Abstract
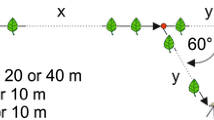
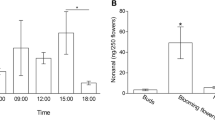
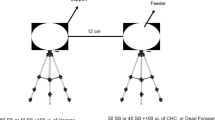
The pheromones used by several species of stingless bees for scent trail communication are generally assumed to be produced by the mandibular glands. Here we present strong evidence that in Trigona recursa these pheromones originate from the labial glands, which are well developed in the heads of foragers. Analysis of the behavior involved in scent marking shows that a bee extends her proboscis and rubs it over the substrate. A single scent marking event lasts for 0.59±0.21 s while the bee runs a stretch of 1.04±0.37 cm on a leaf. According to choice experiments the bees are attracted by a feeder baited with labial gland extract (84.2±6% of the bees choose this feeder) but repelled from a feeder baited with mandibular gland extract (only 27.5±13.1% of the bees choose this feeder). They do not discriminate between two clean feeders (49.6±3% of the bees at a feeder). 87±5.1% of bees already feeding leave the feeder after the application of mandibular gland extract whereas only 6.2±4.9% and 2.6±4% do so when labial gland extract or pure solvent was applied.




Similar content being viewed by others
References
Bergman P, Bergström G (1997) Scent marking, scent origin, and species specificity in male premating behavior of two Scandinavian bumblebees. J Chem Ecol 23:1235–1251
Biesmeijer JC, Vries H de (2001) Exploration and exploitation of food sources by social insect colonies: a revision of the scout-recruit concept. Behav Ecol Sociobiol 49:89–99
Blum MS, Brand JM (1972) Social insect pheromones: their chemistry and function. Am Zool 12:553–576
Blum MS, Crewe RM, Kerr WE, Keith LH, Garrison AW, Walker MM (1970) Citral in stingless bees: isolation and functions in trail-laying and robbing. J Insect Physiol 16:1637–1648
Cruz Landim C da (1967) Estudo comparativo de algumas glândulas das abelhas (Hymenoptera, Apoidea) e respectivas implicações evolutivas. Arq Zool S Paulo15:177–290
Esch H (1967) Die Bedeutung der Lauterzeugung für die Verständigung der stachellosen Bienen. Z Vergl Physiol 56:199–220
Jarau S, Hrncir M, Schmidt VM, Zucchi R, Barth FG (2003) Effectiveness of recruitment behavior in stingless bees (Apidae, Meliponini). Insectes Soc 50:365–374
Johnson LK (1987) Communication of food source location by the stingless bee Trigona fulviventris. In: Eder J, Rembold H (eds.) Chemistry and biology of social insects. Peperny, München, pp 698–699
Johnson LK, Wiemer DF (1982) Nerol: an alarm substance of the stingless bee, Trigona fulviventris (Hymenoptera: Apidae). J Chem Ecology 8:1167–1181
Johnson LK, Haynes LW, Carlson MA, Fortnum HA, Gorgas DL (1985) Alarm substances of the stingless bee, Trigona silvestriana. J Chem Ecology 11:409–416
Keeping MG, Crewe RM, Field BI (1982) Mandibular gland secretions of the old world stingless bee, Trigona gribodoi Magretti: isolation, identification, and compositional changes with age. J Apic Res 21:65–73
Kerr WE, Costa Cruz C da (1961) Funções diferentes tomadas pela glândula mandibular na evolução das abelhas em geral e em “Trigona (Oxytrigona) tataira” em especial. Rev Brasil Biol 21:1–16
Kerr WE, Esch H (1965) Comunicação entre as abelhas sociais brasileiras e sua contribuição para o entendimento da sua evolução. Ciên Cult (Sao Paolo) 17:529–538
Kerr WE, Ferreira A, Mattos NS de (1963) Communication among stingless bees—additional data (Hymenoptera: Apidae). J N Y Entomol Soc 71:80–90
Kerr WE, Blum M, Fales HM (1981) Communication of food source between workers of Trigona (Trigona) spinipes. Rev Brasil Biol 41:619–613
Korte M de, Weissenbacher KH, Crewe RM (1988) Chemical signals in a stingless bee Trigona (Meliplebeia) denoiti Vachal (Hymenoptera: Apidae: Meliponinae). J Entomol Soc South Afr 51:9–16
Kullenberg B, Bergström G, Bringer B, Carlberg B, Cederberg B (1973) Observations on scent marking by Bombus Latr. and Psithyrus Lep. males (Hym., Apidae) and localization of site of production of the secretion. Zoon [Suppl] 1:23–30
Lindauer M, Kerr WE (1958) Die gegenseitige Verständigung bei den stachellosen Bienen. Z Vergl Physiol 41:405–434
