Abstract.
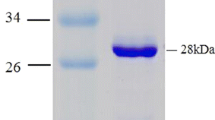
Agrobacterium tumefaciens RS5 harbors two different hydrolytic haloalkanoic acid dehalogenase genes, one coding for a nonstereospecific enzyme (DhlS5II) and a second for a cryptic L-isomer-specific dehalogenase (DhlS5I). The latter gene was cloned and expressed in Escherichia coli. Biochemical characterization and sequence analysis of dhlS5I shows its membership to the class of the L-isomer-specific hydrolytic dehalogenases. Highest homology of 72% was found to the dehalogenase LdexYL from Pseudomonas sp. YL. Both enzymes share an unusual high temperature optimum of 65°C. Controlled by a vector promoter, high specific dehalogenase activities up to 32 U mg−1 protein were obtained in E. coli, and putatively, owing to its own σ70-dependent promoter, a constitutive low-level expression of dhlS5I of 0.4 U mg−1 protein was measured.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 27 June 1997 / Accepted: 4 August 1997
Rights and permissions
About this article
Cite this article
Köhler, R., Brokamp, A., Schwarze, R. et al. Characteristics and DNA-Sequence of a Cryptic Haloalkanoic Acid Dehalogenase from Agrobacterium tumefaciens RS5. Curr Microbiol 36, 96–101 (1998). https://doi.org/10.1007/s002849900286
Issue Date:
DOI: https://doi.org/10.1007/s002849900286