Abstract
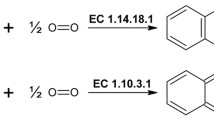
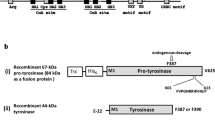
Using primers designed on the basis of sequence homologies in the copper-binding domains for a number of plant and fungal tyrosinases, two tyrosinase encoding cDNAs were cloned from an Agaricus bisporus U1 cDNA-library. The sequences AbPPO1 and AbPPO2 were, respectively, 1.9 and 1.8 kb in size and encoded proteins of approximately 64 kDa. The cDNAs represent different loci. Both AbPPO1 and AbPPO2 occur as single copies on the genomes of the U1 parental strains H39 and H97. The genomic size of AbPPO1 and AbPPO2 is minimally 2.3 and 2.2 kb, respectively. Alignment and phylogenetic analysis of 35 tyrosinase and polyphenol oxidase sequences of animal, plant, fungal, and bacterial origin indicated conserved copper-binding domains, and stronger conservation within genera than between them. The translation products of AbPPO1 and AbPPO2 possess putative N-glycosylation and phosphorylation sites and are recognised by antibodies directed against a 43-kDa tyrosinase. The observations are consistent with previously proposed maturation and activation models for plant and fungal tyrosinases.




Similar content being viewed by others
References
Ausubel F, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1995) Short protocols in molecular biology. John Wiley & Sons, Inc., New York City
Bucheli CS, Robinson SP (1994) Contribution of enzymatic browning to color in sugarcane juice. J Agric Food Chem 42:257–261
Burton KS (1988) The effects of pre- and post-harvest development on mushroom tyrosinase. J Hort Sci 63:255–260
Burton KS, Noble R (1993) The influence of flush number, bruising and storage temperature on mushroom quality. Post-harvest Biol Tech 3:39–47
Cary JW, Lax AR (1995) Biology and molecular biology of polyphenol oxidase. In: Lee CY, Whitaker JR (eds) Enzymatic browning and its prevention. ACS Symposium Series 600, Washington DC, pp 120–128
Espín JC, Wichers HJ (1999) Activation of a latent mushroom (Agaricus bisporus) tyrosinase isoform by sodium dodecyl sulfate (SDS). Kinetic properties of the SDS-activated isoform. J Agric Food Chem 47:3518–3525
Espín JC, Van Leeuwen J, Wichers HJ (1999) Kinetic study of the activation process of a latent mushroom (Agaricus bisporus) tyrosinase by serine proteases. J Agric Food Chem 47:3509–3517
Espín JC, Veltman R, Wichers HJ (2000) The oxidation of L-ascorbic acid catalysed by pear tyrosinase. Physiol Plant 109:1–6
Gerritsen YAM, Chapelon CGJ, Wichers HJ (1994) Low-isoelectric point tyrosinase of Agaricus bisporus appears to be a glycoprotein. Phytochemistry 35:573–577
Hammond JBW, Nichols R (1976) Changes in respiration and soluble carbohydrates during the post-harvest storage of mushrooms (Agaricus bisporus). J Sc. Food Agric 26:835–842
Hunt MD, Eanetta NT, Yu H, Newman SM, Steffens JC (1993) cDNA cloning and expression of potato polyphenol oxidase. Plant Mol Biol 21:59–68
Ichishima E, Maeba H, Amikura T, Sakata H (1984) Multiple forms of protyrosinase from Aspergillus oryzae and their mode of activation at pH 3.0. Biochim Biophys Acta 786:25–31
Kupper U, Niedermann DM, Travaglini G, Lerch K (1989) Isolation and characterization of the tyrosinase gene from Neurospora crassa. J Biol Chem 264:17250–17258
Maniatis T, Fritsch EF, Sambrook J (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory , Cold Spring Harbor, New York
Martínez MV, Whitaker JR (1995) The biochemistry and control of enzymatic browning. Trends Food Sci Technol 6:195–200
Mauerer MJ, Storkus WJ, Kirkwood JM, Lotze MT (1996) New treatment options for patients with melanoma: review of melanoma-derived T-cell epitope-based peptide vaccines. Melanoma Res 6:11–24
Oetting WS, King RA (1992) Molecular analysis of type 1-A (tyrosinase negative) oculocutaneous albinism. Human Genetics 90:258–262
Robinson SP, Dry IB (1992) Broad bean leaf polyphenol oxidase is a 60-kilodalton protein susceptible to proteolytic cleavage. Plant Physiol 99:317–323
Shahar T, Hennig N, Gutfinger T, Hareven D, Lifschitz E (1992) The tomato 66.3 kD polyphenoloxidase gene: molecular identification and developmental expression. The Plant Cell 4:135–147
Sommer A, Ne'eman E, Steffens JC, Mayer AM, Harel E (1994) Import, targeting, and processing of a plant polyphenol oxidase. Plant Physiol 105:1301–1311
Soler-Rivas C, Möller AC, Arpin N, Olivier JM, Wichers HJ (2001) Induction of a tyrosinase mRNA in Agaricus bisporus upon treatment with a tolaasin preparation from Pseudomonas tolaasii. Physiol Mol Plant Pathol 58:95–99
Tobin DJ, Paus R (2001) Graying: gerontobiology of the hair follicle pigmentary unit. Exp Gerontol 36:229–254
Van Gelder CWG, Flurkey WH, Wichers HJ (1997) Sequence and structural features of plant and fungal tyrosinases. Phytochemistry 45:1309–1323
Van Leeuwen J, Wichers HJ (1999) Tyrosinase activity and isoform composition in separate tissues during development of Agaricus bisporus fruitbodies. Mycol Res 103:413–418
Wichers HJ, Gerritsen YAM, Chapelon CGJ (1996) Tyrosinase isoforms from the fruitbodies of Agaricus bisporus. Phytochemistry 43:333–337
Acknowledgements
The technical assistance of Gerard van Duijnhoven, Tanja van den Bosch and Janet Haasjes is gratefully acknowledged. This work was supported by the European Commission (AIR CT 93 0953). The experiments performed comply with the current laws in the Netherlands.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wichers, H.J., Recourt, K., Hendriks, M. et al. Cloning, expression and characterisation of two tyrosinase cDNAs from Agaricus bisporus . Appl Microbiol Biotechnol 61, 336–341 (2003). https://doi.org/10.1007/s00253-002-1194-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-002-1194-2