Abstract
Introduction
Glucocorticoids are widely used to treat various diseases including rheumatoid arthritis (RA); however, one of the most frequent and severe adverse effects is glucocorticoid-induced osteoporosis (GIOP). Iguratimod (IGU) is a novel conventional synthetic disease-modifying anti-rheumatic drug developed in Japan. The aim of this study is to investigate the effects of IGU on glucocorticoid-induced disorder of bone metabolism in vitro.
Materials and methods
In osteoclastogenesis of mouse bone marrow-derived cells, tartrate-resistant acid phosphatase staining, resorption pit assay, western blotting, real-time polymerase chain reaction (PCR), and mRNA sequencing were performed. In osteoblastogenesis of MC3T3-E1 cells, alkaline phosphatase (ALP) staining and activity, alizarin red staining, and mRNA sequencing were performed, and real-time PCR and western blotting were conducted in MC3T3-E1 cells and murine osteocyte-like cell line MLO-Y4 cells.
Results
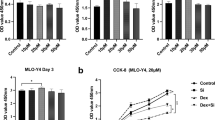
IGU significantly suppressed a dexamethasone-induced increase in osteoclasts, differentiation, and bone resorption activity by inhibition of the receptor activator of the nuclear factor kappa-B (RANK)/tumor necrosis factor receptor (TNFR)-associated factor 6 (TRAF6)/nuclear factor kappa-B (NFκB)-p52 pathway. In MC3T3-E1 cells, IGU significantly upregulated dexamethasone-induced downregulation of ALP activity, bone mineralization, and osteoblast-related gene and protein expression. In MLO-Y4 cells, IGU significantly upregulated dexamethasone-induced downregulation of the gene expression of ALP and osteocalcin, and also downregulated receptor activator of NFκB ligand (RANKL)/osteoprotegerin gene expression ratio without dexamethasone.
Conclusion
These results suggest that IGU may improve glucocorticoid-induced disorder of bone metabolism and may exhibit positive effects against GIOP associated with RA.




Similar content being viewed by others
References
Canalis E, Mazziotti G, Giustina A, Bilezikian JP (2007) Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 18:1319–1328. https://doi.org/10.1007/s00198-007-0394-0
Naganathan V, Jones G, Nash P, Nicholson G, Eisman J, Sambrook PN (2000) Vertebral fracture risk with long-term corticosteroid therapy: prevalence and relation to age, bone density, and corticosteroid use. Arch Intern Med 160:2917–2922. https://doi.org/10.1001/archinte.160.19.2917
Hansen KE, Kleker B, Safdar N, Bartels CM (2014) A systematic review and meta-analysis of glucocorticoid-induced osteoporosis in children. Semin Arthritis Rheum 44:47–54. https://doi.org/10.1016/j.semarthrit.2014.02.002
Buckley L, Guyatt G, Fink HA, Cannon M, Grossman J et al (2017) 2017 American College of Rheumatology Guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Rheumatol 69:1521–1537. https://doi.org/10.1002/art.40137
Russell RG, Watts NB, Ebetino FH, Rogers MJ (2008) Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int 19:733–759. https://doi.org/10.1007/s00198-007-0540-8
Smolen JS, Landewe R, Bijlsma J, Burmester G, Chatzidionysiou K et al (2017) EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 76:960–977. https://doi.org/10.1136/annrheumdis-2016-210715
Du F, Lu LJ, Fu Q, Dai M, Teng JL, Fan W, Chen SL, Ye P, Shen N, Huang XF, Qian J, Bao CD (2008) T-614, a novel immunomodulator, attenuates joint inflammation and articular damage in collagen-induced arthritis. Arthritis Res Ther 10:R136. https://doi.org/10.1186/ar2554
