Abstract
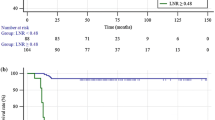
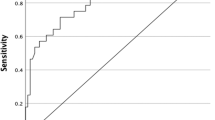
The purpose of this study was to demonstrate the utility of a personalized risk stratification and radioactive iodine (RAI) selection protocol (PRSP) using post-operative stimulated thyroglobulin (Stim-Tg) and neck ultrasound in low- and intermediate-risk papillary thyroid carcinoma (PTC) patients. Patients with PTC tumors ≥1 cm were prospectively followed after total thyroidectomy and selective therapeutic central compartment neck dissection. Low/intermediate risk was defined as PTC confined to the thyroid or central (level VI) lymph nodes. Stim-Tg and neck ultrasound were performed approximately 3 months after surgery and used to guide RAI selection. Patients with Stim-Tg < 1 µg/L did not receive RAI, while those with Stim-Tg >5 µg/L routinely did. Those with Stim-Tg 1–5 µg/L received RAI on the basis of several clinical risk factors. Patients were followed for >6 years with serial neck ultrasound and basal/stimulated thyroglobulin. Among the 129 patients, 84 (65 %) had undetectable Stim-Tg after initial surgery, 40 (31 %) had Stim-Tg of 1–5 µg/L, and 5 (4 %) had Stim-Tg >5 µg/L. RAI was administered to 8 (20 %) patients with Stim-Tg 1–5 µg/L and 5 (100 %) with Stim-Tg >5 µg/L. Using this approach, RAI therapy was avoided in 17/20 (85 %) patients with tumors >4 cm, in 72/81 (89 %) patients older than 45 years, and in 6/9 (67 %) patients with central lymph node involvement. To date, 116 (90 %) patients in this cohort have not received RAI therapy with no evidence of residual/recurrent disease, whereas among the 13 patients who received RAI, 1 (8 %) had pathologic residual/recurrence disease. Using the proposed PRSP, RAI can be avoided in the majority of low/intermediate-risk PTC patients. Moreover, traditional risk factors considered to favor RAI treatment were not always concordant with the PRSP and may lead to overtreatment.

Similar content being viewed by others
References
S. Orlov, J.L. Freeman, P.G. Walfish, Radioiodine ablation in low-risk thyroid cancer. N Engl J Med 367(7), 672 (2012). doi:10.1056/NEJMc1206712#SA1. author reply 673-675
P.W. Rosario, A.F. Mineiro, B.S. Prates, L.C. Silva, M.R. Calsolari, Postoperative Stimulated Thyroglobulin </= 1 ng/ml as a Criterion to Spare Low-Risk Patients with Papillary Thyroid Cancer from Radioiodine Ablation. Thyroid (2012). doi:10.1089/thy.2012-0190
P.W. Rosario, A.C. Xavier, M.R. Calsolari, Value of postoperative thyroglobulin and ultrasonography for the indication of ablation and (1)(3)(1)I activity in patients with thyroid cancer and low risk of recurrence. Thyroid 21(1), 49–53 (2011). doi:10.1089/thy.2010.0145
A.M. Sawka, J.D. Brierley, R.W. Tsang, L. Thabane, L.Rotstein, A. Gafni, S.Straus, D.P.Goldstein, An updated systematic review and commentary examining the effectiveness of radioactive iodine remnant ablation in well-differentiated thyroid cancer. Endocrinol. Metab. Clin. North Am. 37(2), 457–480, x (2008). doi:10.1016/j.ecl.2008.02.007
A.M. Sawka, K. Thephamongkhol, M. Brouwers, L. Thabane, G. Browman, H.C. Gerstein, Clinical review 170: a systematic review and metaanalysis of the effectiveness of radioactive iodine remnant ablation for well-differentiated thyroid cancer. J. Clin. Endocrinol. Metab. 89(8), 3668–3676 (2004). doi:10.1210/jc.2003-031167
D.S. Cooper, G.M. Doherty, B.R. Haugen, R.T. Kloos, S.L. Lee, S.J. Mandel, E.L. Mazzaferri, B. McIver, F. Pacini, M. Schlumberger, S.I. Sherman, D.L. Steward, R.M. Tuttle, Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19(11), 1167–1214 (2009). doi:10.1089/thy.2009.0110
