Advanced Nanobiomaterials: Vaccines, Diagnosis and Treatment of Infectious Diseases
Abstract
:1. Introduction
2. Nanoparticles and Vaccines
- (i)
- Polymers, divided in turn into nanoparticles containing synthetic polymers, such as poly(d,l-lactic-co-glycolic acid)(PLGA) [21], polyethylene glycol (PGE) [22] or polyester bio-beads [23], and natural polymers based on polysaccharides such as alginate [24], inulin [25] or chitosan [26]. Synthetic and natural polymers have been used to synthesize hydrogel nanoparticles, which have favorable properties including but not limited to flexible mesh size, large surface area for multivalent conjugation, high water content, and high antigen loading capacity [27];
- (ii)
- Liposomes, which are biodegradable and non-toxic phospholipids. They encapsulate antigens and incorporate viral envelope glycoproteins to form virosomes. The combination of a modified cationic liposome and a cationic polymer (such as protamine)-condensed DNA is called liposome-polycation-DNA nanoparticles (LPD). They are commonly used as adjuvant delivery system in DNA vaccine studies [28];
- (iii)
- Nanosized emulsions are those nanoparticles that can exist as oil-in-water or water-in-oil form. Emulsions can carry antigens inside their core to increase the efficiency of vaccine delivery or they can also be simply mixed with antigen [20];
- (iv)
- Inorganic nanoparticles are non-biodegradable, they have rigid structure and controllable synthesis. Silica-based nanoparticles (SiNPs) offer the advantage of biocompatibility and have excellent properties as nanocarriers. SiNPs particles such as mesoporous silica nanoparticles (MSNs) could potentially become high-efficient, controlled-release nanocarriers in future vaccine formulations [20].
- (v)
- Immuno-stimulating complexes (ISCOM). They are composed of supra-molecular structures of the adjuvant Quil A and immunizing peptides, which allows selective incorporation of viral envelope proteins by hydrophobic interaction [29].
- (vi)
- Virus-like particles (VLP) are optimized for interaction with the immune system, avoiding the infectious components. They can induce potent immune responses, even in the absence of adjuvant [30]. VLP based vaccines have been the first nanoparticle class to reach market [31], found for example under the following Engerix®, RECOMBIVAX®HB against to HBV [32].
- (vii)
- Self-assembling systems emerged as a consequence of an attempt to drive higher levels of protein, and consequently better immunological properties. A variety of natural proteins can be self-assembled into nanoparticles, conferring highly symmetric, stable and organized structure [32].
3. Microbial Detection by Using Nanoparticles
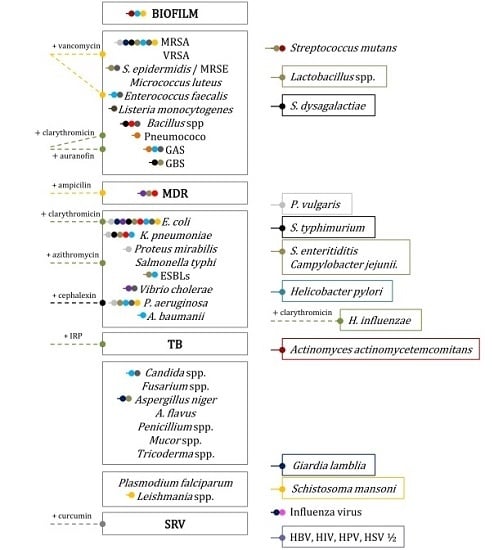
4. Nanoparticles for Fighting Superbugs
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACP | Antigen Presenting Cells |
| Ag | Antigen |
| BT | Botulinum toxin |
| CF | Cystic fibrosis |
| CFU | Colony Forming Unit |
| ELISA | Enzyme Linked Immunosorbent Assay |
| FSNP | Fluorescent Silica Nanoparticles |
| HBV | Hepatitis B Virus |
| HCV | Hepatitis C Virus |
| HIV | Human Immunodeficiency Virus |
| Ig | Immunoglobulin |
| IONP | Iron oxide nanoparticles |
| MALDI-TOF MS | Matrix Assisted Laser Desorption Ionization—Time of Flight Mass Spectrometry |
| MNP | Magnetic nanoparticles |
| QD | Quantum dots |
| ROS | Reactive Oxygen Species |
| SiNP | Silica-based nanoparticles |
| VLP | Virus-like particles |
| WHO | Word Health Organization |
References
- Feynman, R.P. Plenty of room at the bottom. Am. Phy. Soc. 1959. Available online: http://www.pa.msu.edu/~yang/RFeynman_plentySpace.pdf (accessed on 30 June 2016). [Google Scholar]
- Plenty of room’ revisited. Nat. Nanotech. 2009, 4, 781. [CrossRef]
- Drexler, K.E. Molecular engineering: An approach to the development of general capabilities for molecular manipulation. Proc. Natl. Acad. Sci. USA 1981, 78, 5275–5278. [Google Scholar] [CrossRef] [PubMed]
- Drexler, K.E. Engines of Creation: The Coming Era of Nanotechnology; Doubleday: New York City, NY, USA, 1986. [Google Scholar]
- Savage, N.; Thomas, T.A.; Duncan, J.S. Nanotechnology applications and implications research supported by the US environmental protection agency star grants program. J. Environ. Monit. 2007, 9, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Medina, C.; Santos-Martinez, M.J.; Radomski, A.; Corrigan, O.I.; Radomski, M.W. Nanoparticles: Pharmacological and toxicological significance. Br. J. Pharmacol. 2007, 150, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Jos, A.; Pichardo, S.; Puerto, M.; Sanchez, E.; Grilo, A.; Camean, A.M. Cytotoxicity of carboxylic acid functionalized single wall carbon nanotubes on the human intestinal cell line caco-2. Toxicol. In Vitro 2009, 23, 1491–1496. [Google Scholar] [CrossRef] [PubMed]
