Abstract
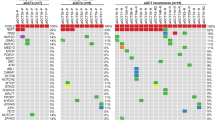
Ovarian granulosa cell tumors (OGCT) are the most frequent kind of sex cord-stromal tumors, and represent ∼2–5% of all ovarian malignancies. OGCTs exist as two entities, juvenile and adult types, with specific clinical and pathological characteristics. The molecular pathogenesis of these tumors has just begun to be unraveled. Indeed, recent studies have indicated that mutation and/or misregulation of the key ovarian transcription factor FOXL2 has a role in OGCT formation, although the mechanisms remain unclear. To better understand the molecular characteristics of OGCT, we studied the transcriptomic profiles of ten human adult-type OGCT samples, as well as ethnically matched granulosa cell (GC) controls. We find that the OGCT samples analyzed herein exhibit several hallmarks of cancer, including increased expression of genes linked to cell proliferation, but decreased expression of those conferring sensitivity to cell death. Moreover, genes differentially expressed in OGCTs are significantly enriched for known FOXL2 target genes, consistently with the prevalence of FOXL2 somatic mutation in these tumors. Expression of these targets is altered in a way expected to promote malignant transformation, for instance, through induction of genes associated with faster cell cycling and downregulation of genes associated with cell death. Over time, such defects may be responsible at least partly for the malignant transformation of healthy GCs into OGCT. These insights into the molecular pathogenesis of OGCTs may open the way to new efforts in the development of more targeted therapeutic strategies for OGCT patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Pectasides D, Pectasides E, Psyrri A . Granulosa cell tumor of the ovary. Cancer Treat Rev 2008; 34: 1–12.
Schumer ST, Cannistra SA . Granulosa cell tumor of the ovary. J Clin Oncol 2003; 21: 1180–1189.
East N, Alobaid A, Goffin F, Ouallouche K, Gauthier P . Granulosa cell tumour: a recurrence 40 years after initial diagnosis. J Obstet Gynaecol Can 2005; 27: 363–364.
Kalfa N, Veitia RA, Benayoun BA, Boizet-Bonhoure B, Sultan C . The new molecular biology of granulosa cell tumors of the ovary. Genome Med 2009; 1: 81.
Benayoun BA, Caburet S, Dipietromaria A, Georges A, D'Haene B, Pandaranayaka PJ et al. Functional exploration of the adult ovarian granulosa cell tumor-associated somatic FOXL2 mutation p.Cys134Trp (c.402C>G). PLoS One 2010; 5: e8789.
Jamieson S, Butzow R, Andersson N, Alexiadis M, Unkila-Kallio L, Heikinheimo M et al. The FOXL2 C134W mutation is characteristic of adult granulosa cell tumors of the ovary. Mod Pathol 2010; 23: 1477–1485.
Kim MS, Hur SY, Yoo NJ, Lee SH . Mutational analysis of FOXL2 codon 134 in granulosa cell tumour of ovary and other human cancers. J Pathol 2010; 221: 147–152.
Schrader KA, Gorbatcheva B, Senz J, Heravi-Moussavi A, Melnyk N, Salamanca C et al. The specificity of the FOXL2 c.402C>G somatic mutation: a survey of solid tumors. PLoS One 2009; 4: e7988.
Shah SP, Kobel M, Senz J, Morin RD, Clarke BA, Wiegand KC et al. Mutation of FOXL2 in granulosa-cell tumors of the ovary. N Engl J Med 2009; 360: 2719–2729.
Crisponi L, Deiana M, Loi A, Chiappe F, Uda M, Amati P et al. The putative forkhead transcription factor FOXL2 is mutated in blepharophimosis/ptosis/epicanthus inversus syndrome. Nat Genet 2001; 27: 159–166.
Cocquet J, Pailhoux E, Jaubert F, Servel N, Xia X, Pannetier M et al. Evolution and expression of FOXL2. J Med Genet. 2002; 39: 916–921.
