Abstract
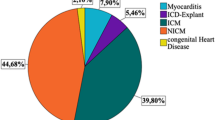
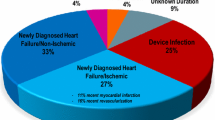
The wearable cardioverter-defibrillator (WCD) was introduced to provide protection from sudden cardiac death (SCD) in patients with transiently elevated risk or during ongoing risk stratification. Benefits and clinical characteristics of routine WCD use remain to be assessed in larger patient populations. This study aims to identify determinants of WCD compliance, therapies, and inappropriate alarms in a real-life cohort. A total of 106 cases (68.9% male) were included between 11/2010 and 04/2016. WCD therapies, automatically recorded arrhythmia episodes, inappropriate WCD alarms, patient compliance, and outcome after WCD prescription were analyzed. Median duration of WCD use was 58.5 days. Average daily wearing time was 22.7 h. Compliance was reduced in patients ≤ 50 years. Three patients received WCD therapies (2.8%). In one case ventricular fibrillation (VF) was appropriately terminated with the first shock. Two patients received inappropriate WCD therapies due to WCD algorithm activation during ventricular pacemaker stimulation. One patient died of asystole while carrying a WCD (0.9%). Additional arrhythmias detected comprised self-terminating sustained ventricular tachycardia (VT; 2.8%), non-sustained VT (2.8%), and supraventricular arrhythmias (5.7%). Inappropriate WCD alarms due to over-/undersensing occurred in 77/106 patients (72.6%), of which 41 (38.7%) experienced ≥ 10 inappropriate WCD alarms during the prescription period. Thirteen patients (12.3%) displayed a mean of > 1 inappropriate alarms/day. WCD use was associated with high compliance and provided protection from VT/VF-related SCD. The majority of patients experienced inappropriate WCD alarms. Alterations in QRS morphology during pacemaker stimulation require consideration in WCD programming to prevent inappropriate alarms.






Similar content being viewed by others
References
Sasaki S, Tomita H, Shibutani S, Izumiyama K, Higuma T, Itoh T, Sasaki K, Horiuchi D, Kimura M, Okumura K (2014) Usefulness of the wearable cardioverter-defibrillator in patients at high risk for sudden cardiac death. Circ J 78:2987–2989
Strauss M, Kouraki K, Skarlos A, Zahn R, Kleemann T (2013) A patient with severely reduced LV function and electrical storm saved by wearable cardioverter-defibrillator: a case report. Herzschrittmacherther Elektrophysiol 24:136–138
Reek S, Geller JC, Meltendorf U, Wollbrueck A, Szymkiewicz SJ, Klein HU (2003) Clinical efficacy of a wearable defibrillator in acutely terminating episodes of ventricular fibrillation using biphasic shocks. Pacing Clin Electrophysiol 26:2016–2022
Epstein AE, Abraham WT, Bianco NR, Kern KB, Mirro M, Rao SV, Rhee EK, Solomon SD, Szymkiewicz SJ (2013) Wearable cardioverter-defibrillator use in patients perceived to be at high risk early post-myocardial infarction. J Am Coll Cardiol 62:2000–2007
Zishiri ET, Williams S, Cronin EM, Blackstone EH, Ellis SG, Roselli EE, Smedira NG, Gillinov AM, Glad JA, Tchou PJ, Szymkiewicz SJ, Chung MK (2013) Early risk of mortality after coronary artery revascularization in patients with left ventricular dysfunction and potential role of the wearable cardioverter defibrillator. Circ Arrhythm Electrophysiol 6:117–128
Duncker D, Haghikia A, Konig T, Hohmann S, Gutleben KJ, Westenfeld R, Oswald H, Klein H, Bauersachs J, Hilfiker-Kleiner D, Veltmann C (2014) Risk for ventricular fibrillation in peripartum cardiomyopathy with severely reduced left ventricular function-value of the wearable cardioverter/defibrillator. Eur J Heart Fail 16:1331–1336
