anti-angiogenic activity of MG624 was assessed by two in vivo models, namely the chicken chorioallantoic membrane model and the nude mice model. In both of these experimental models, MG624 inhibited angiogenesis of human SCLC tumors. Most importantly, the administration of MG624 was not associated with any toxic side effects, lethargy or discomfort in the mice. The anti-angiogenic activity of MG624 was mediated via the suppression of nicotine-induced FGF2 levels in HMEC-Ls. MG624 decreased nicotine-induced early growth response gene 1 (Egr-1) levels in HMEC-Ls, and reduced the levels of Egr-1 on the FGF2 promoter. Consequently, this process decreased FGF2 levels and angiogenesis. Our findings suggest that the anti-angiogenic effects of MG624 could be useful in anti-angiogenic therapy of human SCLCs.








Similar content being viewed by others
References
Bhadada SV, Goyal BR, Patel MM (2011) Angiogenic targets for potential disorders. Fundam Clin Pharmacol 25(1):29–47. doi:10.1111/j.1472-8206.2010.00814.x
Blann AD, Ramcharan KS, Stonelake PS, Luesley D, Lip GY (2011) The angiome: a new concept in cancer biology. J Clin Pathol. doi:10.1136/jcp.2011.088948
Folkman J (2003) Fundamental concepts of the angiogenic process. Curr Mol Med 3(7):643–651
Folkman J (2003) Angiogenesis inhibitors: a new class of drugs. Cancer Biol Ther 2(4 Suppl 1):S127–S133
Folkman J (2007) Angiogenesis: an organizing principle for drug discovery? Nat Rev Drug Discov 6(4):273–286
Cai J, Han S, Qing R, Liao D, Law B, Boulton ME (2011) In pursuit of new anti-angiogenic therapies for cancer treatment. Front Biosci 16:803–814
Kitamura H, Yazawa T, Sato H, Okudela K, Shimoyamada H (2009) Small cell lung cancer: significance of RB alterations and TTF-1 expression in its carcinogenesis, phenotype, and biology. Endocr Pathol 20(2):101–107. doi:10.1007/s12022-009-9072-4
Martinez-Garcia E, Irigoyen M, Gonzalez-Moreno O, Corrales L, Teijeira A, Salvo E, Rouzaut A (2010) Repetitive nicotine exposure leads to a more malignant and metastasis-prone phenotype of SCLC: a molecular insight into the importance of quitting smoking during treatment. Toxicol Sci 116(2):467–476. doi:10.1093/toxsci/kfq138
Heeschen C, Jang JJ, Weis M, Pathak A, Kaji S, Hu RS, Tsao PS, Johnson FL, Cooke JP (2001) Nicotine stimulates angiogenesis and promotes tumor growth and atherosclerosis. Nat Med 7(7):833–839
Heeschen C, Weis M, Aicher A, Dimmler S, Cooke JP (2002) A novel angiogenic pathway mediated by non-neuronal nicotinic acetylcholine receptors. J Clin Invest 110:527–536
Arias HR, Richards VE, Ng D, Ghafoori ME, Le V, Mousa SA (2009) Role of non-neuronal nicotinic acetylcholine receptors in angiogenesis. Int J Biochem Cell Biol 41(7):1441–1451. doi:10.1016/j.biocel.2009.01.013
Costa F, Soares R (2009) Nicotine: a pro-angiogenic factor. Life Sci 84(23–24):785–790. doi:10.1016/j.lfs.2009.03.002
Singh S, Pillai S, Chellappan S (2011) Nicotinic acetylcholine receptor signaling in tumor growth and metastasis. J Oncol 2011:456743. doi:10.1155/2011/456743
Dasgupta P, Chellappan SP (2006) Nicotine-mediated cell proliferation and angiogenesis: new twists to an old story. Cell Cycle 5(20):2324–2328
