Abstract
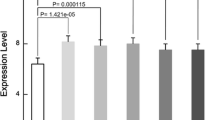
The purpose of this study was to use a cDNA microarray to identify new genes involved in healing of spinal cord injury. C57BL/6 mice (7–8 weeks, male) were subjected to spinal cord compression injury (SCI) at the T7/8 level (20 g, 5 min; SCI group). For the control group, mice underwent only laminectomy. Mice were killed at 1, 3 and 7 days. cDNA transcribed from mRNA was hybridized to NIA mice 15K microarrays at each time point. We found 84 genes showing significant expressional changes, including higher and lower expression levels in the SCI groups than in the control [more than 1.0 or less than −1.0 using log ratio (base 2)]. Five genes were selected for further quantitative gene expression analysis by real-time reverse transcription (RT)-PCR. For histological examination, we applied in situ hybridization and fluorescence immunohistochemistry. Cathepsin D, metallothionein-1 (MT-1), metallothionein-2 (MT-2), osteopontin (OPN), and tenascin-C were selected for quantitative and histological analysis. Microarray analysis revealed that SCI led to the up-regulation of OPN and cathepsin D expression at 7 days and also of MT-1, MT-2, and tenascin-C expression at 1 day. Tenascin-C was re-up-regulated at 7 days. These values agreed with those of real-time RT-PCR analysis. By double labeling with in situ hybridization and fluorescence immunohistochemistry, MT-1, MT-2 and tenascin-C expression was observed in neurons and glial cells at 1 day, whereas at 7 days the main MT-2 and tenascin-C expression was found in fibronectin-positive fibroblasts. The main cathepsin D and OPN expression was observed in activated macrophages/microglia at 3 and 7 days. The five genes picked up by microarray gene expression profiling were shown to exhibit temporal and spatial changes of expression after SCI. This system is potentially useful for identifying genes that are involved in the response to SCI.








Similar content being viewed by others
References
Abe Y, Nakamura H, Yoshino O, Oya T, Kimura T (2003) Decreased neural damage after spinal cord injury in tPA-deficient mice. J Neurotrauma 20:43–57
Abel J, Ruiter N de (1989) Inhibition of hydroxyl-radical-generated DNA degradation by metallothionein. Toxicol Lett 47:191–196
Baldwin SA, Broderik R, Blades DA, Scheff SW (1998) Alterations in temporal/spatial distribution of GFAP- and vimentin-positive astrocytes after spinal cord contusion with the New York University spinal cord injury device. J Neurotrauma 15:1015–1026
Banik NL, Hogan EL, Powers JM, Smith KP (1986) Proteolytic enzymes in experimental spinal cord injury. J Neurol Sci 73:245–256
Blight AR (1985) Delayed demyelination and macrophage invasion: a candidate for secondary cell damage in spinal cord injury. Cent Nerv Syst Trauma 2:299–315
Blum JS, Fiani ML, Stahl PD (1991) Proteolytic cleavage of ricin A chain in endosomal vesicles. Evidence for the action of endosomal proteases at both neutral and acidic pH. J Biol Chem 266:22091–22095
Boehme DH, Umezawa H, Hashim G, Marks N (1978) Treatment of experimental allergic encephalomyelitis with an inhibitor of cathepsin D (pepstatin). Neurochem Res 3:185–194
Brostoff SW, Reuter W, Hichens M, Eylar EH (1974) Specific cleavage of the A1 protein from myelin with cathepsin D. J Biol Chem 249:559–567
Campagne MV, Thibodeaux H, Bruggen N van, Cairns B, Gerlai R, Palmer JT, Williams SP, Lowe DG (1999) Evidence for protective role of metallothionein-1 in focal cerebral ischemia. Proc Natl Acad Sci USA 96:12870–12875
Campagne MV, Thibodeaux H, Bruggen N van, Cairns B, Lowe DG (2000) Increased binding activity at an antioxidant-responsive element in the metallothionein-1 promoter and rapid induction of metallothionein-1 and −2 in response to cerebral ischemia and reperfusion. J Neurosci 20:5200–5207