Rao M, Goldenberg I, Moss AJ, Klein H, Huang DT, Bianco NR, Szymkiewicz SJ, Zareba W, Brenyo A, Buber J, Barsheshet A (2011) Wearable defibrillator in congenital structural heart disease and inherited arrhythmias. Am J Cardiol 108:1632–1638
Opreanu M, Wan C, Singh V, Salehi N, Ahmad J, Szymkiewicz SJ, Thakur RK (2015) Wearable cardioverter-defibrillator as a bridge to cardiac transplantation: a national database analysis. J Heart Lung Transplant 34:1305–1309
Schmitt J, Abaci G, Johnson V, Erkapic D, Gemein C, Chasan R, Weipert K, Hamm CW, Klein HU (2017) Safety of the wearable cardioverter-defibrillator (WCD) in patients with implanted pacemakers. Pacing Clin Electrophysiol 40:271–277
Klein HU, Goldenberg I, Moss AJ (2013) Risk stratification for implantable cardioverter defibrillator therapy: the role of the wearable cardioverter-defibrillator. Eur Heart J 34:2230–2242
Wassnig NK, Gunther M, Quick S, Pfluecke C, Rottstadt F, Szymkiewicz SJ, Ringquist S, Strasser RH, Speiser U (2016) Experience with the wearable cardioverter-defibrillator in patients at high risk for sudden cardiac death. Circulation 134:635–643
Kutyifa V, Moss AJ, Klein H, Biton Y, McNitt S, MacKecknie B, Zareba W, Goldenberg I (2015) Use of the wearable cardioverter defibrillator in high-risk cardiac patients: data from the prospective registry of patients using the wearable cardioverter defibrillator (WEARIT-II registry). Circulation 132:1613–1619
Chung MK, Szymkiewicz SJ, Shao M, Zishiri E, Niebauer MJ, Lindsay BD, Tchou PJ (2010) Aggregate national experience with the wearable cardioverter-defibrillator: event rates, compliance, and survival. J Am Coll Cardiol 56:194–203
Erath JW, Vamos M, Sirat AS, Hohnloser SH (2017) The wearable cardioverter-defibrillator in a real-world clinical setting: experience in 102 consecutive patients. Clin Res Cardiol 106:300–306
Duncker D, König T, Hohmann S, Bauersachs J, Veltmann C (2017) Avoiding untimely implantable cardioverter/defibrillator implantation by intensified heart failure therapy optimization supported by the wearable cardioverter/defibrillator—the PROLONG study. J Am Heart Assoc 6:e004512. https://doi.org/10.1161/JAHA.116.004512
Acknowledgements
This work was supported in part by research grants from the German Center for Cardiovascular Research, DZHK (Rotation Grant to MMZ), from the Cardiology Career Program by the Department of Cardiology, University Hospital Heidelberg, to H.H., from the German Cardiac Society and the Hengstberger Foundation (Klaus-Georg and Sigrid Hengstberger Scholarship to DT), from the German Heart Foundation/German Foundation of Heart Research (project F/08/14 to DT), from the Else Kröner-Fresenius-Stiftung (2014_A242 to DT), from the Joachim Siebeneicher Foundation (to DT), and from the Ministry of Science, Research and the Arts Baden-Wuerttemberg (Sonderlinie Medizin to DT).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
MMZ reports receiving travel support from Medtronic and ZOLL CMS. EZ reports receiving lecture fees/honoraria from Bayer Vital, Medtronic and St. Jude Medical. DT reports receiving lecture fees/honoraria from Bayer Vital, Bristol-Myers Squibb, Daiichi Sankyo, Medtronic, Pfizer Pharma, Sanofi-Aventis, St. Jude Medical and ZOLL CMS, and research grant support from Daiichi Sankyo. The remaining authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zylla, M.M., Hillmann, H.A.K., Proctor, T. et al. Use of the wearable cardioverter-defibrillator (WCD) and WCD-based remote rhythm monitoring in a real-life patient cohort. Heart Vessels 33, 1390–1402 (2018). https://doi.org/10.1007/s00380-018-1181-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-018-1181-x