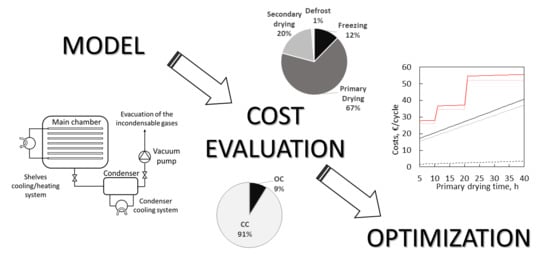
Economic Analysis of a Freeze-Drying Cycle
Abstract
:1. Introduction
2. Model Description
2.1. Power Consumption
2.1.1. Freezing
2.1.2. Primary Drying
2.1.3. Secondary Drying
2.1.4. Condenser Defrosting
2.2. Operational Costs
2.2.1. Refrigerating Apparatus
2.2.2. Other Electrical Consumptions
2.2.3. Operational Costs
2.3. Capital Costs
2.4. Case Studies
3. Results
3.1. Operational Costs in Laboratory-Scale and Industrial Apparatus
3.2. Operational vs. Capital Costs
3.3. Manufacturing Cost per Unit Dose
3.4. Refrigeration with Liquid Nitrogen
3.5. Optimization and Reduction of Primary Drying Duration
- Control of nucleation during the freezing stage;
- Optimization of shelf temperature during primary drying.
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Variable | Laboratory Freeze-Drier | Industrial Freeze-Drier | Units |
|---|---|---|---|
| nvials/shelf | 200 | 6000 | - |
| nshelf | 4 | 17 | - |
| tF | 7 | 7 | h |
| tPD | 28 | 28 | h |
| tSD | 9 | 9 | h |
| tdead time | 4 | 4 | h |
| Tamb | 25 | 25 | °C |
| Tn | 0 | 0 | °C |
| TF | −50 | −50 | °C |
| TF,walls | −15 | −15 | °C |
| TPD | −20 | −20 | °C |
| TPD,walls | 15 | 15 | °C |
| TSD | 10 | 10 | °C |
| TSD,walls | 15 | 15 | °C |
| Tcond | −70 | −70 | °C |
| PPD | 10 | 10 | Pa |
| PSD | 2 | 2 | Pa |
| Patm | 101,325 | 101,325 | Pa |
| mprod/vial | 3 | 3 | g |
| mglass/vial | 10 | 10 | g |
| mstop/vial | 1 | 1 | g |
| cp,w | 4.186 | 4.186 | kJ/kg°C |
| cp,i | 2.090 | 2.090 | kJ/kg°C |
| cp,sol | 1.383 | 1.383 | kJ/kg°C |
| cv,air | 0.715 | 0.715 | kJ/kg°C |
| cp,glass | 0.840 | 0.840 | kJ/kg°C |
| cp,stop | 0.240 | 0.240 | kJ/kg°C |
| cp,steel | 0.502 | 0.502 | kJ/kg°C |
| cp,oil | 1.5 | 1.5 | kJ/kg°C |
| λf,w | 333.6 | 333.6 | kJ/kg |
| λsub,w | 2838.0 | 2838.0 | kJ/kg |
| λdes,w | 2687.4 | 2687.4 | kJ/kg |
| λboil,LN | 198.9 | 198.9 | kJ/kg |
| ρair | 1.4 | 1.4 | kg/m3 |
| ρsteel | 8000 | 8000 | kg/m3 |
| ρoil | 971 | 971 | kg/m3 |
| ρLN | 807 | 807 | kg/m3 |
| xw | 0.95 | 0.95 | - |
| xsol | 0.05 | 0.05 | - |
| φPD | 0.92 | 0.92 | - |
| φSD | 0.08 | 0.08 | - |
| Wch | 600 | 2000 | mm |
| Lch | 600 | 2000 | mm |
| Hch | 700 | 2000 | mm |
| swalls | 10 | 20 | mm |
| Wsh | 380 | 1300 | mm |
| Lsh | 450 | 1300 | mm |
| sshelf | 4 | 4 | mm |
| soil | 12 | 12 | mm |
| sins | 50 | 100 | mm |
| sdoor | 40 | 50 | mm |
| ksteel | 15 | 15 | W/m°C |
| kins | 0.03 | 0.03 | W/m°C |
| kdoor | 0.20 | 0.20 | W/m°C |
| hin | 5 | 5 | W/m2°C |
| hout | 20 | 20 | W/m2°C |
| PM | 3.70 | 40 | kW |
| Pn | 0.75 | 9.2 | kW |
| Vcond | 0.012 | 1 | m3 |
| pump | 20.5 | 200 | m3/h |
| ηel | 0.5 | 0.5 | - |
| ηis | 0.7 | 0.7 | - |
| h1 | 323 | 323 | kJ/kg |
| h2 | 423 | 423 | kJ/kg |
| h3 | 168 | 168 | kJ/kg |
| h4 | 168 | 168 | kJ/kg |
| h5 | 349 | 349 | kJ/kg |
