Abstract
Ethylene receptors are key factors for ethylene signal transduction. In tomato, six ethylene receptor genes (SlETR1–SlETR6) have been identified. Mutations in different ethylene receptor genes result in different phenotypes that are useful for elucidating the roles of each gene. In this study, we screened mutants of two ethylene receptor genes, SLETR4 and SLETR5, from a Micro-Tom mutant library generated by TILLING. We identified two ethylene receptor mutants with altered phenotypes and named them Sletr4-1 and Sletr5-1. Sletr4-1 has a mutation between the transmembrane and GAF domains, while Sletr5-1 has a mutation within the GAF domain. Sletr4-1 showed increased hypocotyl and root lengths, compared to those of wild type plants, under ethylene exposure. Moreover, the fruit shelf life of this mutant was extended, titratable acidity was increased and total soluble solids were decreased, suggesting a reduced ethylene sensitivity. In contrast, in the absence of exogenous ethylene, the hypocotyl and root lengths of Sletr5-1 were shorter than those of the wild type, and the fruit shelf life was shorter, suggesting that these mutants have increased ethylene sensitivity. Gene expression analysis showed that SlNR was up-regulated in the Sletr5-1 mutant line, in contrast to the down-regulation observed in the Sletr4-1 mutant line, while the down-regulation of SlCTR1, SlEIN2, SlEIL1, SlEIL3, and SlERF.E4 was observed in Sletr4-1 mutant allele, suggesting that these two ethylene receptors have functional roles in ethylene signalling and demonstrating, for the first time, a function of the GAF domain of ethylene receptors. These results suggest that the Sletr4-1 and Sletr5-1 mutants are useful for elucidating the complex mechanisms of ethylene signalling through the analysis of ethylene receptors in tomato.









Similar content being viewed by others
References
Abeles FB, Morgan PW, Saltveit ME (1992) Ethylene in Plant Biology 2nd ed. Academic Press, Sandiego. ISBN:0-12-041451-1, pp 83–103
Alexander L, Grierson D (2002) Ethylene biosynthesis and action in tomato: a model for climacteric fruit ripening. J Exp Bot 53:2039–2055
Alonso JM, Hirayama T, Roman G, Nourizadeh S, Ecker JR (1999) EIN2, a bifunctional transducer of ethylene and stress responses in Arabidopsis. Science 284:2148–2152
Aravind L, Ponting CP (1997) The GAF domain: An evolutionary link between diverse phototransducing proteins. Trends Biochem Sci 22:458–459
Baldwin EA, Biggs RH (1988) Cell-wall lysing enzymes and products of cell-wall digestion elicit ethylene in citrus. Physiol Plant 73:58–64
Blecker AB, Patterson SE (1997) Last exit: senescence, abscission, and meristem arrest in Arabidopsis. Plant Cell 9:1169–1179
Brummell DA, Harpster MH (2001) Cell wall metabolism in fruit softening and quality and its manipulation in transgenic plants. Plant Mol Biol 47:311–340
Cameron AC, Reid MS (2001) 1-MCP blocks ethylene induced petal abscission of Pelargonium peltatum but the effect is transients. Postharvest Biol Technol 22:169–177
Carrari F, Fernie AR (2006) Metabolic regulation underlying tomato fruit development. J Exp Bot 57:1883–1897
Choi SW, Hoshikawa K, Fujita S, Thi DP, Mizoguchi T, Ezura H, Ito E (2018) Evaluation of internal control genes for quantitative realtime PCR analyses for studying fruit development of dwarf tomato cultivar ‘Micro-Tom’. Plant Biotech. 18–0525
Ciardi J, Klee H (2001) Regulation of ethylene-mediated responses at the level of the receptor. Ann Bot 88:813–822
Crocker W, Knight LI, Rose RCA (1913) Delicate seedling test. Science 37:380–381
Crouch I (2003) 1-Methylcyclopropene (Smartfreshtm) as an alternative to modified atmosphere and controlled atmosphere storage of apples and pears. Acta Horticlturae 600:433–436
