Abstract
Cold habitats are diminishing as a result of climate change, while at the same time little is known of the diversity or biogeography of microbes that thrive in such environments. Furthermore, despite the evident importance of cyanobacteria in polar areas, there are hardly any studies focusing on the phylogenetic relationship between the Arctic and Antarctic cyanobacteria. Here, we described cyanobacterial mats as well as epi- and endoliths collected from shallow streams and rocks, respectively, in the northwestern part of Svalbard. Thirteen populations were identified and characterized by employing morphological and molecular (16S rRNA gene sequences) techniques. Our results were compared to analogous information (available from the GenBank) and related to organisms from similar environments located in the Northern and Southern Hemispheres. In general, the morphological and molecular characterizations complemented each other, and the identified Arctic populations belonged to the following orders: Oscillatoriales (6), Nostocales (6), and Chroococcales (1). Twelve of the identified polar populations showed high similarity (94–99% 16S rRNA gene sequence) when compared to other Arctic and Antarctic cyanobacteria. Mat builder Phormidium autumnale shared only 88% similarity with sequences deposited in the GenBank. Our results demonstrate remarkable similarities of microbial life of Svalbard to that in Antarctica and the High Himalayas. Our findings are a starting point for future comparative research of the benthic as well as endolithic populations of cyanobacteria from the Arctic and Antarctica that will yield new insights into the cold and dry limits of life on Earth. They imply global distributions of the low-temperature cyanobacterial populations throughout the cold terrestrial biosphere.



Similar content being viewed by others
References
Abed RMM, Palinska KA, Camoin G, Golubic S (2006) Common evolutionary origin of planktonic and benthic nitrogen fixing oscillatoriacean cyanobacteria from tropical oceans. FEMS Microbiol Lett 260:171–177
Anagnostidis K, Komarek J (1988) Modern approach to the classification system of cyanophytes: 3—Oscillatoriales. Arch Hydrobiol Suppl 50:327–472
Berrendero E, Perona E, Mateo P (2008) Genetic and morphological characterization of Rivularia and Calothrix (Nostocales, Cyanobacteria) from running water. Int J Syst Evol Microbiol 58:447–460
Bhaya D, Grossman A, Steunou A-S, Khuri N, Cohan FM, Hamamura N, Melendrez MC, Bateson MM, Ward DM, Heidelberg JF (2007) Population level functional diversity in a microbial community revealed by comparative genomic and metagenomic analyses. ISME J 1:703–713
Bonilla S, Villeneuve V, Vincent WF (2005) Benthic and planktonic algal communities in a high Arctic lake: pigment structure and contrasting responses to nutrient enrichment. J Phycol 41:1120–1130
Borin S, Ventura S, Tambone F, Mapelli F, Schubotz F, Brusetti L, Scaglia B, D’Acqui LP, Solheim B, Turicchia S, Marasco R, Hinrichsw KU, Baldi F, Adani F, Daffonchio D (2010) Rock weathering creates oases of life in a high Arctic desert. Environ Microbiol 12:293–303
Broady PA, Kibblewhite AL (1991) Morphological characterization of Oscillatoriales (Cyanobacteria) from Ross Island and southern Victoria Land, Antarctica. Antarct Sci 3:35–45
Chacon E, Berrendero E, Garcia-Pichel F (2006) Biogeological signatures of microboring cyanobacterial communities in marine carbonates from Cabo Rojo, Puerto Rico. Sediment Geol 185:215–228
Cohan FM, Perry E (2007) A systematics for discovering the fundamental units of bacterial diversity. Curr Biol 17:373–386
Comte K, Sabacka M, Carre-Mlouka A, Elster J, Komarek J (2007) Relationships between the Arctic and the Antarctic cyanobacteria; three Phormidium-like strains evaluated by a polyphasic approach. FEMS Microbiol Ecol 59:366–376