Uhlenhaut NH, Jakob S, Anlag K, Eisenberger T, Sekido R, Kress J et al. Somatic sex reprogramming of adult ovaries to testes by FOXL2 ablation. Cell 2009; 139: 1130–1142.
Batista F, Vaiman D, Dausset J, Fellous M, Veitia RA . Potential targets of FOXL2, a transcription factor involved in craniofacial and follicular development, identified by transcriptomics. Proc Natl Acad Sci USA 2007; 104: 3330–3335.
Benayoun BA, Batista F, Auer J, Dipietromaria A, L'Hote D, De Baere E et al. Positive and negative feedback regulates the transcription factor FOXL2 in response to cell stress: evidence for a regulatory imbalance induced by disease-causing mutations. Hum Mol Genet 2009; 18: 632–644.
Benayoun BA, Georges AB, L'Hote D, Andersson N, Dipietromaria A, Todeschini AL et al. Transcription factor FOXL2 protects granulosa cells from stress and delays cell cycle: role of its regulation by the SIRT1 deacetylase. Hum Mol Genet 2011; 20: 1673–1686.
Lee K, Pisarska MD, Ko JJ, Kang Y, Yoon S, Ryou SM et al. Transcriptional factor FOXL2 interacts with DP103 and induces apoptosis. Biochem Biophys Res Commun 2005; 336: 876–881.
Kim JH, Yoon S, Park M, Park HO, Ko JJ, Lee K et al. Differential apoptotic activities of wild-type FOXL2 and the adult-type granulosa cell tumor-associated mutant FOXL2 (C134W). Oncogene 2011; 30: 1653–1663.
Benayoun BA, Kalfa N, Sultan C, Veitia RA . The forkhead factor FOXL2: a novel tumor suppressor? Biochim Biophys Acta 2010; 1805: 1–5.
Fleming NI, Knower KC, Lazarus KA, Fuller PJ, Simpson ER, Clyne CD . Aromatase is a direct target of FOXL2: C134W in granulosa cell tumors via a single highly conserved binding site in the ovarian specific promoter. PLoS One 2010; 5: e14389.
Johnson WE, Li C, Rabinovic A . Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 2007; 8: 118–127.
Nagaraja AK, Middlebrook BS, Rajanahally S, Myers M, Li Q, Matzuk MM et al. Defective gonadotropin-dependent ovarian folliculogenesis and granulosa cell gene expression in inhibin-deficient mice. Endocrinology 2010; 151: 4994–5006.
Tusher VG, Tibshirani R, Chu G . Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 2001; 98: 5116–5121.
Suzuki R, Shimodaira H . Pvclust: an R package for assessing the uncertainty in hierarchical clustering. Bioinformatics 2006; 22: 1540–1542.
Saldanha AJ . Java Treeview--extensible visualization of microarray data. Bioinformatics 2004; 20: 3246–3248.
Dennis G, Sherman BT, Hosack DA, Yang J, Gao W, Lane HC et al. DAVID: database for annotation, visualization, and integrated discovery. Genome Bio. 2003; 4: P3.
Huangda W, Sherman BT, Lempicki RA . Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 2009; 4: 44–57.
Hanahan D, Weinberg RA . The hallmarks of cancer. Cell 2000; 100: 57–70.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Kalfa N, Philibert P, Patte C, Ecochard A, Duvillard P, Baldet P et al. Extinction of FOXL2 expression in aggressive ovarian granulosa cell tumors in children. Fertil Steril 2007; 87: 896–901.
Pangas SA, Li X, Umans L, Zwijsen A, Huylebroeck D, Gutierrez C et al. Conditional deletion of Smad1 and Smad5 in somatic cells of male and female gonads leads to metastatic tumor development in mice. Mol Cell Biol 2008; 28: 248–257.
Eden E, Navon R, Steinfeld I, Lipson D, Yakhini Z . GOrilla: a tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinformatics 2009; 10: 48.
Blount AL, Schmidt K, Justice NJ, Vale WW, Fischer WH, Bilezikjian LM . FoxL2 and Smad3 coordinately regulate follistatin gene transcription. J Biol Chem 2009; 284: 7631–7645.
