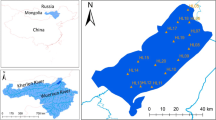
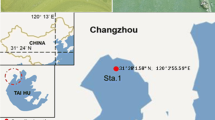
Abstract
Molecular mechanism of interaction between the bloom-forming cyanobacterial species and attached microbios within cyanobacterial aggregates has not been elucidated yet and understanding of which would help to unravel the cyanobacteria bloom-forming mechanism. In this study, we profiled the metabolically active community by high-throughput metatranscriptome sequencing from cyanobacterial aggregates during cyanobacterial bloom period in Lake Taihu, China. A total of 308 million sequences were obtained using the HiSeq 2500 sequencing platform, which provided a great sequence coverage to carry out the in-depth taxonomic classification, functional classification, and metabolic pathway analysis of the cyanobacterial aggregates. The results show that bacteria dominated in cyanobacterial aggregates, accounting for more than 96.66% of total sequences. Microcystis was the most abundant genus, accounted for 26.80% of total assigned sequences at the genus level in cyanobacterial aggregates community; however, Proteobacteria (46.20%) was found to be as the most abundant active bacterial populations at the phylum level. More importantly, nitrogen, phosphonate, and phosphinate metabolism which associated with eutrophication were found in this study. Especially, the enzymes and organisms relating to denitrification and anammox of nitrogen metabolism, which reduced nitrogen concentration by reducing nitrate to nitrogen to inhibit the eutrophication, were first discovered in Lake Taihu during cyanobacterial bloom period. The present study provides a snapshot of metatranscriptome for cyanobacterial aggregates in Lake Taihu and demonstrates that cyanobacterial aggregates could play a key role in the nitrogen cycle in eutrophic water.








Similar content being viewed by others
References
Al Shehhi MR, Gherboudj I, Ghedira H (2014) An overview of historical harmful algae blooms outbreaks in the Arabian Seas. Mar Pollut Bull 86(1-2):314–324. https://doi.org/10.1016/j.marpolbul.2014.06.048
Blomqvist JE, Isaksson S (1994) Skeletal stability after mandibular advancement: a comparison of two rigid internal fixation techniques. J Oral Maxillofac Surg 52(11):1133–1137. https://doi.org/10.1016/0278-2391(94)90529-0
Booijink CC, Boekhorst J, Zoetendal EG, Smidt H, Kleerebezem M, de Vos WM (2010) Metatranscriptome analysis of the human fecal microbiota reveals subject-specific expression profiles, with genes encoding proteins involved in carbohydrate metabolism being dominantly expressed. Appl Environ Microbiol 76(16):5533–5540. https://doi.org/10.1128/AEM.00502-10
Buchfink B, Xie C, Huson DH (2015) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12(1):59–60. https://doi.org/10.1038/nmeth.3176
Cai H, Jiang H, Krumholz LR, Yang Z (2014) Bacterial community composition of size-fractioned aggregates within the phycosphere of cyanobacterial blooms in a eutrophic freshwater lake. PLoS One 9(8):e102879. https://doi.org/10.1371/journal.pone.0102879
Cai HY, Yan ZS, Wang AJ, Krumholz LR, Jiang HL (2013a) Analysis of the attached microbial community on mucilaginous cyanobacterial aggregates in the eutrophic Lake Taihu reveals the importance of Planctomycetes. Microb Ecol 66(1):73–83. https://doi.org/10.1007/s00248-013-0224-1
Cai HY, Yan ZS, Wang AJ, Krumholz LR, Jiang HL (2013b) Analysis of the attached microbial community on mucilaginous cyanobacterial aggregates in the eutrophic Lake Taihu reveals the importance of Planctomycetes. Microb Ecol 66(1):73–83. https://doi.org/10.1007/s00248-013-0224-1
Celaj A, Markle J, Danska J, Parkinson J (2014) Comparison of assembly algorithms for improving rate of metatranscriptomic functional annotation. Microbiome 2(1):39. https://doi.org/10.1186/2049-2618-2-39
Chao JY, Gao G, Tang XM, Dai JY, Zhuang W, Zhang YM (2011) Effects of wind-induced wave on organic aggregates physical and chemical characteristics in a shallow eutrophic lake (Lake Taihu) in China. Huan Jing Ke Xue 32(10):2861–2867