Dillon JG, Castenholz RW (2003) The synthesis of the UV-screening pigment, scytonemin and photosynthetic performance in isolates from closely related natural populations of cyanobacteria (Calothrix sp.). Environ Microbiol 5:484–491
Dojani S, Kauff F, Weber B, Buedel B (2014) Genotypic and phenotypic diversity of cyanobacteria in biological soil crusts of the Succulent Karoo and Nama Karoo of Southern Africa. Microb Ecol 67:286–301
Geitler L (1932) Cyanophyceae. In: Kolkwitz R. (ed) Rabenhorst‘s Kryptogamen Flora von Deutschland, Osterreich und der Schweiz, vol 14. Akademische Verlagsgesellschaft, Leipzig, pp 1–1196
Heath MW, Wood SA, Ryan KG (2010) Polyphasic assessment of fresh-water benthic mat-forming cyanobacteria isolated from New Zealand. FEMS Microbiol Ecol 73:95–109
Holzinger A, Karsten U, Lutz L, Wiencke C (2006) Ultrastructure and photosynthesis in the supralittoral green macroalga Prasiola crispa from Spitsbergen (Norway) under UV exposure. Phycologia 45:168–177
Holzinger A, Roleda MY, Lutz C (2009) The vegetative arctic freshwater green alga Zygnema is insensitive to experimental UV exposure. Micron 40:831–838
Hop H, Pearson T, Hegseth EN, Kovacs KM, Wiencke C, Kwasniewski S, Ketil E et al (2002) The marine ecosystem of Kongsfjorden, Svalbard. Polar Res 21:167–208
Howard-Wiliams C, Pridmore R, Downes M, Vincent WF (1989) Microbial biomass, photosynthesis and chlorophyll a related pigments in the ponds of the McMurdo Ice Shelf, Antarctica. Anatarct Sci 1:125–131
Jaag O (1945) Untersuchungen über die Vegetation und Biologie der Algen des nackten Gesteins in den Alpen, im Jura und im schweizerischen Mittelland. In Beiträge zur Kryptogamenflora der Schweiz 9:1–560
Jungblut AD, Hawes I, Mountfort D, Hitzfeld B, Dietrich DR, Burns BP, Neilan BA (2005) Diversity within cyanobacterial mat communities in variable salinity meltwater ponds of McMurdo Ice Shelf, Antarctica. Environ Microbiol 7:519–529
Jungblut AD, Lovejoy C, Vincent WF (2010) Global distribution of cyanobacterial ecotypes in the cold biosphere. ISME J 4:191–202
Kaasalainen U, Olsson S, Rikkinen J (2015) Evolution of the tRNALeu (UAA) Intron and congruence of genetic markers in lichen-symbiotic Nostoc. PLoS One 10:e0131223
Kastovska K, Elster J, Stibal M, Santruckova H (2005) Microbial assemblages in soil microbial succession after glacial retreat in Svalbard (high Arctic). Microb Ecol 50:396–407
Kastovska K, Stibal M, Sabacka M, Cerna B, Santruckova H, Elster J (2007) Microbial community structure and ecology of subglacial sediments in two polythermal Svalbard glaciers characterized by epifluorescence microscopy and PLFA. Polar Biol 30:277–287
Kim GH, Klochkova TA, Kang S-H (2008) Notes on freshwater and terrestrial algae from Ny-Ålesund, Svalbard (High Arctic sea area). J Environ Biol 29:485–491
Kleinteich J, Wood SA, Kupper FC, Camacho A, Quesada A, Frickey T, Dietrich DR (2012) Temperature-related changes in polar cyanobacterial mat diversity and toxin production. Nat Clim Chang 2:356–360
Komarek J (1999) Diversity of cyanoprokaryota (cyanobacteria) of King George Island, maritime Antarctica—a survey. Arch Hydrobiol 94:181–193
Komarek J (2006) Cyanobacterial taxonomy: current problems and prospects for the integration of traditional and molecular approaches. Algae 21:349–375
Komarek J (2010) Recent changes in cyanobacterial taxonomy based on a combination of molecular background with phenotype and ecological consequences (genus and species concept). Hydrobiologia 63:245–259
Komarek J, Anagnostidis K (1999) Cyanoprokaryota 1. Teil Chroococcales. In: Ettl H, Gartner G, Heynig H, Mollenhauer D (eds) Sußwasserflora von Mitteleuropa. Gustav Fischer, Jena-Stuttgart -Lubeck-Ulm
Komarek J, Anagnostidis K (2005) Cyanoprokaryota 2. Teil Oscillatoriales. In: Budel B, Gartner G, Krienitz L, Schagerl M (eds), Sußwasserflora von Mitteleuropa. Elselvier/Spectrum, Heidelberg
Kubeckova K, Elster J, Kanda H (2001) Periphyton ecology of glacial and snowfed streams, Ny-Ålesund, Svalbard: the influence of discharge disturbances due to sloughing, scraping and peeling. Nova Hedwigia Beiheft 123:139–170