- Holban, A.M.; Gestal, M.C.; Grumezescu, A.M. Control of biofilm-associated infections by signaling molecules and nanoparticles. Int. J. Pharm. 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Holban, A.M.; Gestal, M.C.; Grumezescu, A.M. New molecular strategies for reducing implantable medical devices associated infections. Current Med. Chem. 2014, 21, 3375–3382. [Google Scholar] [CrossRef]
- Lara, H.H.; Romero-Urbina, D.G.; Pierce, C.; Lopez-Ribot, J.L.; Arellano-Jimenez, M.J.; Jose-Yacaman, M. Effect of silver nanoparticles on candida albicans biofilms: An ultrastructural study. J. Nanobiotechnol. 2015, 13, 91. [Google Scholar] [CrossRef] [PubMed]
- Veigas, B.; Fernandes, A.R.; Baptista, P.V. Aunps for identification of molecular signatures of resistance. Front. Microbiol. 2014, 5, 455. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, C.A.; Letsinger, R.L.; Mucic, R.C.; Storhoff, J.J. A DNA-based method for rationally assembling nanoparticles into macroscopic materials. Nature 1996, 382, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.C.; Nam, J.M.; Mirkin, C.A.; Hupp, J.T. Real-time multicolor DNA detection with chemoresponsive diffraction gratings and nanoparticle probes. J. Am. Chem. Soc. 2003, 125, 13541–13547. [Google Scholar] [CrossRef] [PubMed]
- Salvador, A.; Igartua, M.; Hernandez, R.M.; Pedraz, J.L. An overview on the field of micro- and nanotechnologies for synthetic peptide-based vaccines. J. Drug Deliv. 2011, 2011, 181646. [Google Scholar] [CrossRef] [PubMed]
- Angsantikul, P.; Thamphiwatana, S.; Gao, W.; Zhang, L. Cell membrane-coated nanoparticles as an emerging antibacterial vaccine platform. Vaccines 2015, 3, 814–828. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, T.M.; Demento, S.L.; Caplan, M.J.; Mellman, I.; Saltzman, W.M. Design opportunities for actively targeted nanoparticle vaccines. Nanomedicine 2008, 3, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Irvine, D.J.; Hanson, M.C.; Rakhra, K.; Tokatlian, T. Synthetic nanoparticles for vaccines and immunotherapy. Chem. Rev. 2015, 115, 11109–11146. [Google Scholar] [CrossRef] [PubMed]
- Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Nanomedicine: Current status and future prospects. FASEB J. 2005, 19, 311–330. [Google Scholar] [CrossRef] [PubMed]
- Mateescu, A.L.; Dimov, T.V.; Grumezescu, A.M.; Gestal, M.C.; Chifiriuc, M.C. Nanostructured bioactive polymers used in food-packaging. Curr. Pharm. Biotechnol. 2015, 16, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Seth, A.; Wibowo, N.; Zhao, C.X.; Mitter, N.; Yu, C.; Middelberg, A.P. Nanoparticle vaccines. Vaccine 2014, 32, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Doh, H.J.; Jang, M.H.; Ha, Y.J.; Chung, S.I.; Park, H.J. Oral immunization with helicobacter pylori-loaded poly(d,l-lactide-co-glycolide) nanoparticles. Helicobacter 1999, 4, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Vila, A.; Sanchez, A.; Evora, C.; Soriano, I.; Vila Jato, J.L.; Alonso, M.J. PEG-PLA nanoparticles as carriers for nasal vaccine delivery. J. Aerosol Med. 2004, 17, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Parlane, N.A.; Rehm, B.H.; Wedlock, D.N.; Buddle, B.M. Novel particulate vaccines utilizing polyester nanoparticles (bio-beads) for protection against mycobacterium bovis infection—A review. Vet. Immunol. Immunopathol. 2014, 158, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Luo, Z.; Liu, P.; Gao, N.; Zhang, Y.; Pan, H.; Liu, L.; Wang, C.; Cai, L.; Ma, Y. Bioreducible alginate-poly(ethylenimine) nanogels as an antigen-delivery system robustly enhance vaccine-elicited humoral and cellular immune responses. J. Control. Release 2013, 168, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Saade, F.; Honda-Okubo, Y.; Trec, S.; Petrovsky, N. A novel hepatitis B vaccine containing advax, a polysaccharide adjuvant derived from delta inulin, induces robust humoral and cellular immunity with minimal reactogenicity in preclinical testing. Vaccine 2013, 31, 1999–2007. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Jiang, Q.; Xia, M.; Lu, Y.; Qiu, W.; Zhao, D.; Lu, L.; Peng, G.; Wang, Y. Enhanced immune response and protective effects of nano-chitosan-based DNA vaccine encoding T cell epitopes of esat-6 and FL against mycobacterium tuberculosis infection. PLoS ONE 2013, 8, e61135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tahara, Y.; Mukai, S.A.; Sawada, S.; Sasaki, Y.; Akiyoshi, K. Nanocarrier-integrated microspheres: Nanogel tectonic engineering for advanced drug-delivery systems. Adv. Mater. 2015, 27, 5080–5088. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Rizzo, M.A.; Bhattacharya, S.; Huang, L. Characterization of cationic lipid-protamine-DNA (lPD) complexes for intravenous gene delivery. Gene Ther. 1998, 5, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Morein, B.; Lovgren, K.; Hoglund, S.; Sundquist, B. The iscom: An immunostimulating complex. Immunol. Today 1987, 8, 333–338. [Google Scholar] [CrossRef]