Carlson SL, Parish ME, Springer JE, Doty K, Dossett L (1998) Acute inflammatory response in spinal cord following impact injury. Exp Neurol 151:77–88
Carmel JB, Galante A, Soteropoulos P, Tolias P, Recce M, Young W, Hart RP (2001) Gene expression profiling of acute spinal cord injury reveals spreading inflammatory signals and neuron loss. Physiol Genomics 7:201–213
Carmel JB, Kakinohana O, Mestril R, Young W, Marsala M, Hart RP (2004) Mediators of ischemic preconditioning identified by microarray analysis of rat spinal cord. Exp Neurol 185:81–96
Cassina P, Peluffo H, Pehar M, Martinez-Palma L, Ressia A, Beckman JS, Estevez AG, Barbeito L (2002) Peroxynitrite triggers a phenotypic transformation in spinal cord astrocytes that induces motor neuron apotosis. J Neurosci Res 67:21–29
Deckner M, Lindholm T, Cullheim S, Risling M (2000) Differential expression of tenascin-C, tenascin-R, tenascin/J1, and tenascin-X in spinal cord scar tissue and in the olfactory system. Exp Neurol 166:350–362
Deiss LP, Galinka H, Berissi H, Cohen O, Kimchi A (1996) Cathepsin D protease mediates programmed cell death induced by interferon-gamma, Fas/APO-1 and TNF-alpha. EMBO J 15:3861–3870
Farooque M, Hillered L, Holtz A, Olsson Y (1996) Effects of methylprednisolone on extracellular lactic acidosis and amino acids after severe compression injury of rat spinal cord. J Neurochem 66:1125–1130
Fawcett JW, Asher RA (1999) The glial scar and central nervous system repair. Brain Res Bull 49:377–391
Ferhat L, Chevassus au Louis N, Jorquera I, Niquet J, Khrestchatisky M, Ben-Ari Y, Represa A (1996) Transient increase of tenascin-C in immature hippocampus: astroglial and neuronal expression. J Neurocytol 25:53–66
Fu Y, Hashimoto M, Ino H, Murakami M, Yamazaki M, Moriya H (2003) Spinal root avulsion–induced up-regulation of osteopontin expression in the adult rat spinal cord. Acta Neuropathol 107:8–16
Gasull T, Giralt M, Garcia A, Hidalgo J (1994) Regulation of metallothionein-I+II levels in specific brain areas and liver in the rat: role of catecholamines. Glia 12:135–143
Giralt M, Penkowa M, Lago N, Molinero A, Hidalgo J (2002) Metallothionein-1+2 protect the CNS after a focal brain injury. Exp Neurol 173:114–128
Hashimoto M, Koda M, Ino H, Murakami M, Yamazaki M, Moriya H (2003) Upregulation of osteopontin expression in rat spinal cord microglia after traumatic injury. J Neurotrauma 20:287–296
Hellmich HL, Frederickson CJ, DeWitt DS, Saban R, Parsley MO, Stephenson R, Velasco M, Uchida T, Shimamura M, Prough DS (2004) Protective effects of zinc chelation in traumatic brain injury correlate with upregulation of neuroprotective genes in rat brain. Neurosci Lett 355:221–225
Herschman HR (1985) A 12-O-tetradecanoylphorbol-13-acetate (TPA)-nonproliferative variant of 3T3 cells is resistant to TPA-enhanced gene amplification. Mol Cell Biol 5:1130–1135
Ikeda O, Murakami M, Ino H, Yamazaki M, Nemoto T, Koda M, Nakayama C Moriya H (2001) Acute up-regulation of brain-derived neurotrophic factor expression resulting from experimentally induced injury in the rat spinal cord. Acta Neuropathol 102:239–245
Ikeda O, Murakami M, Ino H, Yamazaki M, Koda M, Nakayama C, Moriya H (2002) Effects of brain-derived neurotrophic factor (BDNF) on compression-induced spinal cord injury: BDNF attenuates down-regulation of superoxide dismutase expression and promotes up-regulation of myelin basic protein expression. J Neuropathol Exp Neurol 61:142–153
Kato H, Kanellopoulos GK, Matsuo S, Wu YJ, Jacquin MF, Hsu CY, Kouchoukos NT, Choi DW (1997) Neuronal apoptosis and necrosis following spinal cord ischemia in the rat. Exp Neurol 148:464–474