Lawes JC, Neilan BA, Brown MV, Clark GF, Johnston EL (2016) Elevated nutrients change bacterial community composition and connectivity: high throughput sequencing of young marine biofilms. Biofouling 32:57–69
Lenzenweger R, Lutz C (2006) A contribution to knowledge of the desmid flora (Desmidiaceae, Zygnemaphyceae) of Spitsbergen. Algol Stud 119:79–89
Lopes VR, Ramos V, Martins A, Sousa M, Welker M, Antunes A, Vasconcelos VM (2012) Phylogenetic, chemical and morphological diversity of cyanobacteria from Portuguese temperate estuaries. Mar Environ Res 73:7–16
Loza V, Berrendero E, Perona E, Mateo P (2013) Polyphasic characterization of benthic cyanobacterial diversity from biofilms of the Guadarrama river (Spain): morphological, molecular and ecological approaches. J Phycol 49:282–297
Martineau E, Wood SA, Miller MR, Jungblut AD, Hawes I, Webster-Brown J, Packer MA (2013) Characterisation of Antarctic cyanobacteria and comparison with New Zealand strains. Hydrobiologia 711:139–154
Mueller DR, Vincent WF, Bonilla S, Laurion I (2005) Extremotrophs, extremophiles and broad pigmentation strategies in a high Arctic ice shelf ecosystem. FEMS Microb Ecol 53:73–87
Mueller DR, Vincent WF, Jeffries MO (2006) Environmental gradients, fragmented habitats, and microbiota of a northern ice shelf cryoecosystem, Ellesmere Island, Canada. Arct Antarct Alp Res 38:593–607
Nadeau T-L, Milbrandt EC, Castenholz RW (2001) Evolutionary relationships of cultivated Antarctic oscillatorians (cyanobacteria). J Phycol 37:650–654
Nuebel U, Garcia-Pichel F, Muyzer G (1997) PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl Environ Microb 63:3327–3332
Palinska KA, Surosz W (2014) Taxonomy of cyanobacteria: a contribution to consensus approach. Hydrobiologia 740:1–11
Papke RT, Ramsing NB, Bateson MM, Ward DM (2003) Geographical isolation in hot spring cyanobacteria. Environ Microbiol 5:650–659
Pocock T, Lachance M-A, Pröschold T, Priscu JC, Kim, Huner NPA (2004) Identification of a psychrophilic green alga from lake Bonney Antarctica: Chlamydomonas raudensis Ettl. (UWO 241) Chlorophyceae. J Phycol 40:1138–1148
Priscu JC, Christner BC (2004) Earth’s icy biosphere. In: Bull A (ed) Microbial diversity and bioprospecting. ASM Press, Washington, DC, pp 130–145
Pushkareva E, Pessi IS, Wilmotte A, Elster J (2015) Cyanobacterial community composition in Arctic soil crusts at different stages of development. FEMS. Microb Ecol 91:1–10
Rippka R, Deruelles J, Waterbury JB, Herdman, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Sawstrom C, Mumford P, Marshall W, Hodson A, Laybourn-Parry J (2002) The microbial communities and primary productivity of cryoconite holes in an Arctic glacier (Svalbard, 79oN). Polar Biol 25:591–596
Schmidt SK, Lynch RC, King AJ, Karki D, Robeson MS, Nagy L, Williams MW, Mitter MS Freeman KR (2011) Phylogeography of microbial phototrophs in the dry valleys oft he high Himalayas and Antarctica. Proc R Soc B Biol Sci 278:702–708
Shams S, Capelli C, Cerasino L, Ballot A, Dietrich DR, Sivonen K, Salmaso N (2014) Anatoxin-a producing Tychonema (cyanobacteria) in European waterbodies. Water Res 69C:68–79
Sihvonen LM, Lyra C, Fewer DP, Rajaniemi-Wacklin P, Lehtimaki JM, Wahlsten M, Sivonen K (2007) Strains of the cyanobacterial genera Calothrix and Rivularia isolated from the Baltic Sea display cryptic diversity and are distantly related to Gloeotrichia and Tolypothrix. FEMS. Microb Ecol 61:74–84
Smith MC, Bowman JP, Scott FJ, Line MA (2000) Sublithic bacteria associated with Antarctic quartz stones. Antarct Sci 12:177–184
Stal L (2000) Microbial mats and Stromatolites. In: Whitton BA, Potts M (eds) The ecology of Cyanobacteria. Their diversity in time and space. Kluwer Academic Publishers, Dordrecht, pp 61–120
Staley JT, Gosink JJ (1999) Poles apart: biodiversity and biogeography of sea ice bacteria. Ann Rev Microbiol 53:189–215
