Abstract
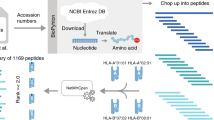
Our genome consists to about 8% of human endogenous retroviral (HERV) sequences. These HERVs have been discussed to be linked to human diseases for decades. Recently, a detailed analysis of a HERV-H sequence located on chromosome Xp22.3 revealed a strong expression in a subset of gastrointestinal cancers whereas expression in normal tissues and in other cancer entities was low. In the present study, we used the reverse immunology approach to test the immunological potential of this HERV-H ORF on Xp22.3. A total of ten peptides displaying HLA-A2.1-binding motifs were selected from the predicted env protein sequence. Stimulation of peripheral T cells with retroviral peptides (RVPs) presented by autologous antigen-presenting cells clearly resulted in sustained proliferation of predominantly CD8+ T cells. High numbers of IFN-γ-secreting T cells were detectable after several weekly stimulations with RVP mixes. Reactivity observed in RVP-Mix–stimulated cultures was attributable to RVP03, RVP09 and to a lower extend to RVP08, suggesting those to be highly immunogenic epitopes. Besides killing of RVP-loaded target cells, up to 40% specific lysis of colorectal carcinoma cell lines endogenously expressing this HERV-H Xp22.3 ORF was achieved. These data demonstrate that human T cells can be sensitized toward HERV peptides and moreover posses a high lytic potential toward HERV-H expressing CRC cells. Additionally, these data hint toward endogenous ENV protein expression followed by proteasomal degradation and presentation in the context of HLA molecules. Finally, our data strengthen the view that HERV-encoded sequences should be considered as a new class of tumor-specific antigens.




Similar content being viewed by others
References
Lander ES, Linton LM, Birren B, Nusbaum C, Zody MC, Baldwin J, Devon K, Doylen M, FitzHugh W, Funke R, Gage D et al (2001) Initial sequencing and analysis of the human genome. Nature 409:860–921. doi:10.1038/35057062
Blikstad V, Benachenhou F, Sperber GO, Blomberg J (2008) Evolution of human endogenous retroviral sequences: a conceptual account. Cell Mol Life Sci 65(21):3348–3365. Review. doi:10.1007/s00018-008-8495-2
Romanish MT, Cohen CJ, Mager DL (2010) Potential mechanisms of endogenous retroviral-mediated genomic instability in human cancer. Semin Cancer Biol 20(4):246–253. Epub 2010 May 26. doi:10.1016/j.semcancer.2010.05.005
Cohen M, Larsson E (1988) Human endogenous retroviruses. Bioessays 9(6):191–196
Alves PM, Lévy N, Stevenson BJ, Bouzourene H, Theiler G, Bricard G, Viatte S, Ayyoub M, Vuilleumier H, Givel JC, Rimoldi D, Speiser DE, Jongeneel CV, Romero PJ, Lévy F (2008) Identification of tumor-associated antigens by large-scale analysis of genes expressed in human colorectal cancer. Cancer Immun 8:11
Christensen T (2010) HERVs in neuropathogenesis. J Neuroimmune Pharmacol 5(3):326–335. doi:10.1007/s11481-010-9214-y
Gimenez J, Montgiraud C, Pichon JP, Bonnaud B, Arsac M, Ruel K, Bouton O, Mallet F (2010) Custom human endogenous retroviruses dedicated microarray identifies self-induced HERV-W family elements reactivated in testicular cancer upon methylation control. Nucleic Acids Res 38(7):2229–2246. doi:10.1093/nar/gkp1214
Christensen T (2005) Association of human endogenous retroviruses with multiple sclerosis and possible interactions with herpes viruses. Rev Med Virol 15(3):179–211. doi:10.1002/rmv.465
Karlsson H, Bachmann S, Schröder J, McArthur J, Torrey EF, Yolken RH (2001) Retroviral RNA identified in the cerebrospinal fluids and brains of individuals with schizophrenia. Proc Natl Acad Sci USA 98(8):4634–4639. doi:10.1073/pnas.061021998
Wang-Johanning F, Frost AR, Jian B, Epp L, Lu DW, Johanning GL (2003) Quantitation of HERV-K env gene expression and splicing in human breast cancer. Oncogene 22(10):1528–1535. doi:10.1038/sj.onc.1206241
Büscher K, Hahn S, Hofmann M, Trefzer U, Ozel M, Sterry W, Löwer J, Löwer R, Kurth R, Denner J (2006) Expression of the human endogenous retrovirus-K transmembrane envelope, Rec and Np9 proteins in melanomas and melanoma cell lines. Melanoma Res 16(3):223–234. doi:10.1097/01.cmr.0000215031.07941.ca
Wentzensen N, Coy JF, Knaebel HP, Linnebacher M, Wilz B, Gebert J, von Knebel Doeberitz M (2007) Expression of an endogenous retroviral sequence from the HERV-H group in gastrointestinal cancers. Int J Cancer 121(7):1417–1423. doi:10.1002/ijc.22826
