Abstract
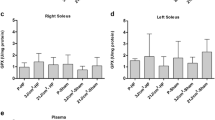
Following heart failure (HF), immune activation leads to an imbalance between pro-inflammatory and anti-inflammatory cytokines. Low-level laser therapy (LLLT) has been used as an anti-inflammatory treatment in several disease conditions. However, the effect of LLLT on the skeletal muscle of rats with HF remains unclear. The present report aimed to evaluate the influence of LLLT on the inflammatory profile of rats with HF. The left coronary artery was ligated to induce HF and a sham operation was performed in the control groups. Male Wistar rats (n = 49) were assigned to one of six groups: placebo sham rats (P-Sham; n = 8), LLLT at a dose of 3 J/cm2 sham rats (3 J/cm2-Sham; n = 8), LLLT at a dose of 21 J/cm2 sham rats (21 J/cm2-Sham; n = 8), placebo HF rats (P-HF; n = 9), LLLT at a dose of 3 J/cm2 HF rats (3 J/cm2-HF; n = 8), and LLLT at a dose of 21 J/cm2 HF rats (21 J/cm2-HF; n = 8). Four weeks after myocardial infarction or sham surgery, rats were subjected to LLLT (InGaAlP 660 nm, spot size 0.035 cm2, output power 20 mW, power density 0.571 W/cm2, energy density 3 or 21 J/cm2, exposure time 5.25 s and 36.75 s) on the right gastrocnemius for 10 consecutive days. LLLT reduced plasma IL-6 levels (61.3 %; P < 0.01), TNF-α/IL-10 (61.0 %; P < 0.01) and IL-6/IL-10 ratios (77.3 %; P < 0.001) and increased IL-10 levels (103 %; P < 0.05) in the 21 J/cm2-HF group. Moreover, LLLT reduced the TNF-α (20.1 % and 21.3 %; both P < 0.05) and IL-6 levels (54.3 % and 37.8 %; P < 0.01 and P < 0.05, respectively) and the IL-6/IL-10 ratio (59.7 % and 42.2 %; P < 0.001 and P < 0.05, respectively) and increased IL-10 levels (81.0 % and 85.1 %; both P < 0.05) and the IL-10/TNF-α ratio (171.5 % and 119.8 %; P < 0.001 and P < 0.05, respectively) in the gastrocnemius in the 3 J/cm2-HF and 21 J/cm2-HF groups. LLLT showed systemic and skeletal muscle anti-inflammatory effects in rats with HF.




Similar content being viewed by others
References
Yndestad A, Damas JK, Oie E, Ueland T, Gullestad L, Aukrust P (2006) Systemic inflammation in heart failure—the whys and wherefores. Heart Fail Rev 11(1):83–92. doi:10.1007/s10741-006-9196-2
Seta Y, Shan K, Bozkurt B, Oral H, Mann DL (1996) Basic mechanisms in heart failure: the cytokine hypothesis. J Card Fail 2(3):243–249. doi:10.1016/S1071-9164(96)80047-9
Kaur K, Dhingra S, Slezak J, Sharma AK, Bajaj A, Singal PK (2009) Biology of TNFalpha and IL-10, and their imbalance in heart failure. Heart Fail Rev 14(2):113–123. doi:10.1007/s10741-008-9104-z
Sharma R, Coats AJ, Anker SD (2000) The role of inflammatory mediators in chronic heart failure: cytokines, nitric oxide, and endothelin-1. Int J Cardiol 72(2):175–186. doi:10.1016/S0167-5273(99)00186-2
Deswal A, Petersen NJ, Feldman AM, Young JB, White BG, Mann DL (2001) Cytokines and cytokine receptors in advanced heart failure: an analysis of the cytokine database from the Vesnarinone trial (VEST). Circulation 103(16):2055–2059. doi:10.1161/01.CIR.103.16.2055
Torre-Amione G, Kapadia S, Benedict C, Oral H, Young JB, Mann DL (1996) Proinflammatory cytokine levels in patients with depressed left ventricular ejection fraction: a report from the Studies of Left Ventricular Dysfunction (SOLVD). J Am Coll Cardiol 27(5):1201–1206. doi:10.1016/0735-1097(95)00589-7
Stumpf C, Seybold K, Petzi S, Wasmeier G, Raaz D, Yilmaz A, Anger T, Daniel WG, Garlichs CD (2008) Interleukin-10 improves left ventricular function in rats with heart failure subsequent to myocardial infarction. Eur J Heart Fail 10(8):733–739. doi:10.1016/j.ejheart.2008.06.007