Egleton RD, Brown KC, Dasgupta P (2009) Angiogenic activity of nicotinic acetylcholine receptors: implications in tobacco-related vascular diseases. Pharmacol Ther 121(2):205–223
Cooke JP (2007) Angiogenesis and the role of the endothelial nicotinic acetylcholine receptor. Life Sci 80(24–25):2347–2351
Cooke JP, Bitterman H (2004) Nicotine and angiogenesis: a new paradigm for tobacco-related diseases. Ann Med 36(1):33–40
Kiuchi K, Matsuoka M, Wu JC, Lima ESR, Kengatharan M, Verghese M, Ueno S, Yokoi K, Khu NH, Cooke JP, Campochiaro PA (2008) Mecamylamine suppresses Basal and nicotine-stimulated choroidal neovascularization. Invest Ophthalmol Vis Sci 49(4):1705–1711
Suner IJ, Espinosa-Heidmann DG, Marin-Castano ME, Hernandez EP, Pereira-Simon S, Cousins SW (2004) Nicotine increases size and severity of experimental choroidal neovascularization. Invest Ophthalmol Vis Sci 45(1):311–317
Paleari L, Catassi A, Ciarlo M, Cavalieri Z, Bruzzo C, Servent D, Cesario A, Chessa L, Cilli M, Piccardi F, Granone P, Russo P (2008) Role of alpha7-nicotinic acetylcholine receptor in human non-small cell lung cancer proliferation. Cell Prolif 41(6):936–959. doi:10.1111/j.1365-2184.2008.00566.x
Paleari L, Cesario A, Fini M, Russo P (2009) Alpha7-Nicotinic receptor antagonists at the beginning of a clinical era for NSCLC and Mesothelioma? Drug Discov Today 14(17–18):822–836. doi:10.1016/j.drudis.2009.06.016
Paleari L, Grozio A, Cesario A, Russo P (2008) The cholinergic system and cancer. Semin Cancer Biol 18(3):211–217. doi:10.1016/j.semcancer.2007.12.009
Grozio A, Paleari L, Catassi A, Servent D, Cilli M, Piccardi F, Paganuzzi M, Cesario A, Granone P, Mourier G, Russo P (2008) Natural agents targeting the alpha7-nicotinic-receptor in NSCLC: a promising prospective in anti-cancer drug development. Int J Cancer 122(8):1911–1915. doi:10.1002/ijc.23298
Paleari L, Sessa F, Catassi A, Servent D, Mourier G, Doria-Miglietta G, Ognio E, Cilli M, Dominioni L, Paolucci M, Calcaterra A, Cesario A, Margaritora S, Granone P, Russo P (2009) Inhibition of non-neuronal alpha7-nicotinic receptor reduces tumorigenicity in A549 NSCLC xenografts. Int J Cancer 125(1):199–211. doi:10.1002/ijc.24299
Alama A, Bruzzo C, Cavalieri Z, Forlani A, Utkin Y, Casciano I, Romani M (2011) Inhibition of the nicotinic acetylcholine receptors by cobra venom alpha-neurotoxins: is there a perspective in lung cancer treatment? PLoS One 6(6):e20695. doi:10.1371/journal.pone.0020695
Gotti C, Balestra B, Moretti M, Rovati GE, Maggi L, Rossoni G, Berti F, Villa L, Pallavicini M, Clementi F (1998) 4-Oxystilbene compounds are selective ligands for neuronal nicotinic alphaBungarotoxin receptors. Br J Pharmacol 124(6):1197–1206. doi:10.1038/sj.bjp.0701957
Gotti C, Carbonnelle E, Moretti M, Zwart R, Clementi F (2000) Drugs selective for nicotinic receptor subtypes: a real possibility or a dream? Behav Brain Res 113(1–2):183–192
Maggi L, Palma E, Eusebi F, Moretti M, Balestra B, Clementi F, Gotti C (1999) Selective effects of a 4-oxystilbene derivative on wild and mutant neuronal chick alpha7 nicotinic receptor. Br J Pharmacol 126(1):285–295. doi:10.1038/sj.bjp.0702299
