Abstract
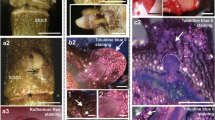
Arabinogalactan proteins (AGPs) are abundant plant proteoglycans implicated in plant growth and development. Here, we report the genetic characterization, partial purification and immunolocalization of a classical AGP (PtaAGP6, accession number AF101785) in loblolly pine (Pinus taeda L.). A PtaAGP6 full-length cDNA clone was expressed in bacteria. PtaAGP6 resembles tomato LeAGP-1 and Arabidopsis AtAGP17-19 in that they all possess a subdomain composed of basic amino acids. The accessibility of this domain in the glycoprotein makes it possible to label the PtaAGP6 epitopes on the cell surface or in the cell wall with polyclonal antibodies raised against this subdomain. The antibodies recognize the peptide of the basic subdomain and bind to the intact protein molecule. A soluble protein-containing fraction was purified from the differentiating xylem of pine trees by using β-glucosyl Yariv reagent (β-glcY) and was recognized by antibodies against the basic subdomain. Immunolocalization studies showed that the PtaAGP6 epitopes are restricted to a file of cells that just precede secondary cell wall thickening, suggesting roles in xylem differentiation and wood formation. The location of apparent labeling of the PtaAGP6 epitopes is separated from the location of lignin deposition. Multiple single nucleotide polymorphisms (SNPs) were detected in EST variants. Denaturing HPLC analysis of PCR products suggests that PtaAGP6 is encoded by a single gene. Mobility variation in denaturing gel electrophoresis was used to map PtaAGP6 SNPs to a site on linkage group 5.
Similar content being viewed by others
References
Allona, I., Quinn, M., Shoop, E., Swope, K., St. Cyr, S., Carlis, J., Riedl, J., Retzel, E., Campbell, M.M., Sederoff, R.R. and Whetten, R.W. 1998. Analysis of xylem formation in pine by cDNA sequencing. Proc. Natl. Acad. Sci. USA 95: 9693–9698.
Alexander, K., Munson, K., Gierdi, D., Haefele, R. and Taylor, P. 1998. Detection of single nucleotide polymorphisms with the WAVETM DNA fragment analysis system. Genet. Test. 1: 201–206.
Audic, S. and Claverie, J. 1997. The significance of digital gene expression profiles. Genome Res. 7: 986–995.
Brown, G.R., Kadel, E.E., Bassoni, D.L., Kiehne, K.L., Temesgen, B., van Buijtenen, J.P., Sewell, M.M., Marshall, K.A. and Neale, D.B. 2001. Anchored reference loci in loblolly pine (Pinus taeda L.) for integrating pine genomics. Genetics 159: 799–809.
Casero, P.J., Casimiro, I. and Knox, J.P. 1998. Occurrence of cell surface arabinogalactan-protein and extensin epitopes in relation to pericycle and vascular tissue development in the root apex of four species. Planta 204: 252–259.
Cassab, G.I. 1998. Plant cell wall proteins. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49: 281–309.
Chapman, A., Blervacq, A.-S., Vasseur, J. and Hilbert J.-L. 2000. Arabinogalactan-proteins in Cichorium somatic embryogenesis: effects of β-glucosyl Yariv reagent and epitope localisation during embryo development. Planta 211: 305–314.
Chen, C.G., Pu, Z.Y., Moritz, R.L., Simpson, R.J., Bacic, A., Clarke, A.E. and Mau, S.L. 1994. Molecular cloning of a gene encoding an arabinogalactan-protein from pear (Pyrus communis) cell suspension culture. Proc. Natl. Acad. Sci. USA 91: 10305–10309.
Cheung, A.Y., Wang, H. and Wu, H.M. 1995. A floral transmitting tissue-specific glycoprotein attracts pollen tubes and stimulates their growth. Cell 82: 383–393.
Ding, L. and Zhu, J.K. 1997. A role for arabinogalactan-proteins in root epidermal cell expansion. Planta 203: 289–294.
Dolan, L., Linstead, P. and Roberts, K. 1995. An AGP epitope distinguishes a central metaxylem initial from other vascular initials in the Arabidopsis root. Protoplasma 189: 149–155.
Du, H., Simpson, R.J., Moritz, R.L., Clarke, A.E. and Bacic, A. 1994. Isolation of the protein backbone of an arabinogalactanprotein from the styles of Nicotiana alata and characterization of a corresponding cDNA. Plant Cell 6: 1643–1653.
Du, H., Simpson, R.J., Clarke, A.E. and Bacic, A. 1996a. Molecular characterization of a stigma-specific gene encoding an arabinogalactan-protein (AGP) from Nicotiana alata. Plant J. 9: 313–323.
Du, H., Clarke, A.E. and Bacic, A. 1996b. Arabinogalactanproteins: a class of extracellular proteoglycans involved in plant growth and development. Trends Cell Biol. 6: 411–414.