- Zhang, L.F.; Zhou, J.; Chen, S.; Cai, L.L.; Bao, Q.Y.; Zheng, F.Y.; Lu, J.Q.; Padmanabha, J.; Hengst, K.; Malcolm, K.; et al. HPV6b virus like particles are potent immunogens without adjuvant in man. Vaccine 2000, 18, 1051–1058. [Google Scholar] [CrossRef]
- Andre, F.E. Overview of a 5-year clinical experience with a yeast-derived hepatitis b vaccine. Vaccine 1990, 8, S74–S78. [Google Scholar] [CrossRef]
- Lopez-Sagaseta, J.; Malito, E.; Rappuoli, R.; Bottomley, M.J. Self-assembling protein nanoparticles in the design of vaccines. Comput. Struct. Biotechnol. J. 2016, 14, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, N.Z.; Wang, M.; Dong, H.; Feng, S.Y.; Guo, H.C.; Zhu, X.Q. A long-lasting protective immunity against chronic toxoplasmosis in mice induced by recombinant rhoptry proteins encapsulated in poly (lactide-co-glycolide) microparticles. Parasitol. Res. 2015, 114, 4195–4203. [Google Scholar] [CrossRef] [PubMed]
- Mohan, T.; Mitra, D.; Rao, D.N. Nasal delivery of plg microparticle encapsulated defensin peptides adjuvanted GP41 antigen confers strong and long-lasting immunoprotective response against HIV-1. Immunol. Res. 2014, 58, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Colonna, C.; Dorati, R.; Conti, B.; Caliceti, P.; Genta, I. Sub-unit vaccine against S. aureus-mediated infections: Set-up of nano-sized polymeric adjuvant. Int. J. Pharm. 2013, 452, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, C.; O’Connor, G.; O’Leary, S.; Gallagher, P.J.; Cryan, S.A.; Keane, J.; O’Sullivan, M.P. Treatment of mycobacterium tuberculosis-infected macrophages with poly(lactic-co-glycolic acid) microparticles drives nfkappab and autophagy dependent bacillary killing. PLoS ONE 2016, 11, e0149167. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Somani, V.K.; Aggarwal, S.; Bhatnagar, R. PLGA (85:15) nanoparticle based delivery of rL7/L12 ribosomal protein in mice protects against Brucella abortus 544 infection: A promising alternate to traditional adjuvants. Mol. Immunol. 2015, 68, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Oberoi, H.S.; Yorgensen, Y.M.; Morasse, A.; Evans, J.T.; Burkhart, D.J. Peg modified liposomes containing CRX-601 adjuvant in combination with methylglycol chitosan enhance the murine sublingual immune response to influenza vaccination. J. Control. Release 2016, 223, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Balachandran, Y.L.; Li, D.; Shao, Y.; Jiang, X. Polyvinylpyrrolidone-poly(ethylene glycol) modified silver nanorods can be a safe, noncarrier adjuvant for HIV vaccine. ACS Nano 2016, 10, 3589–3596. [Google Scholar] [CrossRef] [PubMed]
- Powles, L.; Xiang, S.D.; Selomulya, C.; Plebanski, M. The use of synthetic carriers in malaria vaccine design. Vaccines 2015, 3, 894–929. [Google Scholar] [CrossRef] [PubMed]
- Parlane, N.A.; Grage, K.; Mifune, J.; Basaraba, R.J.; Wedlock, D.N.; Rehm, B.H.; Buddle, B.M. Vaccines displaying mycobacterial proteins on biopolyester beads stimulate cellular immunity and induce protection against tuberculosis. Clin. Vaccine Immunol. 2012, 19, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Feinen, B.; Petrovsky, N.; Verma, A.; Merkel, T.J. Advax-adjuvanted recombinant protective antigen provides protection against inhalational anthrax that is further enhanced by addition of murabutide adjuvant. Clin. Vaccine Immunol. 2014, 21, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Del Rio, E.; Marradi, M.; Calderon-Gonzalez, R.; Frande-Cabanes, E.; Penades, S.; Petrovsky, N.; Alvarez-Dominguez, C. A gold glyco-nanoparticle carrying a listeriolysin o peptide and formulated with advax delta inulin adjuvant induces robust T-cell protection against listeria infection. Vaccine 2015, 33, 1465–1473. [Google Scholar] [CrossRef] [PubMed]
- Honda-Okubo, Y.; Saade, F.; Petrovsky, N. Advax, a polysaccharide adjuvant derived from delta inulin, provides improved influenza vaccine protection through broad-based enhancement of adaptive immune responses. Vaccine 2012, 30, 5373–5381. [Google Scholar] [CrossRef] [PubMed]