Stibal M, Sabacka M, Kastovska K (2006) Microbial communities on glacier surfaces in Svalbard: Impact on physical and chemical properties on abundance and structure of cyanobacteria and algae. Microb Ecol 52:644–654
Strunecky O, Elster J, Komarek J (2010) Phylogenetic relationships between geographically separate Phormidium cyanobacteria: is the realink between north and south polar regions? Polar Biol 33:1419–1428
Strunecky O, Elster J, Komarek J (2012a) Molecular clock evidence for survival of Antarctic cyanobacteria (Oscillatoriales, Phormidium autumnale) from Paleozoic times. FEMS Microb Ecol 82:482–490
Strunecky O, Komarek J, Elster J (2012b) Biogeography of Phormidium autumnale (Oscillatoriales, Cyanobacteria) in western and central Spitsbergen. Pol Polar Res 33:369–382
Tang EPY, Tremblay R, Vincent WF (1997) Cyanobacterial dominance of polar freshwater ecosystems: are high-latitude mat-formers adapted to low temperature? J Phycol 33:171–181
Taton A, Grubisic S, Brambilla E, De Witt R, Wilmotte A (2003) Cyanobacterial diversity in natural and artificial microbial mats of Lake Fryxell (Mc Murdo Dry Valleys, Antarctica): a morphological and molecular approach. Appl Environ Microbiol 69:5157–5169
Taton A, Grubisic S, Balthazart P, Hodgson DA, Laybourn-Parry J, Wilmotte A (2006a) Biogeographical distribution and ecological range of benthic cyanobacteria in East Antarctic lakes. FEMS Microbiol Ecol 57:272–289
Taton A, Grubisic S, Ertz D, Hodgson DA, Piccardi R, Biondi N, Tredici MR, Mainini M, Losi D, Marinelli F, Wilmotte A (2006b) Polyphasic study of Antarctic cyanobacterial strains. J Phycol 42:1257–1270
Thomazeau S, Houdan-Fourmont A, Coute A, Duval C, Couloux A, Rousseau F, Bernard C (2010) The contribution of sub-Saharan African strains to the phylogeny of cyanobacteria: focusing on the Nostocaceae family (Nostocales, Cyanobacteria). J Phycol 46:564–579
Vincent WF (2000) Cyanobacterial dominance in the Polar Regions. In: Whitton BA, Potts M (eds) The ecology of cyanobacteria. Kluwer Academic Publisher, Dordrecht, pp 321–340
Vopel K, Hawes I (2006) Photosynthetic performance of benthic microbial mats in Lake Hoare, Antarctica. Limnol Oceanogr 51:1801–1812
Wade BD, Garcia-Pichel F (2003) Evaluation of DNA extraction methods for molecular analyses of microbial communities in modern microbialites. Geomicrobiol J 40:1–134
Walker JJ, Pace NR (2007) Phylogenetic composition of Rocky Mountain endolithic microbial ecosystems. Appl Environ Microb 73:3497–3504
Ward DM, Cohan FM, Bhaya D, Heidelberg JF, Kuhl M, Grossman A (2008) Genomics, environmental genomics and the issue of microbial species. Heredity 100:207–219
Whitaker RJ, Grogan DW, Taylor JW (2003) Geographic barriers isolate endemic populations of hyperthermophilic archaea. Scince 301:976–978
Zakhia F, Jungblut AD, Taton A, Vincent WF, Wilmotte A (2009) Cyanobacteria in cold environments. In: Margesin R, Schinner F, Marx JC, Gerday C (eds) Psychrophiles: from biodiversity to biotechnology. Springer Verlag, Berlin, pp 121–135
Zhang L, Jungblut AD, Hawes I, Andersen DT, Sumner DY, Mackey TJ (2015) Cyanobacterial diversity in benthic mats of the McMurdo Dry Valley lakes, Antarctica. Polar Biol 38:1097–1110
Acknowledgements
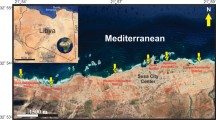
Research was supported by the DFG project PA 842/9-1. The authors thank Dr. Stefano Ventura and CNR-Polarnet for the use of the Italian polar station “Dirigibile Italia” in Ny-Ålesund; Dr. Ewa Jarosz for the proof-reading of the manuscript and English improvement, and Jozef Wiktor jr for the preparation of the Svalbard map.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palinska, K.A., Schneider, T. & Surosz, W. Phenotypic and phylogenetic studies of benthic mat-forming cyanobacteria on the NW Svalbard. Polar Biol 40, 1607–1616 (2017). https://doi.org/10.1007/s00300-017-2083-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-017-2083-6