Freshour, G., Clay, R.P., Fuller, M.S., Albersheim, P., Darvill, A.G. and Hahn, G. 1996. Developmental and tissue-specific structural alterations of the cell-wall polysaccharides of Arabidopsis thaliana roots. Plant Physiol. 110: 1413–1429.
Fukuda, H., Watanabe, Y., Kuriyama, H., Aoyagi, S., Sugiyama, M., Yamamoto, R., Demura, T. and Minami, A. 1998. Programming of cell death during xylogenesis. J. Plant Res. 111: 253–256.
Gao, M. and Showalter, A.M. 1999. Yariv reagent treatment induces programmed cell death in Arabidopsis cell cultures and implicates arabinogalactan protein involvement. Plant J. 19: 321–332.
Gao, M. and Showalter, A.M. 2000. Immunolocalization of LeAGP-1, a modular arabinogalactan-protein, reveals its developmentally regulated expression in tomato. Planta 210: 865–874.
Gao, M., Kieliszewski, M.J., Lamport, D.T.A. and Showalter, A.M. 1999. Isolation, characterization and immunolocalization of a novel modular tomato arabinogalactan-protein corresponding to the LeAGP-1 gene. Plant J. 18: 43–55.
Gilson, P., Gaspar, Y.M., Oxley, D., Youl, J.J. and Bacic, A. 2001. NaAGP4 is an arabinogalactan protein whose expression is suppressed by wounding and fungal infection in Nicotiana alata. Protoplasma 215: 128–139.
Groover, A., Devey, M.E., Fiddler, T., Lee, J., Megraw, R.A., Mitchell-Olds, T., et al. 1994. Identification of quantitative trait loci influencing wood specific gravity in an outbred pedigree of loblolly pine. Genetics 138: 1293–1300.
Hofmann, K. and Stoffel, W. 1993. TMbase - a database of membrane spanning proteins segments. Biol. Chem. Hoppe-Seyler 347: 166.
Jauh, G.Y. and Lord, E.M. 1996. Localization of pectins and arabinogalactan-proteins in lily (Lilium longiflorum L.) pollen tube and style, and their possible roles in pollination. Planta 199: 251–261.
Knox, J.P. 1995. The use of antibodies to study the architecture and developmental regulation of plant cell walls. Int. J. Cytol. 171: 79–120.
Knox, J.P., Day, S. and Roberts, K. 1989. A set of surface glycoproteins forms an early marker of cell position but not cell type in the root apical meristem of Daucus carota L. Development 106: 47–56.
Knox, J.P., Linstead, P.J., Peart, J., Cooper, C. and Roberts, K. 1991. Developmentally regulated epitopes of cell surface arabinogalactan proteins and their relation to root tissue pattern formation. Plant J. 1: 317–326.
Kreuger, M. and van Holst, G.-J. 1993. Arabinogalactan-proteins are essential in somatic embryogenesis of Daucus carota L. Planta 189: 243–248.
Kreuger, M. and van Holst, G.-J. 1995. Arabinogalactan-protein epitopes in somatic embryogenesis of Daucus carota L. Planta 197: 135–141.
Loopstra, C.A., Puryear, J.D. and No, E.-G. 2000. Purification and cloning of an arabinogalactan-protein from xylem of loblolly pine. Planta 210: 686–689.
Loopstra, C.A. and Sederoff, R.R. 1995. Xylem-specific gene expression in loblolly pine. Plant Mol. Biol. 27: 277–291.
Majewska-Sawka, A. and Nothnagel, E.A. 2000. The multiple roles of arabinogalactan proteins in plant development. Plant Physiol. 122: 3–9.
Mau, S.L., Chen, C.G., Pu, Z.Y., Moritz, R.L., Simpson, R.J., Bacic, A. and Clarke, A.E. 1995. Molecular cloning of cDNAs encoding the protein backbones of arabinogalactan-proteins from the filtrate of suspension-cultured cells of Pyrus communis and Nicotiana alata. Plant J. 8: 269–281.
McCann, M.C. 1997. Tracheary element formation: building up to a dead end. Trends Plant Sci. 2: 333–338.
Megraw, R.A. 1985. Wood quality factors in loblolly pine: the influence of tree age, position in tree, and cultural practice on wood specific gravity, fiber length, and fibril angle. TAPPI Press, Atlanta, GA, 88 pp.
Nielsen, H., Engelbrecht, J., Brunak, S. and von Heijne, G. 1997. Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng. 10: 1–6.
Nothnagel, E.A. 1997. Proteoglycans and related components in plant cells. Int. Rev. Cytol. 174: 195–291.
Oxley, D. and Bacic, A. 1999. Structure of the glycosylphosphatidylinositol anchor of an arabinogalactan protein from Pyrus communis suspension-cultured cells. Proc. Natl. Acad. Sci. USA 96: 14246–14251.
Page, R.D.M. 1996. TREEVIEW: an application to display phylogenetic trees on personal computers. Comp. Appl. Biosci. 12: 357–358.