Wang-Johanning F, Radvanyi L, Rycaj K, Plummer JB, Yan P, Sastry KJ, Piyathilake CJ, Hunt KK, Johanning GL (2008) Human endogenous retrovirus K triggers an antigen-specific immune response in breast cancer patients. Cancer Res 68(14):5869–5877. doi:10.1158/0008-5472.CAN-07-6838
Linnebacher M, Gebert J, Rudy W, Woerner S, Yuan YP, Bork P, von Knebel Doeberitz M (2001) Frameshift peptide-derived T-cell epitopes: a source of novel tumor-specific antigens. Int J Cancer 93(1):6–11. doi:10.1002/ijc.1298
Rammensee H-G, Bachmann J, Emmerich NN, Bachor OA, Stevanovic S (1999) SYFPEITHI: database for MHC ligands and peptide motifs. Immunogenetics 50:213–219
Linnebacher M, Wienck A, Boeck I, Klar E (2010) Identification of an MSI-H tumor-specific cytotoxic T cell epitope generated by the (−1) frame of U79260(FTO). J Biomed Biotechnol 2010:841451. doi:10.1155/2010/841451
Vonderheide RH, Hahn WC, Schultze JL, Nadler LM (1999) The telomerase catalytic subunit is a widely expressed tumor-associated antigen recognized by cytotoxic T lymphocytes. Immunity 10:673–679. doi:10.1016/S1074-7613(00)80066-7
Schultze JL, Michalak S, Seamon MJ, Dranoff G, Jung K, Daley J, Delgado JC, Gribben JG, Nadler LM (1997) CD40-activated human B cells: an alternative source of highly efficient antigen presenting cells to generate autologous antigen-specific T cells for adoptive immunotherapy. J Clin Invest 100(11):2757–2765. doi:10.1172/JCI119822
von Bergwelt-Baildon MS, Vonderheide RH, Maecker B, Hirano N, Anderson KS, Butler MO, Xia Z, Zeng WY, Wucherpfennig KW, Nadler LM, Schultze JL (2002) Human primary and memory cytotoxic T lymphocyte responses are efficiently induced by means of CD40-activated B cells as antigen-presenting cells: potential for clinical application. Blood 99(9):3319–3325. doi:10.1182/blood.V99.9.3319
Ahn K, Kim HS (2009) Structural and quantitative expression analyses of HERV gene family in human tissues. Mol Cells 28(2):99–103. doi:10.1007/s10059-009-0107-y
Wang-Johanning F, Liu J, Rycaj K, Huang M, Tsai K, Rosen DG, Chen DT, Lu DW, Barnhart KF, Johanning GL (2007) Expression of multiple human endogenous retrovirus surface envelope proteins in ovarian cancer. Int J Cancer 120(1):81–90. doi:10.1002/ijc.22256
Szpakowski S, Sun X, Lage JM, Dyer A, Rubinstein J, Kowalski D, Sasaki C, Costa J, Lizardi PM (2009) Loss of epigenetic silencing in tumors preferentially affects primate-specific retroelements. Gene 448(2):151–167. doi:10.1016/j.gene.2009.08.006
Schulz WA, Steinhoff C, Florl AR (2006) Methylation of endogenous human retroelements in health and disease. Curr Top Microbiol Immunol 310:211–250
Ostwald C, Linnebacher M, Weirich V, Prall F (2009) Chromosomally and microsatellite stable colorectal carcinomas without the CpG island methylator phenotype in a molecular classification. Int J Oncol 35(2):321–327. doi:10.3892/ijo_00000343
Takahashi Y, Harashima N, Kajigaya S, Yokoyama H, Cherkasova E, McCoy JP, Hanada K, Mena O, Kurlander R, Tawab A, Srinivasan R, Lundqvist A, Malinzak E, Geller N, Lerman MI, Childs RW (2008) Regression of human kidney cancer following allogeneic stem cell transplantation is associated with recognition of an HERV-E antigen by T cells. J Clin Invest 118(3):1099–1109. doi:10.1172/JCI34409
SenGupta D, Tandon R, Vieira RG, Ndhlovu LC, Lown-Hecht R, Ormsby CE, Loh L, Jones RB, Garrison KE, Martin JN, York VA, Spotts G, Reyes-Terán G, Ostrowski MA, Hecht FM, Deeks SG, Nixon DF (2011) Strong human endogenous retrovirus-specific T cell responses are associated with control of HIV-1 in chronic infection. J Virol 85(14):6977–6985. doi:10.1128/JVI.00179-11
de Parseval N, Casella J, Gressin L, Heidmann T (2001) Characterization of the three HERV-H proviruses with an open envelope reading frame encompassing the immunosuppressive domain and evolutionary history in primates. Virology 279(2):558–569. doi:10.1006/viro.2000.0737
Acknowledgments
Thanks to Michael Mullins for his editing advice as a native speaker and proofreading of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The authors declare that all experiments on human subjects were conducted in accordance with the Declaration of Helsinki and that all procedures were carried out with the adequate understanding and written consent of the subjects.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mullins, C.S., Linnebacher, M. Endogenous retrovirus sequences as a novel class of tumor-specific antigens: an example of HERV-H env encoding strong CTL epitopes. Cancer Immunol Immunother 61, 1093–1100 (2012). https://doi.org/10.1007/s00262-011-1183-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-011-1183-3