Kenessy A, Banay-Schwartz M, DeGuzman T, Lajtha A (1989) Increase in cathepsin D activity in rat brain in aging. J Neurosci Res 23:454–456
Koike M, Nakanishi H, Saftig P, Ezaki J, Isahara K, Ohsawa Y, Schulz-Schaeffer W, Watanabe T, Waguri S, Kametaka S, Shibata M, Yamamoto K, Kominami E, Peters C, Figura K von, Uchiyama Y (2000) Cathepsin D deficiency induces lysosomal storage with ceroid lipofuscin in mouse CNS neurons. J Neurosci 15:6898–6906
Kondo Y, Rusnak JM, Hoyt DG, Settineri CE, Pitt BR, Lazo JS (1997) Enhanced apoptosis in metallothionein null cells. Mol Pharmacol 52:195–201
Laywell ED, Dorries U, Bartsch U, Faissner A, Schachner M, Steindler DA (1992) Enhanced expression of the developmentally regulated extracellular matrix molecule tenascin following adult brain injury. Proc Natl Acad Sci USA 89:2634–2638
Liu XZ, Xu XM, Hu R, Du C, Zhang SX, McDonald JW, Dong HX, Wu YJ, Fan GS, Jacquin MF, Hsu CY, Choi DW (1997) Neuronal and glial apoptosis after traumatic spinal cord injury. J Neurosci 17:5395–5406
Long Y, Zou L, Liu H, Lu H, Yuan X, Robertson CS, Yang K (2003) Altered expression of randomly selected genes in mouse hippocampus after traumatic brain injury. J Neurosci Res 71:710–720
Matsuda A, Yoshiki A, Tagawa Y, Matsuda H, Kusakabe M (1999) Corneal wound healing in tenascin knockout mouse. Invest Ophthalmol Vis Sci 40:1071–1080
McKee MD, Nanci A (1996) Secretion of osteopontin by macrophages and it’s accumulation at tissue surfaces during wound healing in mineralized tissues : a potential requirement for macrophage adhesion and phagocytosis. Anat Rec 245:394–409
Montoliu C, Monfort P, Carrasco J, Palacios O, Capdevila M, Hidalgo J, Felipo V (2000) Metallothionein-III prevents glutamate and nitric oxide neurotoxicity in primary cultures of cerebellar neurons. J Neurochem 75:266–273
Mufson EJ, Counts SE, Ginsberg SD (2002) Gene expression profiles of cholinergic nucleus basalis neurons in Alzheimer’s disease. Neurochem Res 27:1035–1048
Pan JZ, Ni L, Sodhi A, Aguanno A, Young W, Hart RP (2002) Cytokine activity contributes to induction of inflammatory cytokine mRNAs in spinal cord following contusion. J Neurosci Res 68:315–322
Pearson CA, Pearson D, Shibahara S, Hofsteenge J, Chiquet-Ehrismann R (1988) Tenascin: cDNA cloning and induction by TGF-beta. EMBO J 7:2977–2982
Penkowa M, Hidalgo J (2003) Treatment with metallothionein prevents demyelination and axonal damage and increases oligodendrocyte precursors and tissue repair during experimental autoimmune encephalomyelitis. J Neurosci Res 72:574–586
Reinholt FP, Hultenby K, Oldberg A, Heinegard D (1990) Osteopontin—a possible anchor of osteoclasts to bone. Proc Natl Acad Sci USA 87:4473–4475
Rollo EE, Laskin DL, Denhardt DT (1996) Osteopontin inhibits nitric oxide production and cytotoxicity by activated RAW264.7 macrophages. J Leukoc Biol 60:397–404
Sasagawa S, Matsubara J, Satow Y (1993) Stress-related induction of hepatic metallothionein synthesis and increase in peripheral polymorphonuclear leukocytes in mice. Immunopharmacal Immunotoxicol 15:217–226
Scatena M, Almeida M, Chaisson ML, Fausto N, Nicosia RF, Giachelli CM (1998) NF-κB mediates αvβ3 integrin-induced endothelial cell survival. J Cell Biol 141:1083–1093
Schmidt C, Beyersmann D (1999) Transient peaks in zinc and metallothionein levels during differentiation of 3T3L1 cells. Arch Biochem Biophys 364:91–98
Schnell L, Fearn S, Klassen H, Schwab ME, Perry VH (1999) Acute inflammatory responses to mechanical lesions in the CNS: differences between brain and spinal cord. Eur J Neurosci 11:3648–3658
Senger DR, Ledbetter SR, Claffey KP (1996) Stimulation of endothelial cell migration by vascular permeability factor/vascular endothelial growth factor through cooperative mechanisms involving the αvβ3 integrin osteopontin, and thrombin. Am J Pathol 149:293–305
