Abstract
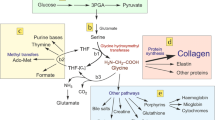
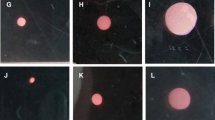
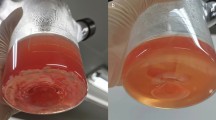
The functional integrity of articular cartilage is dependent on the maintenance of the extracellular matrix (ECM), a process which is controlled by chondrocytes. The regulation of ECM biosynthesis is complex and a variety of substances have been found to influence chondrocyte metabolism. In the present study we have investigated the effect of degraded collagen on the formation of type II collagen by mature bovine chondrocytes in a cell culture model. The culture medium was supplemented with collagen hydrolysate (CH) and biosynthesis of type II collagen by chondrocytes was compared to control cells treated with native type I and type II collagen and a collagen-free protein hydrolysate. The quantification of type II collagen by means of an ELISA technique was confirmed by immunocytochemical detection as well as by the incorporation of 14C-proline in the ECM after a 48 h incubation. Chondrocytes in the control group were maintained in the basal medium for 11 days. The presence of extracellular CH led to a dose-dependent increase in type II collagen secretion. However, native collagens as well as a collagen-free hydrolysate of wheat proteins failed to stimulate the production of type II collagen in chondrocytes. These results clearly indicate a stimulatory effect of degraded collagen on the type II collagen biosynthesis of chondrocytes and suggest a possible feedback mechanism for the regulation of collagen turnover in cartilage tissue.






Similar content being viewed by others
References
Adyelotte MB, Schmid TR, Greenhill RR, Luchene L, Schumacher BL, Kuettner KE (1991) Synthesis of collagen by cultured bovine chondrocytes derived from different depth of articular cartilage. Trans Orthop Res Soc 16:26
Aigner T, Glückert K, von der Mark K (1997) Activation of fibrillar collagen synthesis and phenotypic modulation of chondrocytes in early human osteoarthritic lesions. Osteoarthritis Cartilage 5:183–189
Aycock RS, Raghow R, Stricklin GP, Seyer JM, Kang AH (1986) Post-transcriptional inhibition of collagen and fibronectin synthesis by a synthetic homolog of a portion of the carboxyl-terminal propeotide of human type I collagen. J Biol Chem 261:14355–14360
Bateman JF, Lamandé SR, Ramshaw JAM (1996) Collagen superfamily. In: Comper WD (ed) Extracellular matrix, vol 2. Harwood Academic, Amsterdam, pp 22–67
Benya PD, Padilla SR, Nimni ME (1987) Independent regulation of collagen types by chondrocytes during the loss of differentiated function in culture. Cell 15:1313–1321
Benz K, Breit S, Lukoschek M, Mau H, Richter W (2002) Molecular analysis of expansion, differentiation, and growth factor treatment of human chondrocytes identifies differentiation markers and growth-related genes. Biochem Biophys Res Commun 293:284–292
Buckwalter JA, Mankin HJ (1998) Articular cartilage: degeneration and osteoarthritis, repair, regeneration, and transplantation. Instr Course Lect 47:487–504
Burstein D, Bashir A, Gray ML (2000) MRI techniques in early stages of cartilage disease. Invest Radiol 35:602–621
Elima K, Vuorio E (1989) Expression of mRNAs for collagen and other matrix components in dedifferentiating and redifferentiating human chondrocytes in culture. FEBS Lett 258:195–198
Eyre DR, Wu JJ, Woods PE (1991) The cartilage collagens: structural and metabolic studies. J Rheumatol 18:49–51
Hardingham TE, Bayliss M (1990) Proteoglycans of articular cartilage: changes in aging and in joint disease. Semin Arthritis Rheum 3:12–33
Huber M, Tratting S, Lintner F (2000) Anatomy, biochemistry, and physiology of articular cartilage. Invest Radiol 35:573–580
Katayama K, Seyer JM, Raghow R, Kang AH (1991) Regulation of extracellular matrix production by chemically synthesized subfragments of type I collagen carboxy propeptide. Biochemistry 30:7097–7104
Lotz M, Blanco FJ, von Kempis J, et al. (1995) Cytokine regulation of chondrocyte functions. J Rheumatol 43:104–108
Mayne R (1989) Collagens: what is their function, are they involved in articular disease. Arthritis Rheum 32:241–246
Mlynarik V, Tratting S (2000) Physicochemical properties of normal articular cartilage and its MR appearance. Invest Radiol 35:589–594
Moskowitz RW (2000) Role of collagen hydrolysate in bone and joint disease. Semin Arthritis Rheum 30:87–99
Muir H (1995) The chondrocyte, architect of cartilage: biomechanics, structure, function and molecular biology of cartilage matrix macromolecules. Bioessays 17:1039–1048
Oesser S, Adam M, Babel W, Seifert J (1999) Oral administration of 14C labeled gelatin hydrolysate leads to an accumulation of radioactivity in cartilage of mice (C57/BL). J Nutr 129:1891–1895
Osborne KD, Trppel SB, Mankin HJ (1989) Growth factor stimulation of adult articular cartilage. J Orthop Res 7:35–42
Pasternak RD, Hubbs SJ, Caccese RG, Marks RL, Conaty JM, DiPasquale G (1986) Interleukin-1 stimulates the secretion of proteoglycan- and collagen-degrading proteases by rabbit articular chondrocytes. Clin Immunol 41:351–367
Phadke K, Nanda S (1983) Secretion of chondrocyte stimulating factor by macrophages as a result of activation with collagen and proteoglycans. Clin Exp Immunol 51:494–500
Qi WN, Scully SP (1997) Extracellular collagen modulates the regulation of chondrocytes by transforming growth factor-β1. J Orthop Res 15:483–490
Rosier RN, O'Keefe RJ (1998) Autocrine regulation of articular cartilage. Instr Course Lect 47:469–475
Roughley PJ, Lee ER (1994) Cartilage proteoglycans: structure and potential function. Microsc Res Technol 28:385–397
Roughley PJ, Nguyen Q, Mort SM (1991) Mechanisms of proteoglycan degradation in human articular cartilage. J Rheum 18:52–54
Sandy PJ, Adams ME, Billingham MEJ, Plaas A, Muir H (1984) In vivo and in vitro stimulation of chondrocyte biosynthetic activity in early experimental osteoarthritis. Arthritis Rheum 27:388–397
Sklatvala J (1986) Tumor necrosis factor-a stimulates resorption and inhibits synthesis of proteoglycan in cartilage. Nature 322:547–549
Trippel SB (1995) Growth factor actions on articular cartilage. J Rheumatol 43:129–132
von der Mark K, Gauss V, von der Mark H, Müller P (1977) Relationship between cell shape and type of collagen synthesised as chondrocytes lose their cartilage phenotype in culture. Nature 267:531–532
Zaucke F, Dinser R, Maurer P, Paulsen M (2001) Cartilage oligomeric matrix protein (COMP) and collagen IX are sensitive markers for the differentiation state of articular primary chondrocytes. Biochem J 258:17–24
Acknowledgements
We wish to thank Anja Raabe for her excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
We wish to thank the DGF Stoess AG for supporting this study.
Rights and permissions
About this article
Cite this article
Oesser, S., Seifert, J. Stimulation of type II collagen biosynthesis and secretion in bovine chondrocytes cultured with degraded collagen. Cell Tissue Res 311, 393–399 (2003). https://doi.org/10.1007/s00441-003-0702-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-003-0702-8