Tanibuchi Y, Wu J, Toyohara J, Fujita Y, Iyo M, Hashimoto K (2010) Characterization of [(3)H]CHIBA-1001 binding to alpha7 nicotinic acetylcholine receptors in the brain from rat, monkey, and human. Brain Res 1348:200–208. doi:10.1016/j.brainres.2010.06.008
Wu J, Toyohara J, Tanibuchi Y, Fujita Y, Zhang J, Chen H, Matsuo M, Wang RF, Hashimoto K (2010) Pharmacological characterization of [(125)I]CHIBA-1006 binding, a new radioligand for alpha7 nicotinic acetylcholine receptors, to rat brain membranes. Brain Res 1360:130–137. doi:10.1016/j.brainres.2010.08.095
Fahmy RG, Dass CR, Sun LQ, Chesterman CN, Khachigian LM (2003) Transcription factor Egr-1 supports FGF-dependent angiogenesis during neovascularization and tumor growth. Nat Med 9(8):1026–1032. doi:10.1038/nm905
Wang D, Mayo MW, Baldwin AS Jr (1997) Basic fibroblast growth factor transcriptional autoregulation requires EGR-1. Oncogene 14(19):2291–2299. doi:10.1038/sj.onc.1201069
Jimenez SK, Sheikh F, Jin Y, Detillieux KA, Dhaliwal J, Kardami E, Cattini PA (2004) Transcriptional regulation of FGF-2 gene expression in cardiac myocytes. Cardiovasc Res 62(3):548–557. doi:10.1016/j.cardiores.2004.01.032
Brown KC, Witte TR, Hardman WE, Luo H, Chen YC, Carpenter AB, Lau JK, Dasgupta P (2010) Capsaicin displays anti-proliferative activity against human small cell lung cancer in cell culture and nude mice models via the E2F pathway. PLoS One 5(4):e10243. doi:10.1371/journal.pone.0010243
Dasgupta P, Rastogi S, Pillai S, Ordonez-Ercan D, Morris M, Haura E, Chellappan S (2006) Nicotine induces cell proliferation by beta-arrestin-mediated activation of Src and Rb-Raf-1 pathways. J Clin Invest 116(8):2208–2217
Dasgupta P, Kinkade R, Joshi B, Decook C, Haura E, Chellappan S (2006) Nicotine inhibits apoptosis induced by chemotherapeutic drugs by up-regulating XIAP and survivin. Proc Natl Acad Sci USA 103(16):6332–6337
Dasgupta P, Sun J, Wang S, Fusaro G, Betts V, Padmanabhan J, Sebti SM, Chellappan SP (2004) Disruption of the Rb–Raf-1 interaction inhibits tumor growth and angiogenesis. Mol Cell Biol 24(21):9527–9541
Kinkade R, Dasgupta P, Carie A, Pernazza D, Carless M, Pillai S, Lawrence N, Sebti SM, Chellappan S (2008) A small molecule disruptor of Rb/Raf-1 interaction inhibits cell proliferation, angiogenesis, and growth of human tumor xenografts in nude mice. Cancer Res 68(10):3810–3818. doi:10.1158/0008-5472.CAN-07-6672
Shafiee A, Penn JS, Krutzsch HC, Inman JK, Roberts DD, Blake DA (2000) Inhibition of retinal angiogenesis by peptides derived from thrombospondin-1. Invest Ophthalmol Vis Sci 41(8):2378–2388
Banumathi E, Haribalaganesh R, Sheik Pran Babu S, Sirish Kumar N, Sangiliyandi G (2009) High-yielding enzymatic method for isolation and culture of microvascular endothelial cells from bovine retinal blood vessels. Microvasc Res 77(3):377–381. doi:10.1016/j.mvr.2008.12.005
Ponce ML, Kleinmann HK (2003) The chick chorioallantoic membrane as an in vivo angiogenesis model. Curr Protoc Cell Biol. Chapter 19:Unit 19 15. doi:10.1002/0471143030.cb1905s18
Gu JW, Bailey AP, Sartin A, Makey I, Brady AL (2005) Ethanol stimulates tumor progression and expression of vascular endothelial growth factor in chick embryos. Cancer 103(2):422–431. doi:10.1002/cncr.20781