Deben C, Zwaenepoel K, Boeckx C, Wouters A, Pauwels P, Peeters M, Lardon F, Baay M, Deschoolmeester V (2013) Expression analysis on archival material revisited: isolation and quantification of RNA extracted from FFPE samples. Diagn Mol Pathol 22(1):59–64. https://doi.org/10.1097/PDM.0b013e318269de3b
Deng J, Qin B, Paerl HW, Zhang Y, Wu P, Ma J, Chen Y (2014) Effects of nutrients, temperature and their interactions on spring phytoplankton community succession in Lake Taihu, China. PLoS One 9(12):e113960. https://doi.org/10.1371/journal.pone.0113960
Eiler A, Bertilsson S (2004) Composition of freshwater bacterial communities associated with cyanobacterial blooms in four Swedish lakes. Environ Microbiol 6(12):1228–1243. https://doi.org/10.1111/j.1462-2920.2004.00657.x
Gilbert JA, Hughes M (2011) Gene expression profiling: metatranscriptomics. Methods Mol Biol 733:195–205. https://doi.org/10.1007/978-1-61779-089-8_14
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29(7):644–652. https://doi.org/10.1038/nbt.1883
Haas BJ, Papanicolaou A, Yassour M, Grabherr M, Blood PD, Bowden J, Couger MB, Eccles D, Li B, Lieber M, Macmanes MD, Ott M, Orvis J, Pochet N, Strozzi F, Weeks N, Westerman R, William T, Dewey CN, Henschel R, Leduc RD, Friedman N, Regev A (2013) De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protoc 8(8):1494–1512. https://doi.org/10.1038/nprot.2013.084
Hove-Jensen B, McSorley FR, Zechel DL (2011) Physiological role of phnP-specified phosphoribosyl cyclic phosphodiesterase in catabolism of organophosphonic acids by the carbon-phosphorus lyase pathway. J Am Chem Soc 133(10):3617–3624. https://doi.org/10.1021/ja1102713
Huang Y, Niu B, Gao Y, Fu L, Li W (2010) CD-HIT Suite: a web server for clustering and comparing biological sequences. Bioinformatics 26(5):680–682. https://doi.org/10.1093/bioinformatics/btq003
Ibelings BW, Backer LC, Kardinaal WE, Chorus I (2015) Current approaches to cyanotoxin risk assessment and risk management around the globe. Harmful Algae 49:63–74. https://doi.org/10.1016/j.hal.2014.10.002
Jagtap PD, Blakely A, Murray K, Stewart S, Kooren J, Johnson JE, Rhodus NL, Rudney J, Griffin TJ (2015) Metaproteomic analysis using the Galaxy framework. Proteomics 15(20):3553–3565. https://doi.org/10.1002/pmic.201500074
Jansson JK, Neufeld JD, Moran MA, Gilbert JA (2012) Omics for understanding microbial functional dynamics. Environ Microbiol 14(1):1–3. https://doi.org/10.1111/j.1462-2920.2011.02518.x
Li N, Zhang L, Li F, Wang Y, Zhu Y, Kang H, Wang S, Qin S (2011a) Metagenome of microorganisms associated with the toxic Cyanobacteria Microcystis aeruginosa analyzed using the 454 sequencing platform. Chin J Oceanol Limnol 29(3):505–513. https://doi.org/10.1007/s00343-011-0056-0
Li W, Godzik A (2006) Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22(13):1658–1659. https://doi.org/10.1093/bioinformatics/btl158
Li Y, Chen JA, Zhao Q, Pu C, Qiu Z, Zhang R, Shu W (2011b) A cross-sectional investigation of chronic exposure to microcystin in relationship to childhood liver damage in the Three Gorges Reservoir Region, China. Environ Health Perspect 119(10):1483–1488. https://doi.org/10.1289/ehp.1002412
Liao X, Ou T, Gao H, Xu X, Zhu R, Zhang Q (2014) Main reason for concentric rings plaque formation of virus infecting cyanobacteria (A-4L) in lawns of Anabaena variabilis. Wei Sheng Wu Xue Bao 54(2):191–199
Liu Y, Xu Y, Wang Z, Xiao P, Yu G, Wang G, Li R (2016) Dominance and succession of Microcystis genotypes and morphotypes in Lake Taihu, a large and shallow freshwater lake in China. Environ Pollut 219:399–408. https://doi.org/10.1016/j.envpol.2016.05.021
Ma J, Qin B, Wu P, Zhou J, Niu C, Deng J, Niu H (2015) Controlling cyanobacterial blooms by managing nutrient ratio and limitation in a large hyper-eutrophic lake: Lake Taihu, China. J Environ Sci (China) 27:80–86. https://doi.org/10.1016/j.jes.2014.05.042
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21(19):3787–3793. https://doi.org/10.1093/bioinformatics/bti430
