Abstract
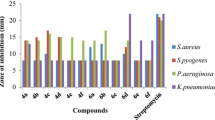
New series of 2-(aryl or alkyl)-N-phenylhydrazine-1-carboxamides 1a–j were synthesized through one-pot reactions of aldehyde or ketones, hydrazine hydrate, and phenylisocyanate in MeOH. The structure of products was confirmed by Fourier transform infrared (FT-IR), proton nuclear magnetic resonance (1H NMR), and 13C NMR spectra. Minimum inhibitory concentration (MIC) of antibacterial activity of 1a–j was screened against five bacterial strains. Compound 1f showed antibacterial activity against Pseudomonas aeruginosa and Staphylococcus aureus.






Similar content being viewed by others
References
S.N. Pandeya, P. Yogeeswari, J.P. Stables, Eur. J. Med. Chem. 35, 879 (2000)
J.R. Dimmock, K.K. Sidhu, S.D. Tumber, S.K. Basran, M. Chen, J.W. Quail, J. Yang, I. Rozas, D.F. Weaver, Eur. J. Med. Chem. 30, 287 (1995)
M. Santagati, M. Modica, A. Santagati, F. Russo, S. Spampinato, Pharmazie 51, 7 (1996)
T. Hemalatha, P.K.M. Imran, A. Gnanamani, S. Na-garajan, Nitric Oxide 19, 303 (2008)
K. Alomar, V. Gaumet, M. Allain, G. Bouet, A. Lan-dreau, J. Inorg. Biochem. 115, 36 (2012)
D.S. Raja, N.S.P. Bhuvanesh, K. Natarajan, Inorg. Chem. 50, 12852 (2011)
L.T.S. Rocha, K.A. Costa, A.C.P. Oliveira, E.B. Nascimento, C.M. Bertollo, F. Araujo, L.R. Teixeira, S.P. Andrade, H. Beraldo, M.M. Coelho, Life Sci. 79, 499 (2006)
S. Shukla, R.S. Srivastava, S.K. Shrivastava, A. Sodhi, P. Kumar, Asian Pac. J. Trop. Biomed. 2, 1040 (2012)
S. Dutta, S. Padhye, K.I. Priyadarsini, C. Newton, Bioorg. Med. Chem. Lett. 15, 2738 (2005)
R.B. de Oliveira, E.M. de Souza-Fagundes, R.P.P. Soares, A.A. Andrade, A.U. Krettli, C.L. Zani, Eur. J. Med. Chem. 43, 1983 (2008)
H. Cerecetto, R. Di Maio, M. González, M. Risso, G. Sagrera, G. Seoane, A. Denicola, G. Peluffo, C. Quijano, A.O.M. Stoppani, M. Paulino, C. Olea-Azar, M.A. Basombrío, Eur. J. Med. Chem. 35, 343 (2000)
C. Gan, J. Cui, S. Su, Q. Lin, L. Jia, L. Fan, Y. Huang, Steroids 87, 99 (2014)
S. Vijaya Laxmi, B. Rajitha, Med. Chem. Res. 21, 85 (2012)
E. Akoz, S. Erdemir, M. Yilmaz, J. Incl. Phenom. Macrocycl. Chem. 73, 449 (2012)
C.M. Goulart, A. Esteves-Souza, C.A. Martinez-Huitle, C.J. Ferreira Rodrigues, M.A. Medeiros Maciel, A. Echevarria, Corros. Sci. 67, 281 (2013)
K. Takagi, H. Hamaguchi, T. Nishimatsu, T. Konno, Vet. Parasitol. 150, 177 (2007)
K. Alomar, V. Gaumet, M. Allain, G. Bouet, A. Landreau, J. Inorg. Biochem. 115, 36 (2012)
P. Anitha, N. Chitrapriya, Y.J. Jang, P. Viswanathamurthi, J. Photochem. Photobiol. B 129, 17 (2013)