Song P, Sekhon HS, Lu A, Arredondo J, Sauer D, Gravett C, Mark GP, Grando SA, Spindel ER (2007) M3 muscarinic receptor antagonists inhibit small cell lung carcinoma growth and mitogen-activated protein kinase phosphorylation induced by acetylcholine secretion. Cancer Res 67(8):3936–3944
Xia G, Kumar SR, Masood R, Koss M, Templeman C, Quinn D, Zhu S, Reddy R, Krasnoperov V, Gill PS (2005) Up-regulation of EphB4 in mesothelioma and its biological significance. Clin Cancer Res 11(12):4305–4315. doi:10.1158/1078-0432.CCR-04-2109
Moilanen E, Thomsen LL, Miles DW, Happerfield DW, Knowles RG, Moncada S (1998) Persistent induction of nitric oxide synthase in tumours from mice treated with the anti-tumour agent 5, 6-dimethylxanthenone-4-acetic acid. Br J Cancer 77(3):426–433
Dasgupta P, Rizwani W, Pillai S, Davis R, Banerjee S, Hug K, Lloyd M, Coppola D, Haura E, Chellappan SP (2011) ARRB1-mediated regulation of E2F target genes in nicotine-induced growth of lung tumors. J Natl Cancer Inst 103(4):317–333. doi:10.1093/jnci/djq541
Chang PY, Lu SC, Lee CM, Chen YJ, Dugan TA, Huang WH, Chang SF, Liao WS, Chen CH, Lee YT (2008) Homocysteine inhibits arterial endothelial cell growth through transcriptional downregulation of fibroblast growth factor-2 involving G protein and DNA methylation. Circ Res 102(8):933–941. doi:10.1161/CIRCRESAHA.108.171082
Ralser M, Querfurth R, Warnatz HJ, Lehrach H, Yaspo ML, Krobitsch S (2006) An efficient and economic enhancer mix for PCR. Biochem Biophys Res Commun 347(3):747–751. doi:10.1016/j.bbrc.2006.06.151
Kundumani-Sridharan V, Niu J, Wang D, Van Quyen D, Zhang Q, Singh NK, Subramani J, Karri S, Rao GN (2010) 15(S)-hydroxyeicosatetraenoic acid-induced angiogenesis requires Src-mediated Egr-1-dependent rapid induction of FGF-2 expression. Blood 115(10):2105–2116. doi:10.1182/blood-2009-09-241802
Fisch TM, Prywes R, Simon MC, Roeder RG (1989) Multiple sequence elements in the c-fos promoter mediate induction by cAMP. Genes Dev 3(2):198–211
Dom AM, Buckley AW, Brown KC, Egleton RD, Marcelo AJ, Proper NA, Weller DE, Shah YH, Lau JK, Dasgupta P (2010) The α7-nicotinic acetylcholine receptor and MMP-2/9 pathway mediate the pro-angiogenic effect of nicotine in human retinal endothelial cells. Invest Ophthalmol Vis Sci. doi:10.1167/iovs.10-5461
Romanelli MN, Gualtieri F (2003) Cholinergic nicotinic receptors: competitive ligands, allosteric modulators, and their potential applications. Med Res Rev 23(4):393–426
Cucina A, Corvino V, Sapienza P, Borrelli V, Lucarelli M, Scarpa S, Strom R, Santoro-D’Angelo L, Cavallaro A (1999) Nicotine regulates basic fibroblastic growth factor and transforming growth factor beta1 production in endothelial cells. Biochem Biophys Res Commun 257(2):306–312
Cucina A, Sapienza P, Corvino V, Borrelli V, Mariani V, Randone B, Santoro-D’Angelo L, Cavallaro A (2000) Nicotine-induced smooth muscle cell proliferation is mediated through bFGF and TGF-b1. Surgery 127(1):316–322
Cucina A, Sapienza P, Corvino V, Borrelli V, Randone B, Santoro-D’Angelo L, Cavallaro A (2000) Nicotine induces platelet-derived growth factor release and cytoskeletal alteration in aortic smooth muscle cells. Surgery 127(1):72–78
