Abstract
Reduced graphene oxide (rGO) in combination with palladium (Pd) thin film for the detection of hydrogen (H2) has been widely reported; Pd-decorated rGO for carbon monoxide (CO) gas sensing is rarely discussed. This study reveals palladium nanoparticle-decorated rGO (rGO-Pd) detects CO gas easily for a wide range of temperatures (RT–150°C). The study also shows that the device detects CO with detection limit of 50 parts per million (ppm). Percentage response (%R) of pure rGO- and rGO-Pd-based sensors increase with both concentration and operating temperature increase. CO sensing of rGO also improves when decorated with Pd nanoparticles. Further, the increase in the %R in rGO-Pd is because of the synergistic electron exchange activity of rGO and Pd. New mechanistic insight into the dependence of sensing performance of rGO and rGO-Pd on pre-adsorbed oxygen species and baseline resistance drift at a particular temperature is proposed. The detailed analysis of the variation in adsorbed oxygen and number of electrons exchanged during CO molecule adsorption at different temperatures is discussed.
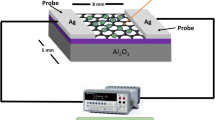
Graphical Abstract














Similar content being viewed by others
References
N.H. Ha, D.D. Thinh, N.T. Huong, N.H. Phuong, P.D. Thach, and H.S. Hong, Fast response of carbon monoxide gas sensors using a highly porous network of ZnO nanoparticles decorated on 3D reduced graphene oxide. Appl. Surf. Sci. 434, 1048 (2018).
Z.U. Abideen, J.H. Kim, A. Mirzaei, H.W. Kim, and S.S. Kim, Sensing behavior to ppm-level gases and synergistic sensing mechanism in metal-functionalized rGO-loaded ZnO nanofibers. Sens. Actuators B Chem. 255, 1884 (2018).
J. Cookson, The preparation of palladium nanoparticles. Platinum Metals Rev. 56, 83 (2012).
C.M. Chang, M.H. Hon, and C. Leu, Improvement in CO sensing characteristics by decorating ZnO nanorod arrays with Pd nanoparticles and the related mechanisms. RSC Adv. 2, 2469 (2012).
N.D. Hoa, P. Van Tong, N. Van Duy, T.D. Dao, H.V. Chung, T. Nagao, and N. Van Hieu, Effective decoration of Pd nanoparticles on the surface of SnO2 nanowires for enhancement of CO gas-sensing performance. J. Hazard. Mater. 265, 124 (2014).
D. Zhang, C. Jiang, J. Liu, and Y. Cao, Carbon monoxide gas sensing at room temperature using copper oxide-decorated graphene hybrid nanocomposite prepared by layer-by-layer self-assembly. Sens. Actuators B Chem. 247, 875 (2017).
F. Liang, S. Chen, W. Xie, and C. Zou, The decoration of Nb-doped TiO2 microspheres by reduced graphene oxide for enhanced CO gas sensing. J. Phys. Chem. Solids 114, 195 (2018).
D. Panda, A. Nandi, S.K. Datta, H. Saha, and S. Majumdar, Selective detection of carbon monoxide (CO) gas by reduced graphene oxide (rGO) at room temperature. RSC Adv. 6, 47337 (2016).
F. Pan, H. Lin, H. Zhai, Z. Miao, Y. Zhang, K. Xu, B. Guan, H. Huang, and H. Zhang, Pd-doped TiO2 film sensors prepared by premixed stagnation flames for CO and NH3 gas sensing. Sens. Actuators B Chem. 261, 451 (2018).
M. Reddeppa, S.B. Mitta, T. Chandrakalavathi, B.G. Park, G. Murali, R. Jeyalakshmi, S.G. Kim, S.H. Park, and M.D. Kim, Solution-processed Au@ rGO/GaN nanorods hybrid-structure for self-powered UV, visible photodetector and CO gas sensors. Curr. Appl. Phys. 19, 938 (2019).
N. Sharma, H.S. Kushwaha, S.K. Sharma, and K. Sachdev, Fabrication of LaFeO3 and rGO-LaFeO3 microspheres based gas sensors for detection of NO2 and CO. RSC Adv. 10, 1297 (2020).
P.A. Pandey, N.R. Wilson, and J.A. Covington, Pd-doped reduced graphene oxide sensing films for H2 detection. Sens. Actuators B Chem. 183, 478 (2013).
P. Bhardwaj, P.B. Barman, and S.K. Hazra, Effect of capping-agent concentration on size and size dispersity of palladium nanoparticles for resistive-type hydrogen sensors. J. Electron. Mater. 49, 6656 (2020).
M.R. Karim, K. Hatakeyama, T. Matsui, H. Takehira, T. Taniguchi, M. Koinuma, Y. Matsumoto, T. Akutagawa, T. Nakamura, S.I. Noro, and T. Yamada, Graphene oxide nanosheet with high proton conductivity. J. Am. Chem. Soc. 135, 8097 (2013).