Stumpf C, Lehner C, Yilmaz A, Daniel WG, Garlichs CD (2003) Decrease of serum levels of the anti-inflammatory cytokine interleukin-10 in patients with advanced chronic heart failure. Clin Sci (Lond) 105(1):45–50. doi:10.1042/CS20020359
Nunes RB, Tonetto M, Machado N, Chazan M, Heck TG, Veiga AB, Dall'Ago P (2008) Physical exercise improves plasmatic levels of IL-10, left ventricular end-diastolic pressure, and muscle lipid peroxidation in chronic heart failure rats. J Appl Physiol 104(6):1641–1647. doi:10.1152/japplphysiol.00062.2008
Gielen S, Adams V, Mobius-Winkler S, Linke A, Erbs S, Yu J, Kempf W, Schubert A, Schuler G, Hambrecht R (2003) Anti-inflammatory effects of exercise training in the skeletal muscle of patients with chronic heart failure. J Am Coll Cardiol 42(5):861–868. doi:10.1016/S0735-1097(03)00848-9
Schulze PC, Gielen S, Adams V, Linke A, Mobius-Winkler S, Erbs S, Kratzsch J, Hambrecht R, Schuler G (2003) Muscular levels of proinflammatory cytokines correlate with a reduced expression of insulinlike growth factor-I in chronic heart failure. Basic Res Cardiol 98(4):267–274. doi:10.1007/s00395-003-0411-1
Batista ML Jr, Rosa JC, Lopes RD, Lira FS, Martins E Jr, Yamashita AS, Brum PC, Lancha AH Jr, Lopes AC, Seelaender M (2010) Exercise training changes IL-10/TNF-alpha ratio in the skeletal muscle of post-MI rats. Cytokine 49(1):102–108. doi:10.1016/j.cyto.2009.10.007
Lunde PK, Sjaastad I, Schiotz Thorud HM, Sejersted OM (2001) Skeletal muscle disorders in heart failure. Acta Physiol Scand 171(3):277–294. doi:10.1046/j.1365-201x.2001.00830.x
Cicoira M, Bolger AP, Doehner W, Rauchhaus M, Davos C, Sharma R, Al-Nasser FO, Coats AJ, Anker SD (2001) High tumour necrosis factor-alpha levels are associated with exercise intolerance and neurohormonal activation in chronic heart failure patients. Cytokine 15(2):80–86. doi:10.1006/cyto.2001.0918
Gielen S, Adams V, Linke A, Erbs S, Mobius-Winkler S, Schubert A, Schuler G, Hambrecht R (2005) Exercise training in chronic heart failure: correlation between reduced local inflammation and improved oxidative capacity in the skeletal muscle. Eur J Cardiovasc Prev Rehabil 12(4):393–400. doi:10.1097/01.hjr.0000174824.94892.43
Anker SD, Sharma R (2002) The syndrome of cardiac cachexia. Int J Cardiol 85(1):51–66. doi:10.1016/S0167-5273(02)00233-4
Lopes-Martins RA, Marcos RL, Leonardo PS, Prianti AC Jr, Muscara MN, Aimbire F, Frigo L, Iversen VV, Bjordal JM (2006) Effect of low-level laser (Ga–Al–As 655nm) on skeletal muscle fatigue induced by electrical stimulation in rats. J Appl Physiol 101(1):283–288. doi:10.1152/japplphysiol.01318.2005
Aimbire F, Albertini R, Pacheco MT, Castro-Faria-Neto HC, Leonardo PS, Iversen VV, Lopes-Martins RA, Bjordal JM (2006) Low-level laser therapy induces dose-dependent reduction of TNFalpha levels in acute inflammation. Photomed Laser Surg 24(1):33–37. doi:10.1089/pho.2006.24.33
Sousa LR, Cavalcanti BN, Marques MM (2009) Effect of laser phototherapy on the release of TNF-alpha and MMP-1 by endodontic sealer-stimulated macrophages. Photomed Laser Surg 27(1):37–42. doi:10.1089/pho.2007.2220
Boschi ES, Leite CE, Saciura VC, Caberlon E, Lunardelli A, Bitencourt S, Melo DA, Oliveira JR (2008) Anti-Inflammatory effects of low-level laser therapy (660nm) in the early phase in carrageenan-induced pleurisy in rat. Lasers Surg Med 40(7):500–508. doi:10.1002/lsm.20658
Pires D, Xavier M, Araujo T, Silva JA Jr, Aimbire F, Albertini R (2011) Low-level laser therapy (LLLT; 780nm) acts differently on mRNA expression of anti- and pro-inflammatory mediators in an experimental model of collagenase-induced tendinitis in rat. Lasers Med Sci 26(1):85–94. doi:10.1007/s10103-010-0811-z