- Honda-Okubo, Y.; Barnard, D.; Ong, C.H.; Peng, B.H.; Tseng, C.T.; Petrovsky, N. Severe acute respiratory syndrome-associated coronavirus vaccines formulated with delta inulin adjuvants provide enhanced protection while ameliorating lung eosinophilic immunopathology. J. Virol. 2015, 89, 2995–3007. [Google Scholar] [CrossRef] [PubMed]
- Petrovsky, N.; Cooper, P.D. Advax, a novel microcrystalline polysaccharide particle engineered from delta inulin, provides robust adjuvant potency together with tolerability and safety. Vaccine 2015, 33, 5920–5926. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.R.; Mehta, M.R.; Bannalikar, A.R.; Menon, M.D. Alginate microparticles loaded with lipopolysaccharide subunit antigen for mucosal vaccination against Klebsiella pneumoniae. Biologicals 2015, 43, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Farjah, A.; Owlia, P.; Siadat, S.D.; Mousavi, S.F.; Ardestani, M.S.; Mohammadpour, H.K. Immunological evaluation of an alginate-based conjugate as a vaccine candidate against Pseudomonas aeruginosa. Acta Pathol. Microbiol. Immunol. Scand. 2015, 123, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Nagatomo, D.; Taniai, M.; Ariyasu, H.; Taniguchi, M.; Aga, M.; Ariyasu, T.; Ohta, T.; Fukuda, S. Cholesteryl pullulan encapsulated TNF-alpha nanoparticles are an effective mucosal vaccine adjuvant against influenza virus. Biomed. Res. Int. 2015, 2015, 471468. [Google Scholar] [CrossRef] [PubMed]
- Cevher, E.; Salomon, S.K.; Somavarapu, S.; Brocchini, S.; Alpar, H.O. Development of chitosan-pullulan composite nanoparticles for nasal delivery of vaccines: In vivo studies. J. Microencapsul. 2015, 32, 769–783. [Google Scholar] [CrossRef] [PubMed]
- Doavi, T.; Mousavi, S.L.; Kamali, M.; Amani, J.; Fasihi Ramandi, M. Chitosan-based intranasal vaccine against Escherichia coli O157:H7. Iran Biomed. J. 2016, 20, 97–108. [Google Scholar] [PubMed]
- Cui, Z.; Han, D.; Sun, X.; Zhang, M.; Feng, X.; Sun, C.; Gu, J.; Tong, C.; Lei, L.; Han, W. Mannose-modified chitosan microspheres enhance oprf-opri-mediated protection of mice against Pseudomonas aeruginosa infection via induction of mucosal immunity. Appl. Microbiol. Biotechnol. 2015, 99, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Khalili, I.; Ghadimipour, R.; Sadigh Eteghad, S.; Fathi Najafi, M.; Ebrahimi, M.M.; Godsian, N.; Sefidi Heris, Y.; Khalili, M.T. Evaluation of immune response against inactivated avian influenza (H9N2) vaccine, by using chitosan nanoparticles. Jundishapur. J. Microbiol. 2015, 8, e27035. [Google Scholar] [CrossRef] [PubMed]
- Lebre, F.; Borchard, G.; Faneca, H.; Pedroso de Lima, M.C.; Borges, O. Intranasal administration of novel chitosan nanoparticle/DNA complexes induces antibody response to hepatitis B surface antigen in mice. Mol. Pharm. 2016, 13, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Malathi, B.; Mona, S.; Thiyagarajan, D.; Kaliraj, P. Immunopotentiating nano-chitosan as potent vaccine carter for efficacious prophylaxis of filarial antigens. Int. J. Biol. Macromol. 2015, 73, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Hunsawong, T.; Sunintaboon, P.; Warit, S.; Thaisomboonsuk, B.; Jarman, R.G.; Yoon, I.K.; Ubol, S.; Fernandez, S. A novel dengue virus serotype-2 nanovaccine induces robust humoral and cell-mediated immunity in mice. Vaccine 2015, 33, 1702–1710. [Google Scholar] [CrossRef] [PubMed]
- Fukuyama, Y.; Yuki, Y.; Katakai, Y.; Harada, N.; Takahashi, H.; Takeda, S.; Mejima, M.; Joo, S.; Kurokawa, S.; Sawada, S.; et al. Nanogel-based pneumococcal surface protein a nasal vaccine induces microRNA-associated TH17 cell responses with neutralizing antibodies against streptococcus pneumoniae in macaques. Mucosal Immunol. 2015, 8, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.H.; Dai, W.J.; Chen, B.; Fan, X.Y. Mucosal immunization with psaa protein, using chitosan as a delivery system, increases protection against acute otitis media and invasive infection by streptococcus pneumoniae. Scand. J. Immunol. 2015, 81, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Tahamtan, A.; Ghaemi, A.; Gorji, A.; Kalhor, H.R.; Sajadian, A.; Tabarraei, A.; Moradi, A.; Atyabi, F.; Kelishadi, M. Antitumor effect of therapeutic hpv DNA vaccines with chitosan-based nanodelivery systems. J. Biomed. Sci. 2014, 21, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dai, C.; Kang, H.; Yang, W.; Sun, J.; Liu, C.; Cheng, G.; Rong, G.; Wang, X.; Wang, X.; Jin, Z.; et al. O-2′-hydroxypropyltrimethyl ammonium chloride chitosan nanoparticles for the delivery of live newcastle disease vaccine. Carbohydr. Polym. 2015, 130, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Hunsawong, T.; Sunintaboon, P.; Warit, S.; Thaisomboonsuk, B.; Jarman, R.G.; Yoon, I.K.; Ubol, S.; Fernandez, S. Immunogenic properties of a bcg adjuvanted chitosan nanoparticle-based dengue vaccine in human dendritic cells. PLoS Negl. Trop. Dis. 2015, 9, e0003958. [Google Scholar] [CrossRef] [PubMed]