Defilippi BG, Dandekar AM, Kader AA (2004) Impact of suppression of ethylene action and biosynthesis on flavor metabolites in apple (Malus domestica Borkh) fruits. J Agric Food Chem 52:5694–5701
Evensen KB (1991) Ethylene responsiveness changes in Pelargonium X domesticum. Physiol. Plant 82:409–412
Gamble RL, Coonfield ML, Schaller GE (1998) Histidine kinase activity of the ETR1 ethylene receptor from Arabidopsis. Proc Natl Acad Sci USA, 95:7825–7829
Gao L, Zhao W, Qu H, Wang Q, Zhao L (2016) The yellow-fruited tomato 1 (yft1) mutant has altered fruit carotenoid accumulation and reduced ethylene production as a result of a genetic lesion in ETHYLENE INSENSITIVE2. Theor Appl Genets 129(4):717–728
Giovannoni JJ (2004) Genetic regulation of fruit development and ripening. Plant Cell (Suppl) 16:S170–S180
Guzmán P, Ecker JR (1990) Exploiting the triple response of Arabidopsis to identify ethylene-related mutants. Plant Cell 2:513–524
Hobson GE (1980) Effect of the introduction of non-ripening mutant genes on the composition and enzyme content of tomato fruit. J Sci Food Agric 31:578–584
Jones ML, Kim ES, Newman SE (2001) Role of ethylene and 1-MCP in flower development and petal abscission in zonal Pelargonium. Hort Sci 36:1305–1309
Kende H (1993) Ethylene biosynthesis. Ann Rev Plant Physiol Plant Mol Biol 44:283–307
Kevany BM, Tieman DM, Taylor MG, Cin VD, Klee HJ (2007) Ethylene receptor degradation controls the timing of ripening in tomato fruit. Plant J 51:458–467
Kieber JJ, Rothenberg M, Roman G, Feldmann KA, Ecker JR (1993) CTR1, a negative regulator of the ethylene response pathway in Arabidopsis, encodes a member of the Raf family of protein kinases. Cell 72:427–441
Klay I, Gouia S, Liu M, Mila I, Khoudi H, Bernadec A, Bouzayen M, Pirello J (2018) Ethylene Response Factors (ERF) are differentially regulated by different abiotic stress types in tomato plants. Plant Sci 274:137–145
Klee H, Tieman D (2002) The tomato ethylene receptor gene family: form and function. Physiol Plant 115:336–341
Lanahan MB, Yen HC, Giovannoni JJ, Klee HJ (1994) The never ripe mutation blocks ethylene perception in tomato. Plant Cell 6:521–530
Lashbrook CC, Tieman DM, Klee HJ (1998) Differential regulation of the ETR gene family throughout plant development. Plant J 15:234–252
Leclercq J, Adams-Phillips LC, Zegzouti H, Jones B, Latché A, Giovannoni JJ, Pech JC, Bouzayen M (2002) LeCTR1, a tomato CTR1-like gene, demonstrates ethylene signaling ability in arabidopsis and novel expression patterns in tomato. Plant Physiol 130:1132–1142
Mubarok S, Okabe Y, Fukuda N, Ariizumi T, Ezura H (2015) Potential use of a weak ethylene receptor mutant, Sletr1-2, as breeding material to extend fruit shelf life of tomato. J Agric Food Chem 63:7995–8007
Mubarok S, Okabe Y, Fukuda N, Ariizumi T, Ezura H (2016) Favourable effect of a weak ethylene receptor mutation Sletr1-2 on postharvest fruit quality changes in tomatoes. Postharvest Biol Technol 120:1–9
Okabe Y, Asamizu E, Saito T, Matsukura C, Ariizumi T, Bres C, Rothan C, Mizaguchi T, Ezura H (2011) Tomato TILLING Technology: Development of a reverse genetic tool for the efficient isolation of mutants from micro-tom mutant libraries. Plant Cell Physiol 52:1994–2005
Payton S, Fray RG, Brown S, Grierson D (1996) Ethylene receptor expression is regulated during fruit ripening, flower senescence and abscission. Plant Mol Biol 31:1227–1231
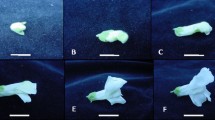
Representative images showing the appearance (upper) and leaves (lower) of the two ethylene receptor mutants. The plant and leaves were taken at 60 days after sowing