Lindauer M, Kerr WE (1960) Communication between the workers of stingless bees. Bee World 41:29–41, 65–71
Luby JM, Regnier FE, Clarke ET, Weaver EC, Weaver N (1973) Volatile cephalic substances of the stingless bees, Trigona mexicana and Trigona pectoralis. J Insect Physiol 19:1111–1127
Michener CD (1974) The Social behavior of the bees: a comparative study. Harvard University Press, Cambridge, Massachusetts
Michener CD (2000) The bees of the world. Johns Hopkins University Press, Baltimore
Nieh JC, Contrera FAL, Nogueira-Neto P (2003) Pulsed mass recruitment by a stingless bee, Trigona hyalinata. Proc R Soc London Ser B 270:2191–2196
Noll FB (2002) Behavioral phylogeny of corbiculate Apidae (Hymenoptera; Apinae), with special reference to social behavior. Cladistics 18:137–153
Roubik DW (1989) Ecology and natural history of tropical bees. Cambridge University Press, Cambridge
Schmidt VM, Zucchi R, Barth FG (2003) A stingless bee marks the feeding site in addition to the scent path (Scaptotrigona aff. depilis Moure 1942). Apidologie 34:237–248
Silva de Moraes RLM (2002) Glândulas salivares do adulto. In: Cruz Landim C da, Camargo Abdalla F (eds) Glândulas Exócrinas das Abelhas. FUNPEC—Editora, Ribeirão Preto São Paulo, pp 51–70
Simpson J, Riedel IBM (1964) Discharge and manipulation of labial gland secretion by workers of Apis mellifera (L.) (Hymenoptera: Apidae). Proc R Entomol Soc London Ser A 39:76–82
Smith BH, Roubik DW (1983) Mandibular glands of stingless bees (Hymenoptera: Apidae): chemical analysis of their contents and biological function in two species of Melipona. J Chem Ecol 9:1465–1472
Weaver N, Weaver EC, Clarke ET (1975) Reactions of five species of stingless bees to some volatile chemicals and to other species of bees. J Insect Physiol 21:479–494
Wilson EO (1965) Chemical communication in the social insects. Science 149:1064–1071
Wilson EO (1971) The insect societies. Belknap Press of Harvard University Press, Cambridge, Massachusetts
Acknowledgements
We are very grateful to Sidnei Mateus for his valuable help in finding and collecting the bee nests in Pedregulho. This study was supported by grant P-14328 of the Austrian Science Foundation (FWF) to F.G.B. This work complies with the current laws of Brazil where the experiments were carried out.
Author information
Authors and Affiliations
Corresponding author
Addendum
Addendum
After the submission of our manuscript a paper by Nieh et al. (2003) appeared which has some relevance in regard of our main finding. According to these authors mandible extracts of T. hyalinata (concentration corresponding to 0.5 bee equivalents) offered at a feeding table released aggressive behavior in newly arriving bees for about 8 min. During this time newcomers did not land to feed. Subsequently, the bees were attracted by the extracts. On the other hand, the authors found a temporal synchronization of recruitment and odor marking. Newcomers arrived at the feeder at the same time foragers scent marked. Despite the mismatch of these findings Nieh et al. (2003) concluded that T. hyalinata recruiters deposit mandibular gland secretions to mark the scent trail.
In order to prepare the “mandibular gland” extract Nieh et al. (2003) pulled out the entire mandibles of the worker bees’ heads together with the mandibular glands and other tissues attached to them (see their Fig. 4). When using this method in T. recursa we frequently pulled out parts of the labial glands together with the mandible. A careful separation of the mandibular gland prior to its extraction is therefore necessary.
Regarding the repellent effect of the mandibular gland extracts the data of Nieh et al. (2003) and our own are in agreement. The attraction of the newcomers after eight minutes in the study by Nieh et al. (2003), however, probably was due to labial gland secretion unknowingly contained in the “mandibular gland” extract.
Rights and permissions
About this article
Cite this article
Jarau, S., Hrncir, M., Zucchi, R. et al. A stingless bee uses labial gland secretions for scent trail communication (Trigona recursa Smith 1863). J Comp Physiol A 190, 233–239 (2004). https://doi.org/10.1007/s00359-003-0489-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-003-0489-9