Tanaka K, Yamamoto T, Aikawa Y, Kizawa K, Muramoto K, Matsuno H, Muraguchi A (2003) Inhibitory effects of an anti-rheumatic agent T-614 on immunoglobulin production by cultured B cells and rheumatoid synovial tissues engrafted into SCID mice. Rheumatology (Oxford) 42:1365–1371. https://doi.org/10.1093/rheumatology/keg381
Kuriyama K, Higuchi C, Tanaka K, Yoshikawa H, Itoh K (2002) A novel anti-rheumatic drug, T-614, stimulates osteoblastic differentiation in vitro and bone morphogenetic protein-2-induced bone formation in vivo. Biochem Biophys Res Commun 299:903–909. https://doi.org/10.1016/s0006-291x(02)02754-7
Gan K, Yang L, Xu L, Feng X, Zhang Q, Wang F, Tan W, Zhang M (2016) Iguratimod (T-614) suppresses RANKL-induced osteoclast differentiation and migration in RAW264.7 cells via NF-kappaB and MAPK pathways. Int Immunopharmacol 35:294–300. https://doi.org/10.1016/j.intimp.2016.03.038
Wu YX, Sun Y, Ye YP, Zhang P, Guo JC, Huang JM, Jing XZ, Xiang W, Yu SY, Guo FJ (2017) Iguratimod prevents ovariectomy induced bone loss and suppresses osteoclastogenesis via inhibition of peroxisome proliferator activated receptor gamma. Mol Med Rep 16:8200–8208. https://doi.org/10.3892/mmr.2017.7648
Noguchi T, Ebina K, Hirao M, Morimoto T, Koizumi K, Kitaguchi K, Matsuoka H, Iwahashi T, Yoshikawa H (2017) Oxygen ultra-fine bubbles water administration prevents bone loss of glucocorticoid-induced osteoporosis in mice by suppressing osteoclast differentiation. Osteoporos Int 28:1063–1075. https://doi.org/10.1007/s00198-016-3830-1
Kohno M, Aikawa Y, Tsubouchi Y, Hashiramoto A, Yamada R, Kawahito Y, Inoue K, Kusaka Y, Kondo M, Sano H (2001) Inhibitory effect of T-614 on tumor necrosis factor-alpha induced cytokine production and nuclear factor-kappaB activation in cultured human synovial cells. J Rheumatol 28:2591–2596
Kaneshiro S, Ebina K, Shi K, Higuchi C, Hirao M, Okamoto M, Koizumi K, Morimoto T, Yoshikawa H, Hashimoto J (2014) IL-6 negatively regulates osteoblast differentiation through the SHP2/MEK2 and SHP2/Akt2 pathways in vitro. J Bone Miner Metab 32:378–392. https://doi.org/10.1007/s00774-013-0514-1
Kato Y, Windle JJ, Koop BA, Mundy GR, Bonewald LF (1997) Establishment of an osteocyte-like cell line, MLO-Y4. J Bone Miner Res 12:2014–2023. https://doi.org/10.1359/jbmr.1997.12.12.2014
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–D484. https://doi.org/10.1093/nar/gkm882
Franzoso G, Carlson L, Xing L, Poljak L, Shores EW, Brown KD, Leonardi A, Tran T, Boyce BF, Siebenlist U (1997) Requirement for NF-kappaB in osteoclast and B-cell development. Genes Dev 11:3482–3496. https://doi.org/10.1101/gad.11.24.3482
Zhu L, Chen J, Zhang J, Guo C, Fan W, Wang YM, Yan Z (2017) Parathyroid hormone (PTH) induces autophagy to protect osteocyte cell survival from dexamethasone damage. Med Sci Monit 23:4034–4040. https://doi.org/10.12659/msm.903432
Yu C, Huang D, Wang K, Lin B, Liu Y, Liu S, Wu W, Zhang H (2017) Advanced oxidation protein products induce apoptosis, and upregulate sclerostin and RANKL expression, in osteocytic MLO-Y4 cells via JNK/p38 MAPK activation. Mol Med Rep 15:543–550. https://doi.org/10.3892/mmr.2016.6047
Li CH, Ma ZZ, Jian LL, Wang XY, Sun L, Liu XY, Yao ZQ, Zhao JX (2020) Iguratimod inhibits osteoclastogenesis by modulating the RANKL and TNF-alpha signaling pathways. Int Immunopharmacol 90:107219. https://doi.org/10.1016/j.intimp.2020.107219
Yamashita T, Yao Z, Li F, Zhang Q, Badell IR, Schwarz EM, Takeshita S, Wagner EF, Noda M, Matsuo K, Xing L, Boyce BF (2007) NF-kappaB p50 and p52 regulate receptor activator of NF-kappaB ligand (RANKL) and tumor necrosis factor-induced osteoclast precursor differentiation by activating c-Fos and NFATc1. J Biol Chem 282:18245–18253. https://doi.org/10.1074/jbc.M610701200