Rick C, Butler L (1956) Cytogenetics of the Tomato. Adv. Genet 8:267–382
Rodríguez GR, Pratta GR, Liberatti DR, Zorzoli R, Picardi LA (2010) Inheritance of shelf life and other quality traits of tomato fruit estimated from F1’s, F2’s and backcross generatios derived from standard cultivar, nor homozygote and wild cherry tomato. Euphytica 176:137–147
Saito T, Ariizumi T, Okabe Y, Asamizu E, Hiwasa-Tanase K, Fukuda N, Mizoguchi T, Yamazaki Y, Aoki K, Ezura H (2011) TOMATOMA: a novel tomato mutant database distributing Micro-Tom mutant collections. Plant Cell Physiol 52:283–296
Sakai H, Hua J, Chen Q, Chang C, Medrano L, Bleecker A, Meyerowitz E (1998) ETR2 is an ETR1-like gene involved in ethylene signaling in Arabidopsis. Proc Natl Acad Sci USA 95:5812–5817
Shinozaki Y, Hao S, Kojima M, Sakakibara H, Ozeki-Iida Y, Zheng Y, Fei Z, Zhong S, Giovannoni JJ, Rose JKC, Okabe Y, Heta Y, Eura H, Ariizumi T (2015) Ethylene suppresses tomato (Solanum lycopersicum) fruit set through modification of gibberellin metabolism. Plant J 83:237–251
Tieman DM, Klee HJ (1999) Differential expression of two novel members of the tomato ethylene-receptor family. Plant Physiol 120:165–172
Tieman DM, Taylor MG, Ciardi JA, Klee HJ (2000) The tomato ethylene receptors NR and LeETR4 are negative regulators of ethylene response and exhibit functional compensation within a multigene family. Proc Natl Acad Sci USA 97:5663–5668
Tieman DM, Ciardi JA, Taylor MG, Klee HJ (2001) Members of the tomato LeEIL (EIN3-like) gene family are functionally redundant and regulate ethylene responses throughout plant development. Plant J 26:47–58
Wang KLC, Li H, Ecker JR (2002) Ethylene biosynthesis and signaling networks. Plant Cell S131–S151
Watanabe S, Mizoguchi T, Aoki K, Kubo Y, Mori H, Imanishi S, Yamazaki Y, Shibata D, Ezura H (2007) Ethylmethanesulfonate (EMS) mutagenesis of Solanum lycopersicum cv. Micro-Tom for large-scale mutant screens. Plant Biotechnol 24:33–38
Wilkinson J, Lanahan M, Yen H, Giovannoni J, Klee H (1995) An ethylene-inducible component of signal transduction encoded by Never-ripe. Science 270:1807–1809
Winsor GW, Davies JN, Massey DM (1962) Composition of tomato fruit. IV. changes in some constituents of the fruit walls during ripening. J Sci Food Agricult 13(3):141–145
Yang SF, Hoffman NE (1984) Ethylene biosysnthesis and its regulation in higher plants. Ann Rev Plant Physiol 35:155–189
Yang X, Song J, Campbell-Palmer L, Fillmore S, Zhang Z (2013) Effect of ethylene and 1-MCP on expression of genes involved in ethylene biosynthesis and perception during ripening of apple fruit. Postharvest BiolTechnol 78:55–66
Zhang ZG, Zhou HL, Chen T, Gong Y, Cao WH, Wang YJ, Zhang JS, Chen SY (2004) Evidence for serine/threonine and histidine kinase activity in the tobacco ethylene receptor protein NTHK2. Plant Physiol 136:2971–2981
Zhou HL, Cao WH, Cao YR, Liu J, Hao YJ, Zhang JS, Chen SY (2006) Roles of ethylene receptor NTHK1 domains in plant growth, stress response and protein phosphorylation. FEBS Lett 580:1239–1250
Acknowledgements
We thank the National BioResearch Project (NBRP), MEXT, Japan for providing seeds from S. lycopersicum cv. Micro-Tom, Sletr4-1,and Sletr5-1. This study was supported by the JSPS KAKENHI [grant number25252008] to H.E. We also thank all members of our laboratory for their helpful discussions throughout the project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mubarok, S., Hoshikawa, K., Okabe, Y. et al. Evidence of the functional role of the ethylene receptor genes SlETR4 and SlETR5 in ethylene signal transduction in tomato. Mol Genet Genomics 294, 301–313 (2019). https://doi.org/10.1007/s00438-018-1505-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-018-1505-7