- Boraschi, D.; Italiani, P. From antigen delivery system to adjuvanticy: The board application of nanoparticles in vaccinology. Vaccines 2015, 3, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C.; Liu, J.; Yang, J.; Li, Y.; Weng, J.; Shao, Y.; Zhang, X. Enhanced non-inflammasome mediated immune responses by mannosylated zwitterionic-based cationic liposomes for HIV DNA vaccines. Biomaterials 2016, 85, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Monaci, E.; Mancini, F.; Lofano, G.; Bacconi, M.; Tavarini, S.; Sammicheli, C.; Arcidiacono, L.; Giraldi, M.; Galletti, B.; Rossi Paccani, S.; et al. Mf59- and al(OH)3-adjuvanted staphylococcus aureus (4C-Staph) vaccines induce sustained protective humoral and cellular immune responses, with a critical role for effector CD4 T cells at low antibody titers. Front. Immunol. 2015, 6, 439. [Google Scholar] [CrossRef] [PubMed]
- Klimka, A.; Michels, L.; Glowalla, E.; Tosetti, B.; Kronke, M.; Krut, O. Montanide isa 71 VG is advantageous to freund’s adjuvant in immunization against S. aureus infection of mice. Scand. J. Immunol. 2015, 81, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.L.; Xiang, S.D.; Plebanski, M. A model to study the impact of polymorphism driven liver-stage immune evasion by malaria parasites, to help design effective cross-reactive vaccines. Front. Microbiol. 2016, 7, 303. [Google Scholar] [CrossRef] [PubMed]
- Cargnelutti, D.E.; Salomon, M.C.; Celedon, V.; Garcia Bustos, M.F.; Morea, G.; Cuello-Carrion, F.D.; Scodeller, E.A. Immunization with antigenic extracts of Leishmania associated with Montanide ISA 763 adjuvant induces partial protection in BALB/c mice against Leishmania (Leishmania) amazonensis infection. J. Microbiol. Immunol. Infect. 2016, 49, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Safari, D.; Marradi, M.; Chiodo, F.; Th Dekker, H.A.; Shan, Y.; Adamo, R.; Oscarson, S.; Rijkers, G.T.; Lahmann, M.; Kamerling, J.P.; et al. Gold nanoparticles as carriers for a synthetic streptococcus pneumoniae type 14 conjugate vaccine. Nanomedicine 2012, 7, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.E.; Judy, B.M.; Qazi, O.; Blumentritt, C.A.; Brown, K.A.; Shaw, A.M.; Torres, A.G.; Titball, R.W. A gold nanoparticle-linked glycoconjugate vaccine against burkholderia mallei. Nanomedicine 2015, 11, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.E.; Williamson, E.D.; Prior, J.L.; Butcher, W.A.; Thompson, I.J.; Shaw, A.M.; Titball, R.W. Conjugation of y. Pestis F1-antigen to gold nanoparticles improves immunogenicity. Vaccine 2012, 30, 6777–6782. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Ray, P.C.; Datta, D.; Bansal, G.P.; Angov, E.; Kumar, N. Nanovaccines for malaria using Plasmodium falciparum antigen Pfs25 attached gold nanoparticles. Vaccine 2015, 33, 5064–5071. [Google Scholar] [CrossRef] [PubMed]
- Gianvincenzo, P.D.; Calvo, J.; Perez, S.; Alvarez, A.; Bedoya, L.M.; Alcami, J.; Penades, S. Negatively charged glyconanoparticles modulate and stabilize the secondary structures of a gp120 V3 loop peptide: Toward fully synthetic HIV vaccine candidates. Bioconj. Chem. 2015, 26, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Yandar, N.; Pastorin, G.; Prato, M.; Bianco, A.; Patarroyo, M.E.; Lozano, J.M. Immunological profile of a Plasmodium vivax AMA-1 N-terminus peptide-carbon nanotube conjugate in an infected plasmodium berghei mouse model. Vaccine 2008, 26, 5864–5873. [Google Scholar] [CrossRef] [PubMed]
- Andrade, G.R.; New, R.R.; Sant’Anna, O.A.; Williams, N.A.; Alves, R.C.; Pimenta, D.C.; Vigerelli, H.; Melo, B.S.; Rocha, L.B.; Piazza, R.M.; et al. A universal polysaccharide conjugated vaccine against O111 E. coli. Hum. Vaccines Immunother. 2014, 10, 2864–2874. [Google Scholar] [CrossRef] [PubMed]
- Neuhaus, V.; Chichester, J.A.; Ebensen, T.; Schwarz, K.; Hartman, C.E.; Shoji, Y.; Guzman, C.A.; Yusibov, V.; Sewald, K.; Braun, A. A new adjuvanted nanoparticle-based H1N1 influenza vaccine induced antigen-specific local mucosal and systemic immune responses after administration into the lung. Vaccine 2014, 32, 3216–3222. [Google Scholar] [CrossRef] [PubMed]