Song J, Liu H, Zhu Q, Miao Y, Wang F, Yang F, Cheng W, Xi Y, Niu X, He D, Chen G (2018) T-614 promotes osteoblastic cell differentiation by increasing Dlx5 expression and regulating the activation of p38 and NF-kappaB. Biomed Res Int 2018:4901591. https://doi.org/10.1155/2018/4901591
Balani DH, Ono N, Kronenberg HM (2017) Parathyroid hormone regulates fates of murine osteoblast precursors in vivo. J Clin Invest 127:3327–3338. https://doi.org/10.1172/JCI91699
Tanaka T, Hoshijima M, Sunaga J, Nishida T, Hashimoto M, Odagaki N, Osumi R, Aadachi T, Kamioka H (2018) Analysis of Ca(2+) response of osteocyte network by three-dimensional time-lapse imaging in living bone. J Bone Miner Metab 36:519–528. https://doi.org/10.1007/s00774-017-0868-x
Humphrey EL, Williams JH, Davie MW, Marshall MJ (2006) Effects of dissociated glucocorticoids on OPG and RANKL in osteoblastic cells. Bone 38:652–661. https://doi.org/10.1016/j.bone.2005.10.004
Wei Y, Sun X, Hua M, Tan W, Wang F, Zhang M (2015) Inhibitory effect of a novel antirheumatic drug T-614 on the IL-6-induced RANKL/OPG, IL-17, and MMP-3 expression in synovial fibroblasts from rheumatoid arthritis patients. Biomed Res Int 2015:214683. https://doi.org/10.1155/2015/214683
Suematsu A, Tajiri Y, Nakashima T, Taka J, Ochi S, Oda H, Nakamura K, Tanaka S, Takayanagi H (2007) Scientific basis for the efficacy of combined use of antirheumatic drugs against bone destruction in rheumatoid arthritis. Mod Rheumatol 17:17–23. https://doi.org/10.1007/s10165-006-0531-1
Okada H, Kajiya H, Omata Y, Matsumoto T, Sato Y et al (2019) CTLA4-Ig directly inhibits osteoclastogenesis by interfering with intracellular calcium oscillations in bone marrow macrophages. J Bone Miner Res 34:1744–1752. https://doi.org/10.1002/jbmr.3754
Eastell R, Rosen CJ, Black DM, Cheung AM, Murad MH, Shoback D (2019) Pharmacological management of osteoporosis in postmenopausal women: an endocrine society* clinical practice guideline. J Clin Endocrinol Metab 104:1595–1622. https://doi.org/10.1210/jc.2019-00221
Acknowledgements
We would like to thank F. Hirayama and Y. Eguchi for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The iguratimod was kindly provided by Toyama Chemical Co., Ltd (Tokyo, Japan). K. Ebina has received research grants and lecture fee from Eisai Co., Ltd. K. Ebina and S. Oyama are affiliated with, and K. Nakata supervises the Department of Musculoskeletal Regenerative Medicine, Osaka University Graduate School of Medicine, which is supported by Taisho Pharmaceutical Co., Ltd. S. Oyama is an employee of Taisho Pharmaceutical Co., Ltd. These companies had no role in the study design, decision to publish, or preparation of the manuscript. A. Miyama, M. Hirao, G. Okamura, Y. Etani, K. Takami, A. Goshima, T. Miura, T. Kanamoto, and H. Yoshikawa declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
774_2021_1206_MOESM1_ESM.doc
Supplementary file1 Supplementary Fig. KEGG map of parathyroid hormone activated pathway in comparison with or without IGU in the absence of Dex. Red rectangles represent significantly upregulated factors. Dex, dexamethasone; IGU, iguratimod; PTH, parathyroid hormone; KEGG, Kyoto Encyclopedia of Genes and Genomes. (DOC 41 KB)
About this article
Cite this article
Miyama, A., Ebina, K., Hirao, M. et al. Effects of iguratimod on glucocorticoid-induced disorder of bone metabolism in vitro. J Bone Miner Metab 39, 639–648 (2021). https://doi.org/10.1007/s00774-021-01206-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-021-01206-5