P. Perros, K. Boelaert, S. Colley, C. Evans, G. Gerrard Ba, J. Gilbert, B. Harrison, S.J. Johnson, T.E. Giles, L. Moss, V. Lewington, K. Newbold, J. Taylor, R.V. Thakker, J. Watkinson, G.R. Williams, Guidelines for the management of thyroid cancer. Clin. Endocrinol. 81(Suppl 1), 1–122 (2014). doi:10.1111/cen.12515
K.A. Heemstra, Y.Y. Liu, M. Stokkel, J. Kievit, E. Corssmit, A.M. Pereira, J.A. Romijn, J.W. Smit, Serum thyroglobulin concentrations predict disease-free remission and death in differentiated thyroid carcinoma. Clin. Endocrinol. 66(1), 58–64 (2007). doi:10.1111/j.1365-2265.2006.02685.x
T.Y. Kim, W.B. Kim, E.S. Kim, J.S. Ryu, J.S. Yeo, S.C. Kim, S.J. Hong, Y.K. Shong, Serum thyroglobulin levels at the time of 131I remnant ablation just after thyroidectomy are useful for early prediction of clinical recurrence in low-risk patients with differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 90(3), 1440–1445 (2005). doi:10.1210/jc.2004-1771
A.M. Sawka, S. Orlov, J. Gelberg, B. Stork, M. Dowar, M. Shaytzag, V. Tabatabaie, J.L. Freeman, P.G. Walfish, Prognostic value of postsurgical stimulated thyroglobulin levels after initial radioactive iodine therapy in well-differentiated thyroid carcinoma. Head Neck 30(6), 693–700 (2008). doi:10.1002/hed.20755
A. Vaisman, S. Orlov, J. Yip, C. Hu, T. Lim, M. Dowar, J.L. Freeman, P.G. Walfish, Application of post-surgical stimulated thyroglobulin for radioiodine remnant ablation selection in low-risk papillary thyroid carcinoma. Head Neck 32(6), 689–698 (2010). doi:10.1002/hed.21371
W. Sacks, C.H. Fung, J.T. Chang, A. Waxman, G.D. Braunstein, The effectiveness of radioactive iodine for treatment of low-risk thyroid cancer: a systematic analysis of the peer-reviewed literature from 1966 to April 2008. Thyroid 20(11), 1235–1245 (2010). doi:10.1089/thy.2009.0455
M.R. Haymart, M. Banerjee, A.K. Stewart, R.J. Koenig, J.D. Birkmeyer, J.J. Griggs, Use of radioactive iodine for thyroid cancer. JAMA 306(7), 721–728 (2011). doi:10.1001/jama.2011.1139
A.M. Sawka, D.P. Goldstein, L. Thabane, J.D. Brierley, R.W. Tsang, L. Rotstein, S. Kamalanathan, B. Zhao, A. Gafni, S. Straus, S. Ezzat, Basis for physician recommendations for adjuvant radioiodine therapy in early-stage thyroid carcinoma: principal findings of the Canadian-American thyroid cancer survey. Endocr. pract. 14(2), 175–184 (2008)
C. Nascimento, I. Borget, A. Al Ghuzlan, D. Deandreis, L. Chami, J.P. Travagli, D. Hartl, J. Lumbroso, C. Chougnet, L. Lacroix, E. Baudin, M. Schlumberger, S. Leboulleux, Persistent disease and recurrence in differentiated thyroid cancer patients with undetectable postoperative stimulated thyroglobulin level. Endocr. Relat. Cancer 18(2), R29–R40 (2011). doi:10.1677/ERC-10-0292
R.C. Webb, R.S. Howard, A. Stojadinovic, D.Y. Gaitonde, M.K. Wallace, J. Ahmed, H.B. Burch, The utility of serum thyroglobulin measurement at the time of remnant ablation for predicting disease-free status in patients with differentiated thyroid cancer: a meta-analysis involving 3947 patients. J Clin Endocrinol Metab 97(8), 2754–2763 (2012). doi:10.1210/jc.2012-1533
L. Giovanella, G. Treglia, R. Sadeghi, P. Trimboli, L. Ceriani, F.A. Verburg, Unstimulated highly sensitive thyroglobulin in follow-up of differentiated thyroid cancer patients: a meta-analysis. J. Clin. Endocrinol. Metab. 99(2), 440–447 (2014). doi:10.1210/jc.2013-3156
F.L. Greene, American Cancer Society, American College of Surgeons, American Joint Committee on Cancer, AJCC cancer staging manual, 6th edn. (Springer, New York, 2002)
A. Toft, G. Beckett, Use of recombinant thyrotropin. Lancet 359(9321), 1874–1875 (2002). doi:10.1016/S0140-6736(02)08785-8