- Skrastina, D.; Petrovskis, I.; Lieknina, I.; Bogans, J.; Renhofa, R.; Ose, V.; Dishlers, A.; Dekhtyar, Y.; Pumpens, P. Silica nanoparticles as the adjuvant for the immunisation of mice using hepatitis B core virus-like particles. PLoS ONE 2014, 9, e114006. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.I.; Omar, A.R.; Hussein, M.Z.; Elkhidir, I.M.; Sekawi, Z. Systemic antibody response to nano-size calcium phospate biocompatible adjuvant adsorbed HEV-71 killed vaccine. Clin. Exp. Vaccine Res. 2015, 4, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, K.; Gopinath, V.P.; Raj, G.D. Formulation of newcastle disease virus coupled calcium phosphate nanoparticles: An effective strategy for oculonasal delivery to chicken. Colloids Surf. B Biointerfaces 2014, 116, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Knuschke, T.; Bayer, W.; Rotan, O.; Sokolova, V.; Wadwa, M.; Kirschning, C.J.; Hansen, W.; Dittmer, U.; Epple, M.; Buer, J.; et al. Prophylactic and therapeutic vaccination with a nanoparticle-based peptide vaccine induces efficient protective immunity during acute and chronic retroviral infection. Nanomedicine 2014, 10, 1787–1798. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.S.; Pereyra, E.A.; Baravalle, C.; Camussone, C.M.; Dallard, B.E.; Marcipar, I.S.; Calvinho, L.F. Functional role of antibodies generated in heifers through immunization with staphylococcus aureus vaccines in invasion and phagocytosis assays. FEMS Microbiol. Lett. 2014, 360, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Bontempi, I.A.; Vicco, M.H.; Cabrera, G.; Villar, S.R.; Gonzalez, F.B.; Roggero, E.A.; Ameloot, P.; Callewaert, N.; Perez, A.R.; Marcipar, I.S. Efficacy of a trans-sialidase-iscomatrix subunit vaccine candidate to protect against experimental chagas disease. Vaccine 2015, 33, 1274–1283. [Google Scholar] [CrossRef] [PubMed]
- Heldens, J.G.; Pouwels, H.G.; Derks, C.G.; Van de Zande, S.M.; Hoeijmakers, M.J. Duration of immunity induced by an equine influenza and tetanus combination vaccine formulation adjuvanted with iscom-matrix. Vaccine 2010, 28, 6989–6996. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.L.; Twyman, R.M.; Fiering, S.; Steinmetz, N.F. Virus-based nanoparticles as platform technologies for modern vaccines. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 554–578. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Liu, J.; Chen, C.; Ye, X.; Xu, L.; Wang, W.; Zhao, Q.; Zhu, H.; Cheng, T.; Xia, N. A highly conserved epitope-vaccine candidate against varicella-zoster virus induces neutralizing antibodies in mice. Vaccine 2016, 34, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, F.; Rostami, S.; Meshkat, Z. Progress in the development of vaccines for hepatitis c virus infection. World J. Gastroenterol. 2015, 21, 11984–12002. [Google Scholar] [CrossRef] [PubMed]
- Yee, P.T.; Poh, C.L. Development of novel vaccines against enterovirus-71. Viruses 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Schwameis, M.; Buchtele, N.; Wadowski, P.P.; Schoergenhofer, C.; Jilma, B. Chikungunya vaccines in development. Hum. Vaccines Immunother. 2016, 12, 716–731. [Google Scholar] [CrossRef] [PubMed]
- Tamborrini, M.; Geib, N.; Marrero-Nodarse, A.; Jud, M.; Hauser, J.; Aho, C.; Lamelas, A.; Zuniga, A.; Pluschke, G.; Ghasparian, A.; et al. A synthetic virus-like particle streptococcal vaccine candidate using B-cell epitopes from the proline-rich region of pneumococcal surface protein A. Vaccines 2015, 3, 850–874. [Google Scholar] [CrossRef] [PubMed]
- McKay, P.F.; Cope, A.V.; Mann, J.F.; Joseph, S.; Esteban, M.; Tatoud, R.; Carter, D.; Reed, S.G.; Weber, J.; Shattock, R.J. Glucopyranosyl lipid a adjuvant significantly enhances HIV specific t and b cell responses elicited by a DNA-mva-protein vaccine regimen. PLoS ONE 2014, 9, e84707. [Google Scholar] [CrossRef] [PubMed]
- Burkhard, P.; Lanar, D.E. Malaria vaccine based on self-assembling protein nanoparticles. Expert Rev. Vaccines 2015, 14, 1525–1527. [Google Scholar] [CrossRef] [PubMed]
- El Bissati, K.; Zhou, Y.; Dasgupta, D.; Cobb, D.; Dubey, J.P.; Burkhard, P.; Lanar, D.E.; McLeod, R. Effectiveness of a novel immunogenic nanoparticle platform for toxoplasma peptide vaccine in HLA transgenic mice. Vaccine 2014, 32, 3243–3248. [Google Scholar] [CrossRef] [PubMed]
- Babapoor, S.; Neef, T.; Mittelholzer, C.; Girshick, T.; Garmendia, A.; Shang, H.; Khan, M.I.; Burkhard, P. A novel vaccine using nanoparticle platform to present immunogenic M2e against avian influenza infection. Influenza Res. Treat. 2011, 2011, 126794. [Google Scholar] [CrossRef] [PubMed]
- Wahome, N.; Pfeiffer, T.; Ambiel, I.; Yang, Y.; Keppler, O.T.; Bosch, V.; Burkhard, P. Conformation-specific display of 4E10 and 2F5 epitopes on self-assembling protein nanoparticles as a potential HIV vaccine. Chem. Biol. Drug Des. 2012, 80, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Qasim, M.; Lim, D.J.; Park, H.; Na, D. Nanotechnology for diagnosis and treatment of infectious diseases. J. Nanosci. Nanotechnol. 2014, 14, 7374–7387. [Google Scholar] [CrossRef] [PubMed]