Pennell, R.I. and Lamb, C. 1997. Programmed cell death in plants. Plant Cell 9: 1157–1168.
Pennell, R.I. and Roberts, K. 1990. Sexual development in the pea is presaged by altered expression of arabinogalactan protein. Nature 344: 547–549.
Roy, S., Jauh, G.Y., Helper, P.K. and Lord, E.M. 1998. Effects of Yariv phenylglycoside on cell wall assembly in the lily pollen tube. Planta 204: 450–458.
Samaj, J., Baluska, F. and Volkmann, D. 1998.Cell-specific expression of two arabinogalactan protein epitopes recognized by monoclonal antibodies JIM8 and JIM13 in maize roots. Protoplasma 204: 1–12.
Schindler, T., Bergfeld, R., Schopfer, P. 1995. Arabinogalactan proteins in maize coleoptiles: developmental relationship to cell death during xylem differentiation but not to extension growth. Plant J. 7: 25–36.
Schultz, C.J., Gilson, P., Oxley, D., Youl J. and Bacic, A. 1998. GPIanchors on arabinogalactan-proteins: implications for signaling in plants. Trends Plant Sci. 3: 426–431.
Schultz, C.J., Johnson, K.L., Currie, G. and Bacic, A. 2000. The classical arabinogalactan protein gene family of Arabidopsis. Plant Cell 12: 1751–1767.
Serpe, M.D. and Nothnagel, E.A. 1994. Effects of Yariv phenylglycosides on Rosa cell suspensions: evidence for the involvement of arabinogalactan-proteins in cell proliferation. Planta 193: 541–550.
Sewell, M.M., Bassoni, D.L., Megraw, R.A., Wheeler, N.C. and Neale, D.B. 2000. Identification of QTLs influencing wood property traits in loblolly pine (Pinus taeda L.). I. Physical wood properties. Theor. Appl. Genet. 101: 1273–1281.
Showalter, A.M. 1993. Structure and function of plant cell wall proteins. Plant Cell 5: 9–23.
Showalter, A.M. and Varner, J.E. 1989. Plant hydroxyproline-rich glycoproteins. In: A. Marcus (Ed.) The Biochemistry of Plants, vol. 15, Academic Press, New York, pp. 485–536.
Stacey, N.J., Roberts, K. and Knox, J.P. 1990. Patterns of expression of the JIM4 arabinogalactan-protein epitopes in cell cultures and during somatic embryogenesis in Daucus carota L. Planta 180: 285–292.
Stam, P. 1993. Construction of integrated genetic linkage maps by means of a new computer package: JoinMap. Plant J. 3: 739–744.
Temesgen, B., Brown, G.R., Harry, D.E., Kinlaw, C.S., Sewell M.M. et al. 2001. Genetic mapping of expressed sequence tag polymorphism (ESTP) markers in loblolly pine (Pinus taeda L.). Theor. Appl. Genet. 102: 664–675.
Thompson, H.J.M. and Knox, J.P. 1998. Stage-specific responses of embryogenic carrot cell suspension cultures to arabinogalactan protein-binding β-glucosyl Yariv reagent. Planta 205: 32–38.
Whetten, R., Sun, Y.H., Zhang, Y. and Sederoff, R.R. 2001. Functional genomics and cell wall biosynthesis in loblolly pine. Plant Mol. Biol. 47: 275–291.
Willats, W.G.T. and Knox, J.P. 1996. A role for arabinogalactanproteins in plant cell expansion: evidence from studies on the interaction of β-glucosyl Yariv reagent with seedlings of Arabidopsis thaliana. Plant J. 9: 919–925.
Wu, H.M., Wong, E., Ogdahl, J. and Cheung, A.Y. 2000. A pollen tube growth-promoting arabinogalactan protein from Nicotiana alata is similar to the tobacco TTS protein. Plant J. 22: 165–176.
Yariv, J., Rapport, M.M. and Graf, L. 1962. The interaction of glycosides and saccharides with antibody to the corresponding phenylazoglycosides. Biochem. J. 85: 383–388.
Youl, J.J., Bacic, A. and Oxley, D. 1998. Arabinogalactan-proteins from Nicotiana alata and Pyrus communis contain glycosylphosphatidylinositol membrane anchors. Proc. Natl. Acad. Sci. USA 95: 7921–7926.
Zhang, Y., Sederoff, R.R. and Allona, I. 2000. Differential expression of genes encoding cell wall proteins in vascular tissues from vertical and bent loblolly pine trees. Tree Physiol. 20: 457–466.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, Y., Brown, G., Whetten, R. et al. An arabinogalactan protein associated with secondary cell wall formation in differentiating xylem of loblolly pine. Plant Mol Biol 52, 91–102 (2003). https://doi.org/10.1023/A:1023978210001
Issue Date:
DOI: https://doi.org/10.1023/A:1023978210001