Mesquita-Ferrari RA, Martins MD, Silva JA Jr, da Silva TD, Piovesan RF, Pavesi VC, Bussadori SK, Fernandes KP (2011) Effects of low-level laser therapy on expression of TNF-alpha and TGF-beta in skeletal muscle during the repair process. Lasers Med Sci 26(3):335–340. doi:10.1007/s10103-010-0850-5
Pfeffer MA, Pfeffer JM, Fishbein MC, Fletcher PJ, Spadaro J, Kloner RA, Braunwald E (1979) Myocardial infarct size and ventricular function in rats. Circ Res 44(4):503–512. doi:10.1161/01.RES.44.4.503
Jaenisch RB, Hentschke VS, Quagliotto E, Cavinato PR, Schmeing LA, Xavier LL, Dal Lago P (2011) Respiratory muscle training improves hemodynamics, autonomic function, baroreceptor sensitivity, and respiratory mechanics in rats with heart failure. J Appl Physiol 111(6):1664–1670. doi:10.1152/japplphysiol.01245.2010
Francis J, Weiss RM, Wei SG, Johnson AK, Felder RB (2001) Progression of heart failure after myocardial infarction in the rat. Am J Physiol Regul Integr Comp Physiol 281(5):R1734–1745
Blalock EM, Phelps JT, Pancani T, Searcy JL, Anderson KL, Gant JC, Popovic J, Avdiushko MG, Cohen DA, Chen KC, Porter NM, Thibault O (2010) Effects of long-term pioglitazone treatment on peripheral and central markers of aging. PLoS One 5(4):e10405. doi:10.1371/journal.pone.0010405
Hedayat M, Mahmoudi MJ, Rose NR, Rezaei N (2010) Proinflammatory cytokines in heart failure: double-edged swords. Heart Fail Rev 15(6):543–562. doi:10.1007/s10741-010-9168-4
Chung ES, Packer M, Lo KH, Fasanmade AA, Willerson JT (2003) Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-alpha, in patients with moderate-to-severe heart failure: results of the anti-TNF Therapy Against Congestive Heart Failure (ATTACH) trial. Circulation 107(25):3133–3140. doi:10.1161/01.CIR.0000077913.60364.D2
Mann DL, McMurray JJ, Packer M, Swedberg K, Borer JS, Colucci WS, Djian J, Drexler H, Feldman A, Kober L, Krum H, Liu P, Nieminen M, Tavazzi L, van Veldhuisen DJ, Waldenstrom A, Warren M, Westheim A, Zannad F, Fleming T (2004) Targeted anticytokine therapy in patients with chronic heart failure: results of the Randomized Etanercept Worldwide Evaluation (RENEWAL). Circulation 109(13):1594–1602. doi:10.1161/01.CIR.0000124490.27666.B2
Mann DL (2005) Targeted anticytokine therapy and the failing heart. Am J Cardiol 95(11A):9C–16C. doi:10.1016/j.amjcard.2005.03.007
Bjordal JM, Johnson MI, Iversen V, Aimbire F, Lopes-Martins RA (2006) Photoradiation in acute pain: a systematic review of possible mechanisms of action and clinical effects in randomized placebo-controlled trials. Photomed Laser Surg 24(2):158–168. doi:10.1089/pho.2006.24.158
Moreira MS, Velasco IT, Ferreira LS, Ariga SK, Barbeiro DF, Meneguzzo DT, Abatepaulo F, Marques MM (2009) Effect of phototherapy with low intensity laser on local and systemic immunomodulation following focal brain damage in rat. J Photochem Photobiol B 97(3):145–151. doi:10.1016/j.jphotobiol.2009.09.002
Zhevago NA, Samoilova KA (2006) Pro- and anti-inflammatory cytokine content in human peripheral blood after its transcutaneous (in vivo) and direct (in vitro) irradiation with polychromatic visible and infrared light. Photomed Laser Surg 24(2):129–139. doi:10.1089/pho.2006.24.129
De Marchi T, Leal Junior EC, Bortoli C, Tomazoni SS, Lopes-Martins RA, Salvador M (2011) Low-level laser therapy (LLLT) in human progressive-intensity running: effects on exercise performance, skeletal muscle status, and oxidative stress. Lasers Med Sci 27(1):231–236. doi:10.1007/s10103-011-0955-5
