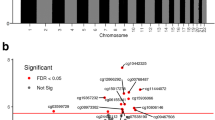
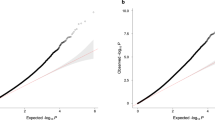
Abstract
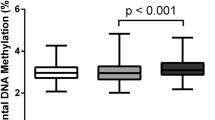
Emerging evidence indicates that maternal medical risk during pregnancy, such as gestational diabetes mellitus (GDM), preeclampsia, and obesity, predisposes the offspring to suboptimal development. However, the underlying biological/epigenetic mechanism in utero is still unknown. The current pilot study (N = 50) compared the levels of global methylation in the placenta and umbilical cord blood among women with and without each risk condition (GDM, preeclampsia, and obesity) and explored whether the levels of global methylation were associated with fetal/infant growth. Results show that global methylation levels in the placenta were lower in patients with gestational diabetes (P = .003) and preeclampsia (P = .05) but higher with obesity (P = .01). Suggestive negative associations were found between global methylation level in the placenta and infant body length and head circumference. While preliminary, it is possible that the placenta tissue, but not umbilical cord blood, may be epigenetically programmed by maternal GDM, preeclampsia, and obesity to carry out its own specific functions that influence fetal growth.
Similar content being viewed by others
References
Berkowitz G, Lapinski R, Wein R, Lee D. Race/ethnicity and other risk factors for gestational diabetes. Am J Epidemiol. 1992;135(9):965–973.
Ferrara A. Increasing prevalence of gestational diabetes mellitus: a public health perspective. Diabetes Care. 2007;30(suppl 2):S141–S146.
Boney C, Verma A, Tucker R, Vohr B. Metabolic syndrome in childhood: association with birth weight, maternal obesity and gestational diabetes mellitus. Pediatrics. 2005;115(3):290–296.
Catalano P, Presley L, Minium J, Hauguel-De S. Fetuses of obese mothers develop insulin resistance in utero. Diabetes Care. 2009;32(6):1076–1080.
Catalano P, Farrell K, Thomas A, et al. Perinatal risk factors for childhood obesity and metabolic dysregulation. Am J Clin Nutr. 2009;90(5):1303–1313.
Knight M; UKOSS. Eclampsia in the United Kingdom 2005. BJOG. 2007;114(9):1072–1078.
Kral J, Naslund E. Surgical treatment of obesity. Nat Clin Pract Endocrinol Metab. 2007;3(8):574–583.
Lawlor D, Timpson N, Harbord R, et al. Exploring the developmental overnutrition hypothesis using parental-offspring associations and FTO as an instrumental variable. PLoS Med. 2008;5(3):84–93.
Taylor P, Poston L. Developmental programming of obesity in mammals. Exp Physiol. 2006;92(2):287–298.
Nomura Y, Marks D, Grossman B, et al. Exposure to gestational diabetes mellitus and low socioeconomic status: effects on neurocognitive development and risk of attention-deficit/hyperactivity disorder in offspring. Arch Pediatr Adolesc Med. 2012;166(4):337–343.
Krakowiak P, Walker C, Bremer A, et al. Maternal metabolic conditions and risk for autism and other neurodevelopmental disorders. Pediatrics. 2012;129(5):1121–1128.
Barker DJ. Mothers, Babies, and Health in Later Life. 2nd ed. Edinburgh, UK: Churchill Livingstone; 1998.
McMillen IC, Robinson JS. Developmental origins of the metabolic syndrome: prediction, plasticity, and programming. Physiol Rev. 2005;85(2):571–633.
Thompson C, Syddall H, Rodin I, Osmond C, Barker D. Birth weight and the risk of depressive disorder in late life. Br J Psychiatry. 2001;179:450–455.
Sallout B, Walker M. The fetal origin of adult diseases. J Obstet Gynecol. 2003;23(5):555–560.
Talge N, Neal C, Glover V. Antenatal maternal stress and long-term effects on child neurodevelopment: how and why? J Child Psychol Psychiatry. 2007;48(3–4):245–261.
Barker D. The developmental origins of adult disease. J Am Coll Nutr. 2004;23(suppl 6):588S-595S.
Barker D. Intrauterine programming of adult disease. Mol Med Today. 1995;1(9):418–423.
Lambertini L, Lee MJ, Marsit CJ, Chen J. Genomic imprinting in human placenta. In: Zheng J, ed. Recent Advances in Research on the Human Placenta. Rijeka, Croatia: InTech Open Access Publisher; 2012:357–377.
Santos F, Dean W. Epigenetic reprogramming during early development in mammals. Reproduction. 2004;127(6):643–651.
Tobi EW, Lumey LH, Talens RP, et al. DNA methylation differences after exposure to prenatal famine are common and timing- and sex-specific. Hum Mol Genet. 2009;18(21):4046–4053.
Cutfield W, Hofman P, Mitchell M, Morison I. Could epigenetics play a role in the developmental origins of health and disease? Pediatr Res. 2007;61(5 pt 2):68R-75R.
Ornoy A, Ratzon N, Greenbaum C, Wolf A, Dulitzky M. School-age children born to diabetic mothers and to mothers with gestational diabetes exhibit a high rate of inattention and fine and gross motor impairment. J Pediatr Endocrinol Metab. 2001;14(suppl 1):681–689.
Dionne G, Boivin M, Séguin JR, Pérusse D, Tremblay RE. Gestational diabetes hinders language development in offspring. Pediatrics. 2008;122(5): 1073–1079.
Veena SR, Krishnaveni GV, Srinivasan K, et al. Childhood cognitive ability: relationship to gestational diabetes mellitus in India. Diabetologia. 2010;53(10):2134–2138.
Xu X, Gammon MD, Herandez-Vargas H, et al. DNA methylation in peripheral blood measured by LUMA is associated with breast cancer in population-based study. FASEB J. 2012;26(6):2657–2666.
Bjornsson HT, Sigurdsson MI, Fallin MD, et al. Intra individual change over time in DNA methylation with familial clustering. JAMA. 2008;299(24):2877–2883.
Simes RJ. An improved Bonferroni procedure for multiple tests of significance. Biometrika. 1986;73(3):751–754.
Ramsey EM. The Placenta: Human and Animal. 1st ed. New York, NY: Praeger; 1982.
Ehrlich M, Gama-Sosa MA, Huang LH, et al. Amount and distribution of 5-methylcytosine in human DNA from different types of tissues of cells. Nucleic Acids Res. 1982;10(8):2709–2721.
Gama-Sosa MA, Midgett RM, Slagel VA, et al. Tissue-specific differences in DNA methylation in various mammals. Biochim Biophys Acta. 1983;740(2):212–219.
Hu D, Cross JC. Development and function of trophoblast giant cells in the rodent placenta. Int J Dev Biol. 2010;54(2–3):341–354.
Williams PL, Seage III GR, van Dyke RB, et al. A trigger-based design for evaluating the safety of in utero antiretroviral exposure in uninfected children of human immunodeficiency virus-infected mothers. Am J Epidemiol. 2012;175(9):950–961.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nomura, Y., Lambertini, L., Rialdi, A. et al. Global Methylation in the Placenta and Umbilical Cord Blood From Pregnancies With Maternal Gestational Diabetes, Preeclampsia, and Obesity. Reprod. Sci. 21, 131–137 (2014). https://doi.org/10.1177/1933719113492206
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719113492206