McCarty GW, Bremner JM (1992) Regulation of assimilatory nitrate reductase activity in soil by microbial assimilation of ammonium. Proc Natl Acad Sci U S A 89(2):453–456. https://doi.org/10.1073/pnas.89.2.453
Mitra S, Rupek P, Richter DC, Urich T, Gilbert JA, Meyer F, Wilke A, Huson DH (2011) Functional analysis of metagenomes and metatranscriptomes using SEED and KEGG. BMC Bioinformatics 1(12 Suppl):S21
Morales SE, Holben WE (2011) Linking bacterial identities and ecosystem processes: can ‘omic’ analyses be more than the sum of their parts? FEMS Microbiol Ecol 75(1):2–16. https://doi.org/10.1111/j.1574-6941.2010.00938.x
Otten TG, Paerl HW (2015) Health effects of toxic cyanobacteria in U.S. drinking and recreational waters: our current understanding and proposed direction. Curr Environ Health Rep 2(1):75–84. https://doi.org/10.1007/s40572-014-0041-9
Paerl HW, Gardner WS, McCarthy MJ, Peierls BL, Wilhelm SW (2014) Algal blooms: noteworthy nitrogen. Science 346(6206):175. https://doi.org/10.1126/science.346.6206.175-a
Paerl HW, Xu H, McCarthy MJ, Zhu G, Qin B, Li Y, Gardner WS (2011a) Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): the need for a dual nutrient (N & P) management strategy. Water Res 45(5):1973–1983. https://doi.org/10.1016/j.watres.2010.09.018
Paerl HW, Xu H, McCarthy MJ, Zhu G, Qin B, Li Y, Gardner WS (2011b) Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): The need for a dual nutrient (N & P) management strategy. Water Res 45:1973–1983. https://doi.org/10.1016/j.watres.2010.09.018
Parveen B, Ravet V, Djediat C, Mary I, Quiblier C, Debroas D, Humbert JF (2013) Bacterial communities associated with Microcystis colonies differ from free-living communities living in the same ecosystem. Environ Microbiol Rep 5(5):716–724. https://doi.org/10.1111/1758-2229.12071
Podzelinska K, He SM, Wathier M, Yakunin A, Proudfoot M, Hove-Jensen B, Zechel DL, Jia Z (2009) Structure of PhnP, a phosphodiesterase of the carbon-phosphorus lyase pathway for phosphonate degradation. J Biol Chem 284(25):17216–17226. https://doi.org/10.1074/jbc.M808392200
Qin BQ, Zhu GW, Gao G, Zhang YL, Li W, Paerl HW, Carmichael WW (2010) A drinking water crisis in Lake Taihu, China: linkage to climatic variability and lake management. Environ Manag 45(1):105–112. https://doi.org/10.1007/s00267-009-9393-6
Shao K, Gao G, Chi K, Qin B, Tang X, Yao X, Dai J (2013) Decomposition of Microcystis blooms: implications for the structure of the sediment bacterial community, as assessed by a mesocosm experiment in Lake Taihu, China. J Basic Microbiol 53(6):549–554. https://doi.org/10.1002/jobm.201100532
Steffen MM, Li Z, Effler TC, Hauser LJ, Boyer GL, Wilhelm SW (2012) Comparative metagenomics of toxic freshwater cyanobacteria bloom communities on two continents. PLoS One 7(8):e44002. https://doi.org/10.1371/journal.pone.0044002
Tao M, Xie P, Chen J, Qin BQ, Zhang DW, Niu Y, Zhang M, Wang Q, Wu LY (2012) Use of a generalized additive model to investigate key abiotic factors affecting microcystin cellular quotas in heavy bloom areas of Lake Taihu. PLoS One 7(2):e32020. https://doi.org/10.1371/journal.pone.0032020
Trinchet I, Cadel-Six S, Djediat C, Marie B, Bernard C, Puiseux-Dao S, Krys S, Edery M (2013) Toxicity of harmful cyanobacterial blooms to bream and roach. Toxicon 71:121–127. https://doi.org/10.1016/j.toxicon.2013.05.019
Wells ML, Trainer VL, Smayda TJ, Karlson BS, Trick CG, Kudela RM, Ishikawa A, Bernard S, Wulff A, Anderson DM, Cochlan WP (2015) Harmful algal blooms and climate change: learning from the past and present to forecast the future. Harmful Algae 49:68–93. https://doi.org/10.1016/j.hal.2015.07.009
Wu S, Ren Y, Peng C, Hao Y, Xiong F, Wang G, Li W, Zou H, Angert ER (2015) Metatranscriptomic discovery of plant biomass-degrading capacity from grass carp intestinal microbiomes. FEMS Microbiol Ecol 91:10. https://doi.org/10.1093/femsec/fiv107