É.A. Enyedy, G.M. Bognár, N.V. Nagy, T. Jakusch, T. Kiss, D. Gambino, Polyhedron 67, 242 (2014)
Y. Li, Z.-Y. Yang, Z.-C. Liao, Z.-C. Han, Z.-C. Liu, Inorg. Chem. Commun. 13, 1213 (2010)
H.L. Singh, J.B. Singh, K.P. Sharma, Res. Chem. Intermed. 38, 53 (2012)
P.R. Verma, S. Mandal, P. Gupta, B. Mukhopadhyay, Tetrahedron Lett. 54, 4914 (2013)
G. Sonia, T.K. Ravi, Med. Chem. Res. 22, 3428 (2013)
M. Shalini, P. Yogeeswari, D. Sriram, J.P. Stables, Biomed. Pharmacother. 63, 187 (2009)
N. Raghav, R. Kaur, Med. Chem. Res. 23, 4669 (2014)
M.E. Jung, B.T. Chamberlain, C.-L.C. Ho, E.J. Gillespie, K.A. Bradley, A.C.S. Med, Chem. Lett. 5, 363 (2014)
C.S. Sharma, T. Verma, H.P. Singh, N. Kumar, Med. Chem. Res. 23, 4814 (2014)
B. Qi, B. Mi, X. Zhai, Z. Xu, X. Zhang, Z. Tian, P. Gong, Bioorg. Med. Chem. 21, 5246 (2013)
S. Jin Tan, H.-W. Xi, S. Bedoura, K.H. Lim, Inorg. Chim. Acta 29, 384 (2012)
K. Jakusová, M. Gáplovský, J. Donovalová, M. Cigáň, H. Stankovičová, R. Sokolík, J. Gašpar, A. Gáplovský, Chem. Pap. 67, 117 (2013)
N.O. Mahmoodi, N. Safari, B. Sharifzadeh, Synth. Commun. 44, 245 (2014)
N.O. Mahmoodi, I. Nikokar, M. Farhadi, A. Ghavidast, Z. Naturforsch. 69b, 715 (2014)
N.O. Mahmoodi, J. Parvizi, B. Sharifzadeh, M. Rassa, Arch. Pharm. Chem. Life Sci. 346, 1 (2013)
N.O. Mahmoodi, B. Sharifzadeh, M. Mamaghani, K. Tabatabaeian, A. Salimi Chirani, I. Nikokar, Bioorg. Med. Chem. Lett. 23, 548 (2013)
N.O. Mahmoodi, S. Shoja, B. Sharifzadeh, M. Rassa, Med. Chem. Res. 23, 1207 (2014)
N.O. Mahmoodi, S. Ramzanpour, F. Ghanbari Pirbasti, Arch. Pharm. 348, 275 (2015)
H. Kiyani, N.O. Mahmoodi, K. Tabatabaeian, M.A. Zanjanchi, J. Phys. Org. Chem. 22, 559 (2009)
N.O. Mahmoodi, K. Tabatabaeian, A. Ghavidast. Chin. Chem. Lett. 21, 1199 (2010)
N.O. Mahmoodi, A. Ghavidast, Chem. Heterocycl. Compd. 49, 1451 (2014)
N.O. Mahmoodi, K. Tabatabaeian, H. Kiyani, Helv. Chim. Acta 95, 536 (2012)
Z. Khodaee, A. Yahyazadeh, N.O. Mahmoodi, J. Het, Chemistry 50, 288 (2013)
Acknowledgments
The authors are grateful to the Research Council of University of Guilan for financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mahmoodi, N.O., Namroudi, M., Ghanbari Pirbasti, F. et al. Practical one-pot synthesis of semicarbazone derivatives via semicarbazide, and evaluation of their antibacterial activity. Res Chem Intermed 42, 3625–3636 (2016). https://doi.org/10.1007/s11164-015-2235-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-2235-7