| h6 | 406 | 406 | kJ/kg |
| h7 | 251 | 251 | kJ/kg |
| h8 | 251 | 251 | kJ/kg |
| tdep | 20 | 20 | y |
| nw/y | 48 | 48 | w |
| CLN | 100 | 100 | €/m3 |
| Cel | 0.06568 | 0.06568 | €/kWh |
| Cfreeze-dryer | 100 | 2000 | k€ |
References
- Rey, L. Glimpses into the Realm of Freeze-Drying: Classical Issues and New Ventures. In Freeze Drying/Lyophilization of Pharmaceuticals and Biological Products, 3rd ed.; Rey, L., May, J.C., Eds.; Informa Healthcare: London, UK, 2010; pp. 1–28. [Google Scholar]
- Oetjen, G.W.; Haseley, P.U. Freeze-Drying, 2nd ed.; John Wiley & Sons: New York, NY, USA, 2008. [Google Scholar]
- Ratti, C. Freeze-Drying Process Design. In Handbook of Food Process Design; Rahman, S., Ahmed, J., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; pp. 621–647. [Google Scholar]
- Zhang, S.Z.; Luo, J.L.; Chen, G.M.; Wang, Q. Thermodynamic Analysis of a Freeze-Dryer Utilizing Hygroscopic Solution. Dry. Technol. 2018, 36, 697–708. [Google Scholar]
- Bruttini, R.; Crosser, O.K.; Liapis, A.I. Exergy Analysis for the Freezing Stage of the Freeze Drying Process. Dry. Technol. 2001, 19, 2303–2313. [Google Scholar] [CrossRef]
- Liapis, A.I.; Bruttini, R. Exergy Analysis of Freeze Drying of Pharmaceuticals in Vials on Trays. Int. J. Heat Mass Transf. 2008, 51, 3854–3868. [Google Scholar] [CrossRef]
- Renteria Gamiz, A.G.; Van Bockstal, P.J.; De Meester, S.; De Beer, T.; Corver, J.; Dewulf, J. Analysis of a pharmaceutical batch freeze dryer: Resource consumption, hotspots, and factors for potential improvement. Dry. Technol. 2019, 37, 1563–1582. [Google Scholar] [CrossRef]
- Duan, X.; Yang, X.T.; Ren, G.Y.; Pang, Y.Q.; Liu, L.L.; Liu, Y.H. Technical Aspects in Freeze-Drying of Foods. Dry. Technol. 2016, 34, 1271–1285. [Google Scholar] [CrossRef]
- Ratti, C. Hot air and freeze-drying of high-value foods: A review. J. Food Eng. 2001, 49, 311–319. [Google Scholar] [CrossRef]
- Millman, M.; Liapis, A.; Marchello, J. Note on the economics of batch freeze dryers. Int. J. Food Sci. Technol. 1985, 5, 541–551. [Google Scholar] [CrossRef]
- Bird, K. Freeze-Drying of Foods: Cost Projections; Marketing Research Report n.639; Marketing Economics Division, Economic Research Service; US Department of Agriculture: Washington, DC, USA, 1964. [Google Scholar]
- Arsiccio, A.; Giorsello, P.; Marenco, L.; Pisano, R. Considerations on Protein Stability During Freezing and Its Impact on the Freeze-Drying Cycle: A Design Space Approach. J. Pharm. Sci. 2019, 109, 464–475. [Google Scholar] [CrossRef] [Green Version]
- Arsiccio, A.; Pisano, R. Application of the Quality by Design Approach to the Freezing Step of Freeze-Drying: Building the Design Space. J. Pharm. Sci. 2018, 107, 1586–1596. [Google Scholar] [CrossRef]