A. Golger, T.R. Fridman, S. Eski, I.J. Witterick, J.L. Freeman, P.G. Walfish, Three-week thyroxine withdrawal thyroglobulin stimulation screening test to detect low-risk residual/recurrent well-differentiated thyroid carcinoma. J. Endocrinol. Invest. 26(10), 1023–1031 (2003)
F.T. Hall, N.J. Beasley, S.J. Eski, I.J. Witterick, P.G. Walfish, J.L. Freeman, Predictive value of serum thyroglobulin after surgery for thyroid carcinoma. Laryngoscope 113(1), 77–81 (2003). doi:10.1097/00005537-200301000-00014
J. Yip, S. Orlov, D. Orlov, A. Vaisman, K.G. Hernandez, D. Etarsky, I. Kak, N. Parvinnejad, J.L. Freeman, P.G. Walfish, Predictive value of metastatic cervical lymph node ratio in papillary thyroid carcinoma recurrence. Head Neck 35(4), 592–598 (2013). doi:10.1002/hed.23047
M.G. Castagna, L. Brilli, T. Pilli, A. Montanaro, C. Cipri, C. Fioravanti, F. Sestini, M. Capezzone, F. Pacini, Limited value of repeat recombinant human thyrotropin (rhTSH)-stimulated thyroglobulin testing in differentiated thyroid carcinoma patients with previous negative rhTSH-stimulated thyroglobulin and undetectable basal serum thyroglobulin levels. J. Clin. Endocrinol. Metab. 93(1), 76–81 (2008). doi:10.1210/jc.2007-1404
R.T. Kloos, E.L. Mazzaferri, A single recombinant human thyrotropin-stimulated serum thyroglobulin measurement predicts differentiated thyroid carcinoma metastases three to five years later. J. Clin. Endocrinol. Metab. 90(9), 5047–5057 (2005). doi:10.1210/jc.2005-0492
K. Gomez Hernandez, D. Etarsky, S. Orlov, P.G. Walfish, Stimulated thyroglobulin and neck ultrasonography facilitates postsurgical radioactive iodine remnant ablation selection in patients with low-risk well-differentiated thyroid carcinoma. Thyroid 22(7), 760–761 (2012). doi:10.1089/thy.2011.0453
A. Iervasi, G. Iervasi, M. Ferdeghini, C. Solimeo, A. Bottoni, L. Rossi, C. Colato, G.C. Zucchelli, Clinical relevance of highly sensitive Tg assay in monitoring patients treated for differentiated thyroid cancer. Clin. Endocrinol. 67(3), 434–441 (2007). doi:10.1111/j.1365-2265.2007.02907.x
R.C. Smallridge, S.E. Meek, M.A. Morgan, G.S. Gates, T.P. Fox, S. Grebe, V. Fatourechi, Monitoring thyroglobulin in a sensitive immunoassay has comparable sensitivity to recombinant human tsh-stimulated thyroglobulin in follow-up of thyroid cancer patients. J. Clin. Endocrinol. Metab 92(1), 82–87 (2007). doi:10.1210/jc.2006-0993
T.E. Angell, C.A. Spencer, B.D. Rubino, J.T. Nicoloff, J.S. LoPresti, In search of an unstimulated thyroglobulin baseline value in low-risk papillary thyroid carcinoma patients not receiving radioactive iodine ablation. Thyroid 24(7), 1127–1133 (2014). doi:10.1089/thy.2013.0691
Acknowledgments
This study was supported in part by unrestricted educational grants from Abbott Laboratories Limited Canada; the Da Vinci Gala Fundraiser; the George Knudson Oakdale Pro-Am Charity Golf Tournament; the Department of Medicine Research Fund, Mount Sinai Hospital; the Mount Sinai Hospital Foundation of Toronto; and the Alex and Simona Shnaider Research Chair in Thyroid Oncology.
Conflict of interest
PGW receives consulting fees from Genzyme Canada, a Sanofi company. SO, FS, JLF, AV, and IJW have no relevant disclosures of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Orlov, S., Salari, F., Kashat, L. et al. Post-operative stimulated thyroglobulin and neck ultrasound as personalized criteria for risk stratification and radioactive iodine selection in low- and intermediate-risk papillary thyroid cancer. Endocrine 50, 130–137 (2015). https://doi.org/10.1007/s12020-015-0575-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-015-0575-0