Smith GM, Hale JH (1997) Macrophage/microglia regulation of astrocytic tenascin: synergistic action of transforming growth factor-beta and basic fibroblast growth factor. J Neurosci 17:9624–9633
Tanaka TS, Jaradat SA, Lim MK, Kargul GJ, Wang X, Grahovac MJ, Pantano S, Sano Y, Piao Y, Nagaraja R, Doi H, Wood III WH, Becker KG, Ko MSH (2000) Genome-wide expression profiling of mid-gestation placenta and embryo using a 15000 mouse developmental cDNA microarray. Proc Natl Acad Sci USA 97:9127–9132
Tang X, Davies JE, Davies SJA (2003) Changes in distribution, cell association, and protein expression levels of NG2, neurocan, phosphacan, brevican, versican V2, and tenascin-C during acute to chronic maturation of spinal cord scar tissue. J Neurosci Res 71:427–444
Terai K, Takano-Yamamoto T, Ohba Y, Hiura K (1999) Role of osteopontin in bone remodeling caused by mechanical stress. J Bone Miner Res 14:839–849
Tsangaris GT, Tzortzatou-Stathopoulou F (1998) Metallothionein expression prevents apoptosis: a study with antisense phosphorothioate oligodeoxynucleotides. Anticancer Res 18:2423-2434
Wada S, Yone K, Ishidou Y, Nagamine T, Nakahara S, Niiyama T, Sakou T (1999) Apoptosis following spinal cord injury in rats and preventative effect of N-methyl-d-aspartate receptor antagonist. J Neurosurg 91(Suppl 1):98–104
Wallner K, Li C, Shah PK, Fishbein MC, Forrester JS, Kaul S, Sharifi BG (1999) Tenascin-C is expressed in macrophage-rich human coronary atherosclerotic plaque. Circulation 99:1284–1289
Wang XK, Louden C, Yue TL, Ellison JA, Barone FC (1998) Delayed expression of osteopontin after focal stroke in the rat. J Neurosci 18:2075–2083
Weber GF, Zawaideh S, Hikita S, Kumar VA, Cantor H, Ashkar S (2002) Phosphorylation-dependent interaction of osteopontin with its receptors regulates macrophage migration and activation. J Leukoc Biol 72:752–761
Whitaker JN, Rhodes RH (1983) The distribution of cathepsin D in rat tissues determined by immunohistochemistry. Am J Anat 166:417–428
Yick LW, Wu W, So KF, Wong SY (1998) Time course of NOS expression and neuronal death in Clarke’s nucleus following traumatic injury in adult rat spinal cord. Neurosci Lett 241:155–158
Yue TL, McKenna PJ, Ohlstein EH, Farach-Carson MC (1994) Osteopontin-stimulated vascular smooth muscle cell migration is mediated by β3 integrin. Exp Cell Res 214:459–464
Zhang Y, Anderson PN, Campbell G, Mohajeri H, Schachner M, Lieberman AR (1995) Tenascin-C expression by neurons and glial cells in the rat spinal cord: changes during postnatal development and after dorsal root or sciatic nerve injury. J Neurocytol 24:585–601
Zhang Y, Winterbottom JK, Schachner M, Lieberman AR, Anderson PN (1997) Tenascin-C expression and axonal sprouting following injury to the spinal dorsal columns in the adult rat. J Neurosci Res 49:433–450
Acknowledgements
This work was supported by grants-in-aid to Katsunori Yoshinaga for scientific research from the Ministry of Education, Science and Culture of Japan. We thank Dr. Shintaro Nomura, Osaka University, Osaka, Japan, for kindly providing the plasmid containing OPN cDNA, and Drs. Masamichi Tahara and Masatoshi Komiyama, Chiba University Graduate School of Medicine, for their technical advice and useful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hashimoto, M., Koda, M., Ino, H. et al. Gene expression profiling of cathepsin D, metallothioneins-1 and -2, osteopontin, and tenascin-C in a mouse spinal cord injury model by cDNA microarray analysis. Acta Neuropathol 109, 165–180 (2005). https://doi.org/10.1007/s00401-004-0926-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-004-0926-z