Kyronlahti A, Kauppinen M, Lind E, Unkila-Kallio L, Butzow R, Klefstrom J et al. GATA4 protects granulosa cell tumors from TRAIL-induced apoptosis. Endocr Relat Cancer 2010; 17: 709–717.
Chen C, Grennan K, Badner J, Zhang D, Gershon E, Jin L et al. Removing batch effects in analysis of expression microarray data: an evaluation of six batch adjustment methods. PLoS One 2011; 6: e17238.
Heinz S, Benner C, Spann N, Bertolino E, Lin YC, Laslo P et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol Cell 2010; 38: 576–589.
Moumne L, Dipietromaria A, Batista F, Kocer A, Fellous M, Pailhoux E et al. Differential aggregation and functional impairment induced by polyalanine expansions in FOXL2, a transcription factor involved in cranio-facial and ovarian development. Hum Mol Genet 2008; 17: 1010–1019.
Kim HP, Roe JH, Chock PB, Yim MB . Transcriptional activation of the human manganese superoxide dismutase gene mediated by tetradecanoylphorbol acetate. J Biol Chem 1999; 274: 37455–37460.
Rishi AK, Sun RJ, Gao Y, Hsu CK, Gerald TM, Sheikh MS et al. Post-transcriptional regulation of the DNA damage-inducible gadd45 gene in human breast carcinoma cells exposed to a novel retinoid CD437. Nucleic Acids Res 1999; 27: 3111–3119.
Zhang H, Vollmer M, De Geyter M, Litzistorf Y, Ladewig A, Durrenberger M et al. Characterization of an immortalized human granulosa cell line (COV434). Mol Hum Reprod 2000; 6: 146–153.
Sambrook J, Russell DW . Molecular Cloning – A Laboratory Manual. 3 edn. Cold Spring Harbor Laboratory Press, New York, 2001.
Acknowledgements
We thank Dr Ralf Bützow and Dr Unkila-Kallio for help and collection of the human OGCT samples. We thank Dr Denis Mestivier for his advice on clustering analysis of the OGCT microarray samples. BAB was funded by AMN/Université Paris Diderot-Paris 7 fellowships. MA was funded by a grant from The Helsinki University Central Hospital Research Funds. NA and MH were funded by Academy of Finland, Sigrid Juselius Foundation and Helsinki University Central Hospital. DL was funded by Université Paris Diderot-Paris 7. MBB was funded by CNRS and Université Lyon 1. RAV was funded by Institut Universitaire de France, Fondation pour la Recherche Médicale, Ligue Nationale Contre le Cancer (Comité de Paris), Groupement d'Entreprises Francaises dans la Lutte contre le Cancer, CNRS and Université Paris VII. This work was supported by grants from Fondation pour la Recherche Médicale (FRM), from The Helsinki University Central Hospital Research Funds and from Academy of Finland, Sigrid Juselius Foundation and Helsinki University Central Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Benayoun, B., Anttonen, M., L’Hôte, D. et al. Adult ovarian granulosa cell tumor transcriptomics: prevalence of FOXL2 target genes misregulation gives insights into the pathogenic mechanism of the p.Cys134Trp somatic mutation. Oncogene 32, 2739–2746 (2013). https://doi.org/10.1038/onc.2012.298
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.298
Keywords
This article is cited by
-
New STAT3-FOXL2 pathway and its function in cancer cells
BMC Molecular and Cell Biology (2019)
-
Molecular manipulation of keratin 8/18 intermediate filaments: modulators of FAS-mediated death signaling in human ovarian granulosa tumor cells
Journal of Ovarian Research (2016)
-
The four and a half LIM domains 2 (FHL2) regulates ovarian granulosa cell tumor progression via controlling AKT1 transcription
Cell Death & Disease (2016)
-
Combined comparative genomic hybridization and transcriptomic analyses of ovarian granulosa cell tumors point to novel candidate driver genes
BMC Cancer (2015)
-
FOXL2 posttranslational modifications mediated by GSK3β determine the growth of granulosa cell tumours
Nature Communications (2014)