Xie C, Mao X, Huang J, Ding Y, Wu J, Dong S, Kong L, Gao G, Li CY, Wei L (2011) KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res 39(suppl_2):W316–W322. https://doi.org/10.1093/nar/gkr483
Xie P, Liu J (2001) Practical success of biomanipulation using filter-feeding fish to control cyanobacteria blooms: a synthesis of decades of research and application in a subtropical hypereutrophic lake. ScientificWorldJournal 1:337–356. https://doi.org/10.1100/tsw.2001.67
Xu H, Paerl HW, Qin B, Zhu G, Hall NS, Wu Y (2015) Determining critical nutrient thresholds needed to control harmful cyanobacterial blooms in eutrophic Lake Taihu, China. Environ Sci Technol 49(2):1051–1059. https://doi.org/10.1021/es503744q
Xu H, Paerl HW, Qin BQ, Zhu GW, Gao G (2010) Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic Lake Taihu, China. Limnol Oceanogr 55(1):420–432. https://doi.org/10.4319/lo.2010.55.1.0420
Yamada T, Letunic I, Okuda S, Kanehisa M, Bork P (2011) iPath2.0: interactive pathway explorer. Nucleic Acids Res 39(suppl_2):W412–W415. https://doi.org/10.1093/nar/gkr313
Yang C, Lin, F, Li, Q, Li, T, Zhao, J (2015) Comparative genomics reveals diversified CRISPR-Cas systems of globally distributed Microcystis aeruginosa, a freshwater bloom-forming cyanobacterium. Front Microbiol 6:394. https://doi.org/10.3389/fmicb.2015.00394
Yang F, Zhou YL, Yin LH, Zhu GC, Liang GY, Pu YP (2014) Microcystin-degrading activity of an indigenous bacterial strain Stenotrophomonas acidaminiphila MC-LTH2 isolated from Lake Taihu. PLoS One 9(12):e115989. https://doi.org/10.1371/journal.pone.0115989
Yang Z, Zhang M, Shi X, Kong F, Ma R, Yu Y (2016) Nutrient reduction magnifies the impact of extreme weather on cyanobacterial bloom formation in large shallow Lake Taihu (China). Water Res 103:302–310. https://doi.org/10.1016/j.watres.2016.07.047
Yoshida T, Takashima Y, Tomaru Y, Shirai Y, Takao Y, Hiroishi S, Nagasaki K (2006) Isolation and characterization of a cyanophage infecting the toxic cyanobacterium Microcystis aeruginosa. Appl Environ Microbiol 72(2):1239–1247. https://doi.org/10.1128/AEM.72.2.1239-1247.2006
Zhai S, Yang L, Hu W (2009) Observations of atmospheric nitrogen and phosphorus deposition during the period of algal bloom formation in northern Lake Taihu, China. Environ Manag 44(3):542–551. https://doi.org/10.1007/s00267-009-9334-4
Zhang J-Y, Guan R, Zhang H-J, Li H, Xiao P, Yu G-L, Du L, Cao D-M, Zhu B-C, Li R-H, Lu Z-H (2016) Complete genome sequence and genomic characterization of Microcystis panniformis FACHB 1757 by third-generation sequencing. Stand Genomic Sci 11:1–13
Zhang XJ, Chen C, Ding JQ, Hou AX, Li Y, Niu ZB, Su XY, Xu YJ, Laws EA (2010) The 2007 water crisis in Wuxi, China: analysis of the origin. J Hazard Mater 182(1-3):130–135. https://doi.org/10.1016/j.jhazmat.2010.06.006
Zhou Q, Chen W, Shan K, Zheng L, Song L (2014) Influence of sunlight on the proliferation of cyanobacterial blooms and its potential applications in Lake Taihu, China. J Environ Sci (China) 26(3):626–635. https://doi.org/10.1016/S1001-0742(13)60457-X
Zhu M, Zhu G, Zhao L, Yao X, Zhang Y, Gao G, Qin B (2013) Influence of algal bloom degradation on nutrient release at the sediment-water interface in Lake Taihu, China. Environ Sci Pollut Res Int 20(3):1803–1811. https://doi.org/10.1007/s11356-012-1084-9
Acknowledgements
This study was supported by projects 61271055, 61372164, and 31600096 of the National Natural Science Foundation of China and also The National Key Research and Development Program of China (2016YFA0501602). The Sequencing services were provided by Metagene Science &Technology Co. Ltd. (Wuxi, China).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Vitor Manuel Oliveira Vasconcelos
Rights and permissions
About this article
Cite this article
Chen, Z., Zhang, J., Li, R. et al. Metatranscriptomics analysis of cyanobacterial aggregates during cyanobacterial bloom period in Lake Taihu, China. Environ Sci Pollut Res 25, 4811–4825 (2018). https://doi.org/10.1007/s11356-017-0733-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0733-4