- Pisano, R.; Arsiccio, A.; Nakagawa, K.; Barresi, A.A. Tuning, measurement and prediction of the impact of freezing on product morphology: A step toward improved design of freeze-drying cycles. Dry. Technol. 2019, 37, 579–599. [Google Scholar] [CrossRef]
- Oddone, I.; Arsiccio, A.; Duru, C.; Malik, K.; Ferguson, J.; Pisano, R.; Matejtschuk, P. Vacuum-Induced Surface Freezing for the Freeze-Drying of the Human Growth Hormone: How Does Nucleation Control Affect Protein Stability? J. Pharm. Sci. 2020, 109, 254–263. [Google Scholar] [CrossRef] [Green Version]
- Fissore, D.; Pisano, R. Computer-Aided Framework for the Design of Freeze-Drying Cycles: Optimization of the Operating Conditions of the Primary Drying Stage. Processes 2015, 3, 406. [Google Scholar] [CrossRef]
- Vilas, C.; Alonso, A.A.; Balsa-Canto, E.; López-Quiroga, E.; Trelea, I.C. Model-Based Real Time Operation of the Freeze-Drying Process. Processes 2020, 8, 325. [Google Scholar] [CrossRef] [Green Version]
- Vanbillemont, B.; Nicolaï, N.; Leys, L.; De Beer, T. Model-Based Optimisation and Control Strategy for the Primary Drying Phase of a Lyophilisation Process. Pharmaceutics 2020, 12, 181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisano, R.; Arsiccio, A.; Capozzi, L.C.; Trout, B.L. Achieving continuous manufacturing in lyophilization: Technologies and approaches. Eur. J. Pharm. Biopharm. 2019, 142, 265–279. [Google Scholar] [CrossRef]
- Adali, M.B.; Barresi, A.A.; Boccardo, G.; Pisano, R. Spray Freeze-Drying as a Solution to Continuous Manufacturing of Pharmaceutical Products in Bulk. Processes 2020, 8, 709. [Google Scholar] [CrossRef]
- Pisano, R. Continuous manufacturing of lyophilized products: Why and how to make it happen. Am. Pharm. Rev. 2020, 23, 20–22. [Google Scholar]
- Maggard, B.N.; Rhyne, D.M. Total productive maintenance. A timely integration of production and maintenance. Prod. Inventory Manag. J. 1992, 33, 6–10. [Google Scholar]
- Löfsten, H. Measuring maintenance performance—In search for a maintenance productivity index. Int. J. Prod. Econ. 2000, 63, 47–58. [Google Scholar] [CrossRef]
- Vishnu, C.R.; Regikumar, V. Reliability Based Maintenance Strategy Selection in Process Plants: A Case Study. Proc. Technol. 2016, 25, 1080–1087. [Google Scholar] [CrossRef] [Green Version]
- Bevilacqua, M.; Braglia, M. The analytic hierarchy process applied to maintenance strategy selection. Reliab. Eng. Syst. Saf. 2000, 70, 71–83. [Google Scholar] [CrossRef]
- Kochs, M.; Körber, C.; Nunner, B.; Heschel, I. The influence of the freezing process on vapour transport during sublimation in vacuum-freeze-drying. Int. J. Heat Mass Transf. 1991, 34, 2395–2408. [Google Scholar] [CrossRef]