Pasumarthi KB, Jin Y, Cattini PA (1997) Cloning of the rat fibroblast growth factor-2 promoter region and its response to mitogenic stimuli in glioma C6 cells. J Neurochem 68(3):898–908
Biesiada E, Razandi M, Levin ER (1996) Egr-1 activates basic fibroblast growth factor transcription. Mechanistic implications for astrocyte proliferation. J Biol Chem 271(31):18576–18581
Jin Y, Sheikh F, Detillieux KA, Cattini PA (2000) Role for early growth response-1 protein in alpha(1)-adrenergic stimulation of fibroblast growth factor-2 promoter activity in cardiac myocytes. Mol Pharmacol 57(5):984–990
Ganti AK, West WW, Lackner RP, Kessinger A (2010) Current concepts in the diagnosis and management of small-cell lung cancer. Oncology (Williston Park) 24(11):1034–1039
Lucchi M, Mussi A, Fontanini G, Faviana P, Ribechini A, Angeletti CA (2002) Small cell lung carcinoma (SCLC): the angiogenic phenomenon. Eur J Cardiothorac Surg 21(6):1105–1110
Zhu BQ, Heeschen C, Sievers RE, Karliner JS, Parmley WW, Glantz SA, Cooke JP (2003) Second hand smoke stimulates tumor angiogenesis and growth. Cancer Cell 4(3):191–196
Conklin BS, Zhao W, Zhong DS, Chen C (2002) Nicotine and cotinine up-regulate vascular endothelial growth factor expression in endothelial cells. Am J Pathol 160(2):413–418
Fukuda N (2010) Cigarette smoking induces vascular proliferative disease through the activation of Egr-1. Cardiovasc Res 88(2):207–208. doi:10.1093/cvr/cvq295
Choi JE, Kim JN, Jeong SH, Son SW (2010) Nicotine induces the expression of early growth response-1 in human skin dermal fibroblasts. Int J Dermatol 49(2):158–161. doi:10.1111/j.1365-4632.2009.04210.x
Acknowledgments
We thank Dr. Srikumar Chellappan and his laboratory for continuous support. We are grateful to Carla Cook, Dr. Travis Salisbury and Dr. Nalini Santanam for their help in the real-time PCR experiments. We also acknowledge Dr. Cattini and Dr. Nilson for providing us the constructs used in this study. This work was supported by the grants Young Clinical Scientist Award (#82115) from the Flight Attendant Medical Association, Miami, FL, Research Starter Grant from the PhRMA Foundation, Washington D.C., ASPET-Astellas Award, NIH-ROI-NIDDK65003 (to RDE). AMD and KCB are recipients of graduate fellowships from the WVSGC.
Conflict of interest
None of the authors have any conflict of interest.
Ethics statement
All experiments were performed in the United States of America and they comply with the laws of the USA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Kathleen C. Brown, Jamie K. Lau and Aaron M. Dom contributed equally to the work presented here and should therefore be regarded as equivalent authors.
An erratum to this article can be found at http://dx.doi.org/10.1007/s10456-012-9258-0.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Brown, K.C., Lau, J.K., Dom, A.M. et al. MG624, an α7-nAChR antagonist, inhibits angiogenesis via the Egr-1/FGF2 pathway. Angiogenesis 15, 99–114 (2012). https://doi.org/10.1007/s10456-011-9246-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-011-9246-9