Abstract
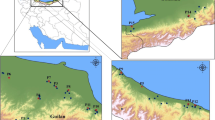
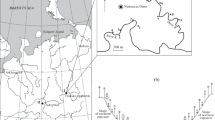
Seven populations of Hordelymus europaeus and four populations of Leymus arenarius from Poland were subjected to examination of 36 morphological characters. This study showed that both species are relatively uniform and that morphological variation of their populations represents a continuum. Of those, three populations of either species were selected for analysis with molecular markers – RAPDs and AFLPs. These populations differed with respect of geographical location as well as syntaxa and habitat. RAPD-PCR was performed for individual plants and clearly grouped them according to the population origin. For either H. europaeus or L. arenarius - the studied populations differed in degree of their intrinsic variation while none of them as a whole was significantly different from the remaining ones. In AFLP analysis the studied populations were represented by DNA pools of several individual plants. Also this approach allowed discrimination among the population samples of both H. europaeus and L. arenarius. Both RAPDs and AFLPs were accordant in indication that H. europaeus exceeds L. arenarius with respect to variation accumulated at the DNA level.
Similar content being viewed by others
References
Abbott LA, Bisby FA and Roger DJ (1985). Taxonomic analysis in biology. Computers, models and databases. Columbia University Press, New York, 311
Anamthawat-Jónsson K (2001). Genetic and genomic relationships in Leymus Hochst. Hereditas 135: 247–253
Anamthawat-Jónsson K, Bragason B Th, Bödvarsdóttir SK and Koebner RMD (1999). Molecular variation in Leymus species and populations. Molec Ecol 8: 309–315
Anamthawat-Jónsson K and Bödvarsdóttir SK (2001). Genomic and genetic relationships among species of Leymus (Poaceae: Triticeae) inferred from 18S–26S ribosomal genes. Amer J Bot 88(4): 553–559
Ascherson P, Graebner P (1898–1902) Synopsis der Mitteleuropäischen Flora. 2(1). W. Engelmann, Leipzig, pp 795.
Barkworth ME and Atkins RJ (1984). Leymus Hochst. (Gramineae: Triticeae) in North America: taxonomy and distribution. Amer J Bot 71: 609–625
Bothmer R von and Jacobsen N (1989). Intergeneric hybridization between Hordeum and Hordelymus (Poaceae). Nordic J Bot 9: 113–117
Bothmer R von, Lu B-R and Linde-Laursen I (1994). Intergeneric hybridization and C-banding patterns in Hordelymus (Triticeae, Poaceae). Pl Syst Evol 189: 259–266
Conert HJ (2000). Pareys Gräserbuch. Die Gräser Deutschlands erkennen und bestimmen. Parey Buchverlag, Berlin, 592
Felsenstein J (1989). PHYLIP: phylogeny interference package, version 3.2. Cladistics 5: 164–166
Gawal NJ and Jarret RL (1991). A modified CTAB DNA extraction procedure for Musa and Ipomea. Pl Molec Biol Rep 9: 262–266
Hammer Ř, Harper DAT and Ryan PD (2001). PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontologia Electronica 4(1): 9
Hitchcock CL, Cronquist A, Ownbey M and Thompson JW (1971). Vascular plants of the Pacific Northwest. Univ. of Washington Press, Seattle and London, 914
Hultén E and Fries M (1986). Atlas of North European vascular plants. North of the Tropic of Cancer. 3. Koelz Scientific Books, Königstein, 969–1172
Humphries CJ (1980). Hordelymus (Jessen) CO Harz. In: Tutin, TG, Heywood, VH, Burges, NA, Moore, DM, Valentine, DH, Walters, SM and Webb, DA (eds) Flora Europaea 5. Alismataceae to Orchidaceae (Monocotyledones), pp 205. Cambridge Univ. Press, Cambridge
Klimko M, Kreft A and Truchan M (2000). Interpopulation variability of Leymus arenarius (L.) Hochst. Rocz. AR Poznań 321. Bot 3: 155–163
Löve A (1982). Generic evolution of the wheatgrasses. Biol Zentralbl 101: 199–212
Macfarlane TD and Watson L (1982). The classification of Poaceae subfamily Pooideae. Taxon 31(2): 178–203
Matuszkiewicz W (2001). Przewodnik do oznaczania zbiorowisk roślinnych Polski. Państwowe Wyd. Naukowe, Warszawa, 298
Mizianty M (2001). The Agropyron-Elymus complex (Poaceae) in Poland: occurrence of Hordelymus europaeus. In: Frey, L (eds) Studies on the grasses in Poland, pp 161–176. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków
Mizianty M (2005). Variability and structure of natural populations of Elymus caninus (L.) L. based on morphology. Pl Syst Evol 251: 199–216
Mizianty M, Bieniek W, Czech A, Strzałka W and Szklarczyk M (2006). Variability and structure of natural populations of Elymus caninus (L.) L. and their possible relationship with Hordelymus europaeus (L.) Jess. ex Harz revealed by AFLP analysis. Pl Syst Evol 256: 193–200
Mizianty M, Frey L and Szczepaniak M (2001). The Agropyron − Elymus complex (Poaceae) in Poland: biosystematics. In: Frey, L (eds) Studies on grasses in Poland, pp 25–77. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków
Nei M and Li W-H (1979). Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76: 5269–5273
Nei M (1978). Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89: 583–590
Page RDM (1996). TREEVIEW: An application to display phylogenetic trees on personal computers. Comput Appl Biosci 12: 357–358
Sneath PH and Sokal RR (1973). Numerical taxonomy. The principles and practice of numerical classification. W. H. Freeman and Co., San Francisko, XV pp. + 573 pp
Stace CA (1991). New flora of the British Isles. Cambridge Univ. Press, Cambridge, XXX + 1226 pp
Svitashev S, Bryngelsson T, Xiaomei Li and Wang R (1998). Genome – specific repetitive DNA and RAPD markers for genome identification in Elymus and Hordelymus. Genome 41(4): 120–128
Szafer W (1977). Zasięgi geograficzne drzew oraz ważniejszych krzewów i krzewinek w Polsce. [Geographical limits of trees and important shrubs in Poland]. In: Szafer, W and Zarzycki, K (eds) Szata roślinna Polski. [The vegetation of Poland.] 2. Map inserted no 2, pp. Państwowe Wydawnictwo Naukowe, Warszawa
Tsvelev NN (1976). Zlaki SSSR [``Grasses of the USRR”]. Izdatel'stvo Nauka, Leningrad, 786 (in Russian)
Vershinin A, Svitashev S, Gummesson P-O, Salomon B, Bothmer R von and Bryngelsson T (1994). Characterisation of a family of tandemly repeated DNA sequences in Triticeae. Theor Appl Genet 89: 217–225
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M and Zabeau M (1995). AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23: 4407–4414
Waga J (1847). Flora polonica phanerogama sive descriptiones plantarum phanerogamicarum. 1. Typis S. Strąbski, Warszawa, 766
Wang RR-C and Jensen KB (1994). Absence of the J genome in Leymus species (Poaceae: Triticeae): evidence from DNA hybridization and meiotic pairing. Genome 37: 231–235
Yeh FC and Boyle TJB (1997). Population genetic analysis of co-dominant and dominant markers and quantitative traits. Belg J Bot 129: 157
Author information
Authors and Affiliations
Corresponding author
Additional information
It is the sixth paper of the series: Biodiversity of wild Triticeae (Poaceae) in Poland. The first is: M. Mizianty (2005). Variability and structure of natural populations of Elymus caninus (L.) L. based on morphology. Pl. Syst. Evol. 251: 199-216. The second: M. Mizianty, and M. Szklarczyk (2005). Systematic significance of Elymus caninus morphological characters revealed by AFLP analysis. In: L. Frey (ed.) Biology of grasses. W. Szafer Institute of Botany, Polish Academy of Sciences. Kraków, pp 9–21. The third: M. Mizianty et al. (2006). Variability and structure of natural populations of Elymus caninus (L.) L. and their possible relationship with Hordelymus europaeus (L.) Jess. ex Harz revealed by AFLP analysis. Pl. Syst. Evol. 256: 193–200. The fourth: M. Mizianty (2006). Variability and structure of natural populations of Hordeum murinum L. based on morphology. Pl. Syst. Evol. 261: 139–150. The fifth: B. Paszko Variability and structure of natural populations of Brachypodium pinnatum and B. sylvaticum based on morphology. Acta Soc. Bot. Pol. (in press).
Rights and permissions
About this article
Cite this article
Mizianty, M., Frey, L., Bieniek, W. et al. Variability and structure of natural populations of Hordelymus europaeus (L.) Jess. ex Harz and Leymus arenarius (L.) Hochst. as revealed by morphology and DNA markers. Plant Syst. Evol. 269, 15–28 (2007). https://doi.org/10.1007/s00606-007-0586-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-007-0586-2