Calderin S, Garcia-Nunez JA, Gomez C (2012) Short-term clinical and osteoimmunological effects of scaling and root planing complemented by simple or repeated laser phototherapy in chronic periodontitis. Lasers Med Sci [Epub ahead of print]. doi:10.1007/s10103-012-1104-5
Aimbire F, Lopes-Martins RA, Castro-Faria-Neto HC, Albertini R, Chavantes MC, Pacheco MT, Leonardo PS, Iversen VV, Bjordal JM (2006) Low-level laser therapy can reduce lipopolysaccharide-induced contractile force dysfunction and TNF-alpha levels in rat diaphragm muscle. Lasers Med Sci 21(4):238–244
de Lima FM, Villaverde AB, Albertini R, Correa JC, Carvalho RL, Munin E, Araujo T, Silva JA, Aimbire F (2011) Dual Effect of low-level laser therapy (LLLT) on the acute lung inflammation induced by intestinal ischemia and reperfusion: Action on anti- and pro-inflammatory cytokines. Lasers Surg Med 43(5):410–420. doi:10.1002/lsm.21053
Laraia EM, Silva IS, Pereira DM, Dos Reis FA, Albertini R, de Almeida P, Leal Junior EC, de Tarso Camillo de Carvalho P (2012) Effect of low-level laser therapy (660 nm) on acute inflammation induced by tenotomy of achilles tendon in rats. Photochem Photobiol. doi:10.1111/j.1751-1097.2012.01179.x, Epub ahead of print
de Lima FM, Moreira LM, Villaverde AB, Albertini R, Castro-Faria-Neto HC, Aimbire F (2011) Low-level laser therapy (LLLT) acts as cAMP-elevating agent in acute respiratory distress syndrome. Lasers Med Sci 26(3):389–400. doi:10.1007/s10103-010-0874-x
Francis J, Chu Y, Johnson AK, Weiss RM, Felder RB (2004) Acute myocardial infarction induces hypothalamic cytokine synthesis. Am J Physiol Heart Circ Physiol 286(6):H2264–H2271. doi:10.1152/ajpheart.01072.2003
Lopes R, Batista M Jr, Rosa J, Lira F, Martins E Jr, Yamashita A (2010) Changes in IL-10 and TNF-alpha skeletal muscle levels from rats with left ventricular disfunction after myocardial infarction. Arq Bras Cardiol 94(3):293–300. doi:10.1590/S0066-782X2010000300006
Kaur K, Sharma AK, Singal PK (2006) Significance of changes in TNF-alpha and IL-10 levels in the progression of heart failure subsequent to myocardial infarction. Am J Physiol Heart Circ Physiol 291(1):H106–H113. doi:10.1152/ajpheart.01327.2005
Aukrust P, Ueland T, Lien E, Bendtzen K, Muller F, Andreassen AK, Nordoy I, Aass H, Espevik T, Simonsen S, Froland SS, Gullestad L (1999) Cytokine network in congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol 83(3):376–382. doi:10.1016/S0002-9149(98)00872-8
Spate U, Schulze PC (2004) Proinflammatory cytokines and skeletal muscle. Curr Opin Clin Nutr Metab Care 7(3):265–269
Saghizadeh M, Ong JM, Garvey WT, Henry RR, Kern PA (1996) The expression of TNF alpha by human muscle. Relationship to insulin resistance. J Clin Invest 97(4):1111–1116. doi:10.1172/JCI118504
Toth MJ, Ades PA, Tischler MD, Tracy RP, LeWinter MM (2006) Immune activation is associated with reduced skeletal muscle mass and physical function in chronic heart failure. Int J Cardiol 109(2):179–187. doi:10.1016/j.ijcard.2005.06.006
Acknowledgments
We are thankful to Prof. Emerson Soldadeli Boschi, Tiago Heck MSc., Dr. Eurico Camargo Neto and André Costa for their support during the development of this study. This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brasília, Brazil. PROAP/UFCSPA.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hentschke, V.S., Jaenisch, R.B., Schmeing, L.A. et al. Low-level laser therapy improves the inflammatory profile of rats with heart failure. Lasers Med Sci 28, 1007–1016 (2013). https://doi.org/10.1007/s10103-012-1190-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-012-1190-4