- Searles, J.A.; Carpenter, J.F.; Randolph, T.W. The Ice Nucleation Temperature Determines the Primary Drying Rate of Lyophilization for Samples Frozen on a Temperature-Controlled Shelf. J. Pharm. Sci. 2001, 90, 860–871. [Google Scholar] [CrossRef]
- Hottot, A.; Vessot, S.; Andrieu, J. Freeze drying of pharmaceuticals in vials: Influence of freezing protocol and sample configuration on ice morphology and freeze-dried cake texture. Chem. Eng. Process. 2007, 46, 666–674. [Google Scholar] [CrossRef]
- Kasper, J.C.; Friess, W. The freezing step in lyophilisation: Physico-chemical fundamentals, freezing methods and consequences on process performance and quality attributes of biopharmaceuticals. Eur. J. Pharm. Biopharm. 2011, 78, 248–263. [Google Scholar] [CrossRef] [PubMed]
- Arsiccio, A.; Matejtschuk, P.; Ezeajughi, E.; Riches-Duit, A.; Bullen, A.; Malik, K.; Raut, S.; Pisano, R. Impact of controlled vacuum induced surface freezing on the freeze drying of human plasma. Int. J. Pharm. 2020, 582, 119290. [Google Scholar] [CrossRef]
- Arsiccio, A.; Barresi, A.; De Beer, T.; Oddone, I.; Van Bockstal, P.J.; Pisano, R. Vacuum Induced Surface Freezing as an effective method for improved inter- and intra-vial product homogeneity. Eur. J. Pharm. Biopharm. 2018, 128, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Pisano, R.; Fissore, D.; Barresi, A. Freeze-Drying Cycle Optimization Using Model Predictive Control Techniques. Ind. Eng. Chem. Res. 2011, 50, 7363–7379. [Google Scholar] [CrossRef]
- Fissore, D.; Pisano, R.; Barresi, A. Advanced Approach to Build the Design Space for the Primary Drying of a Pharmaceutical Freeze-Drying Process. J. Pharm. Sci. 2011, 100, 4922–4933. [Google Scholar] [CrossRef]
- Fissore, D.; Pisano, R.; Barresi, A. Applying quality-by-design to develop a coffee freeze-drying process. J. Food Eng. 2014, 123, 179–187. [Google Scholar] [CrossRef] [Green Version]
- Pikal, M.J.; Shah, S.; Roy, M.L.; Putman, R. The secondary drying of freeze-drying: Drying kinetics as a function of the temperature and chamber pressure. Int. J. Pharm. 1990, 60, 203–217. [Google Scholar] [CrossRef]





| Freeze-Dryer | OC, €/cycle | CC, €/cycle | Total Cost, €/cycle | Total Cost, €/dose |
|---|---|---|---|---|
| Laboratory | 3.28 | 29.76 | 33.04 | 0.041 |
| Industrial | 107.27 | 595.24 | 702.51 | 0.007 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stratta, L.; Capozzi, L.C.; Franzino, S.; Pisano, R. Economic Analysis of a Freeze-Drying Cycle. Processes 2020, 8, 1399. https://doi.org/10.3390/pr8111399
Stratta L, Capozzi LC, Franzino S, Pisano R. Economic Analysis of a Freeze-Drying Cycle. Processes. 2020; 8(11):1399. https://doi.org/10.3390/pr8111399
Chicago/Turabian StyleStratta, Lorenzo, Luigi C. Capozzi, Simone Franzino, and Roberto Pisano. 2020. "Economic Analysis of a Freeze-Drying Cycle" Processes 8, no. 11: 1399. https://doi.org/10.3390/pr8111399