S.I. El-Hout, S.M. El-Sheikh, H.M. Hassan, F.A. Harraz, I.A. Ibrahim, and E.A. El-Sharkawy, A green chemical route for synthesis of graphene supported palladium nanoparticles: a highly active and recyclable catalyst for reduction of nitrobenzene. Appl. Catal. A: Gen. 503, 176 (2015).
Z. Wei, R. Pan, Y. Hou, Y. Yang, and Y. Liu, Graphene-supported Pd catalyst for highly selective hydrogenation of resorcinol to 1, 3-cyclohexanedione through giant π-conjugate interactions. Sci. Rep. 5, 1 (2015).
Z. Cui and X. Bai, Ultrasonic-assisted synthesis of two dimensional coral-like Pd nanosheets supported on reduced graphene oxide for enhanced electrocatalytic performance. Ultrason. Sonochem. 70, 105309 (2021).
K. Bramhaiah and N.S. John, Hybrid films of reduced graphene oxide with noble metal nanoparticles generated at a liquid/liquid interface for applications in catalysis. RSC Adv. 3, 7765 (2013).
T. Kuila, S. Bose, A.K. Mishra, P. Khanra, N.H. Kim, and J.H. Lee, Chemical functionalization of graphene and its applications. Prog. Mater. Sci. 57, 1061 (2012).
R. Kumar, Doping and stress induced raman shifts in Pd-decorated CVD grown graphene. ECS J. Solid State Sci. Technol. 10, 061002 (2021).
S. Drewniak, R. Muzyka, A. Stolarczyk, T. Pustelny, M. Kotyczka-Morańska, and M. Setkiewicz, Studies of reduced graphene oxide and graphite oxide in the aspect of their possible application in gas sensors. Sensors (Basel). 16, 103 (2016).
L.J. Wang, J. Zhang, X. Zhao, L.L. Xu, Z.Y. Lyu, M. Lai, and W. Chen, Palladium nanoparticle functionalized graphene nanosheets for Li–O2 batteries: enhanced performance by tailoring the morphology of the discharge product. RSC Adv. 5, 73451 (2015).
S. Muralikrishna, K. Sureshkumar, T.S. Varley, D.H. Nagaraju, and T. Ramakrishnappa, In situ reduction and functionalization of graphene oxide with L-cysteine for simultaneous electrochemical determination of cadmium (II), lead (II), copper (II), and mercury (II) ions. Anal. Methods 6, 8698 (2014).
Y. Zheng, Q. Qiao, J. Wang, X. Li, and J. Jian, Gas sensing behavior of palladium oxide for carbon monoxide at low working temperature. Sens. Actuators B Chem. 212, 256 (2015).
S. Zhu, Y. Liu, G. Wu, L. Fei, S. Zhang, Y. Hu, Z. Yan, Y. Wang, H. Gu, and W. Chen, Mechanism study on extraordinary room-temperature CO sensing capabilities of Pd-SnO2 composite nanoceramics. Sens. Actuators B Chem. 285, 49 (2019).
S.W. Choi and S.S. Kim, Room temperature CO sensing of selectively grown networked ZnO nanowires by Pd nanodot functionalization. Sens. Actuators B Chem. 168, 8 (2012).
Q. Hu, S. Liu, and Y. Lian, Sensors for carbon monoxide based on Pd/SnO2/CNT nanocomposites. Phys. Status Solidi A 211, 2729 (2014).
H.Y. Lai and C.H. Chen, Highly sensitive room-temperature CO gas sensors: Pt and Pd nanoparticle-decorated In2O3 flower-like nanobundles. J. Mater. Chem. 22, 13204 (2012).
M. Shojaee, S. Nasresfahani, and M.H. Sheikhi, Hydrothermally synthesized Pd-loaded SnO2/partially reduced graphene oxide nanocomposite for effective detection of carbon monoxide at room temperature. Sens. Actuators B Chem. 254, 457 (2018).
M. Wang, B. Sun, Z. Jiang, Y. Liu, X. Wang, Z. Tang, Y. Wang, and W. Chen, Preparation and extraordinary room-temperature co sensing capabilities of Pd–SnO2 composite nanoceramics. J. Nanosci. Nanotechnol. 18, 4176 (2018).
A.K. Basu, P.S. Chauhan, M. Awasthi, and S. Bhattacharya, α-Fe2O3 loaded rGO nanosheets based fast response/recovery CO gas sensor at room temperature. Appl. Surf. Sci. 465, 56 (2019).
S.K. Hazra and S. Basu, Graphene-oxide nano composites for chemical sensor applications. C 2, 12 (2016).
U. Latif and F.L. Dickert, Graphene hybrid materials in gas sensing applications. Sensors 15, 30504 (2015).
Acknowledgments
The authors thank NIT, Hamirpur, for Raman and IIT, Mandi, for XPS studies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kashyap, A., Barman, P.B. & Hazra, S.K. Response of rGO and Pd-Decorated rGO to Carbon Monoxide Gas. J. Electron. Mater. 52, 1999–2011 (2023). https://doi.org/10.1007/s11664-022-10161-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-022-10161-4