- Pedrosa, P.; Baptista, P.V. Gold and silver nanoparticles for diagnostics of infection. In Nanotechnology in Diagnosis, Treatment and Prophylaxis of Infectious Diseases, 1st ed; Elsevier: London, UK, 2015; Chapter: 1. [Google Scholar] [CrossRef]
- Hauck, T.S.; Giri, S.; Gao, Y.; Chan, W.C. Nanotechnology diagnostics for infectious diseases prevalent in developing countries. Adv. Drug Deliv. Rev. 2010, 62, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Blecher, K.; Nasir, A.; Friedman, A. The growing role of nanotechnology in combating infectious disease. Virulence 2011, 2, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Qin, D.; He, X.; Wang, K.; Zhao, X.J.; Tan, W.; Chen, J. Fluorescent nanoparticle-based indirect immunofluorescence microscopy for detection of mycobacterium tuberculosis. J. Biomed. Biotechnol. 2007, 2007, 89364. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, J.; Li, Y.; He, D.; He, X.; Wang, K.; Zou, Z.; Shi, H. A combination of positive dielectrophoresis driven on-line enrichment and aptamer-fluorescent silica nanoparticle label for rapid and sensitive detection of staphylococcus aureus. Analyst 2015, 140, 4489–4497. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.Y.; Kang, Y.J. Bioprobes based on aptamer and silica fluorescent nanoparticles for bacteria salmonella typhimurium detection. Nanoscale Res. Lett. 2016, 11, 150. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Harrison, S.H.; Schoeniger, J.S. Gangliosides as receptors for biological toxins: Development of sensitive fluoroimmunoassays using ganglioside-bearing liposomes. Anal. Chem. 2000, 72, 6019–6024. [Google Scholar] [CrossRef] [PubMed]
- Ahn-Yoon, S.; DeCory, T.R.; Baeumner, A.J.; Durst, R.A. Ganglioside-liposome immunoassay for the ultrasensitive detection of cholera toxin. Anal.Chem. 2003, 75, 2256–2261. [Google Scholar] [CrossRef] [PubMed]
- Ahn-Yoon, S.; DeCory, T.R.; Durst, R.A. Ganglioside-liposome immunoassay for the detection of botulinum toxin. Anal. Bioanal. Chem. 2004, 378, 68–75. [Google Scholar] [CrossRef] [PubMed]
- De Mello Donega, C. Synthesis and properties of colloidal heteronanocrystals. Chem. Soc. Rev. 2011, 40, 1512–1546. [Google Scholar] [CrossRef] [PubMed]
- Conde, J.; Dias, J.T.; Grazu, V.; Moros, M.; Baptista, P.V.; de la Fuente, J.M. Revisiting 30 years of biofunctionalization and surface chemistry of inorganic nanoparticles for nanomedicine. Front. Chem. 2014, 2, 48. [Google Scholar] [CrossRef] [PubMed]
- Medintz, I.L.; Mattoussi, H.; Clapp, A.R. Potential clinical applications of quantum dots. Int. J. Nanomed. 2008, 3, 151–167. [Google Scholar]
- Zhang, C.Y.; Hu, J. Single quantum dot-based nanosensor for multiple DNA detection. Anal. Chem. 2010, 82, 1921–1927. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Pang, D.W.; Chen, Z.; Shao, J.B.; Xiong, L.H.; Xiang, Y.P.; Xiong, Y.; Wu, K.; Ai, H.W.; Zhang, H. Visual and efficient immunosensor technique for advancing biomedical applications of quantum dots on salmonella detection and isolation. Nanoscale 2016, 8, 4688–4698. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, K.K.; Al-Sheikh, Y.A. Role and implications of nanodiagnostics in the changing trends of clinical diagnosis. Saudi J. Biol. Sci. 2014, 21, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Goldman, E.R.; Balighian, E.D.; Mattoussi, H.; Kuno, M.K.; Mauro, J.M.; Tran, P.T.; Anderson, G.P. Avidin: A natural bridge for quantum dot-antibody conjugates. J. Am. Chem. Soc. 2002, 124, 6378–6382. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Tripp, R.A.; Anderson, L.J.; Nie, S. Real-time detection of virus particles and viral protein expression with two-color nanoparticle probes. J. Virol. 2005, 79, 8625–8628. [Google Scholar] [CrossRef] [PubMed]
- Klostranec, J.M.; Xiang, Q.; Farcas, G.A.; Lee, J.A.; Rhee, A.; Lafferty, E.I.; Perrault, S.D.; Kain, K.C.; Chan, W.C. Convergence of quantum dot barcodes with microfluidics and signal processing for multiplexed high-throughput infectious disease diagnostics. Nano Lett. 2007, 7, 2812–2818. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, S.; Reda, M.; Hussien, A.; Zayed, D. Cdte quantum dots as a novel biosensor for serratia marcescens and lipopolysaccharide. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 150, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Goluch, E.D.; Nam, J.M.; Georganopoulou, D.G.; Chiesl, T.N.; Shaikh, K.A.; Ryu, K.S.; Barron, A.E.; Mirkin, C.A.; Liu, C. A bio-barcode assay for on-chip attomolar-sensitivity protein detection. Lab Chip 2006, 6, 1293–1299. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Wang, Y.; Li, S.S.; Wan, Z.; Zhai, J. Rapid and simultaneous detection of human hepatitis B virus and hepatitis C virus antibodies based on a protein chip assay using nano-gold immunological amplification and silver staining method. BMC Infect. Dis. 2005, 5, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bohara, R.A.; Pawar, S.H. Innovative developments in bacterial detection with magnetic nanoparticles. Appl. Biochem. Biotechnol. 2015, 176, 1044–1058. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.W.; Doshi, N.; Mitragotri, S. Adaptive micro and nanoparticles: Temporal control over carrier properties to facilitate drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 1247–1256. [Google Scholar] [CrossRef] [PubMed]
- Tsourkas, A.; Hofstetter, O.; Hofstetter, H.; Weissleder, R.; Josephson, L. Magnetic relaxation switch immunosensors detect enantiomeric impurities. Angew. Chem. 2004, 43, 2395–2399. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.C.; Tsai, P.J.; Lin, Y.S.; Chen, Y.C. Using biofunctionalized nanoparticles to probe pathogenic bacteria. Anal. Chem. 2004, 76, 7162–7168. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.S.; Tsai, P.J.; Weng, M.F.; Chen, Y.C. Affinity capture using vancomycin-bound magnetic nanoparticles for the maldi-ms analysis of bacteria. Anal. Chem. 2005, 77, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Gopal, J.; Muthu, M.; Chun, S.C.; Wu, H.F. State-of-the-art nanoplatform-integrated MALDI-MS impacting resolutions in urinary proteomics. Proteom. Clin. Appl. 2015, 9, 469–481. [Google Scholar] [CrossRef] [PubMed]
- Azzazy, H.M.; Mansour, M.M.; Kazmierczak, S.C. Nanodiagnostics: A new frontier for clinical laboratory medicine. Clin. Chem. 2006, 52, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Mungroo, N.; Daikuara, L.; Neethirajan, S. Label-free nir-sers discrimination and detection of foodborne bacteria by in situ synthesis of Ag colloids. J. Nanobiotechnol. 2015, 13, 45. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Qin, T.; Jia, X.X.; Deng, A.H.; Zhang, X.; Fan, W.H.; Huo, S.D.; Wen, T.Y.; Liu, W.J. Rapid identification of legionella pathogenicity by surface-enhanced raman spectroscopy. Biomed. Environ. Sci. 2015, 28, 437–444. [Google Scholar] [PubMed]
- Sivanesan, A.; Witkowska, E.; Adamkiewicz, W.; Dziewit, L.; Kaminska, A.; Waluk, J. Nanostructured silver-gold bimetallic sers substrates for selective identification of bacteria in human blood. Analyst 2014, 139, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Zhang, X.; Kumar, A.; Zou, G.; Zhang, X.; Liang, X.J. Long genomic DNA amplicons adsorption onto unmodified gold nanoparticles for colorimetric detection of bacillus anthracis. Chem. Commun. 2013, 49, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Kalidasan, K.; Neo, J.L.; Uttamchandani, M. Direct visual detection of salmonella genomic DNA using gold nanoparticles. Mol. Biosyst. 2013, 9, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.A.; Azzazy, H.M.; Attia, A.S.; Hashem, A.G. A sensitive colorimetric assay for identification of acinetobacter baumannii using unmodified gold nanoparticles. J. Appl. Microbiol. 2014, 117, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Veigas, B.; Pedrosa, P.; Carlos, F.F.; Mancio-Silva, L.; Grosso, A.R.; Fortunato, E.; Mota, M.M.; Baptista, P.V. One nanoprobe, two pathogens: Gold nanoprobes multiplexing for point-of-care. J. Nanobiotechnol. 2015, 13, 48. [Google Scholar] [CrossRef] [PubMed]
- Pedrosa, P.; Veigas, B.; Machado, D.; Couto, I.; Viveiros, M.; Baptista, P.V. Gold nanoprobes for multi loci assessment of multi-drug resistant tuberculosis. Tuberculosis 2014, 94, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kong, H.; Mout, R.; Saha, K.; Moyano, D.F.; Robinson, S.M.; Rana, S.; Zhang, X.; Riley, M.A.; Rotello, V.M. Rapid identification of bacterial biofilms and biofilm wound models using a multichannel nanosensor. ACS Nano 2014, 8, 12014–12019. [Google Scholar] [CrossRef] [PubMed]
- Pohlmann, C.; Dieser, I.; Sprinzl, M. A lateral flow assay for identification of Escherichia coli by ribosomal RNA hybridisation. Analyst 2014, 139, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Luo, P.; Liu, Y.; Xia, Y.; Xu, H.; Xie, G. Aptamer biosensor for sensitive detection of toxin a of clostridium difficile using gold nanoparticles synthesized by bacillus stearothermophilus. Biosens. Bioelectron. 2014, 54, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Jiang, Y.; Jia, F.; Yu, Y.; Chen, J.; Wang, Z. An aptamer-based electrochemical biosensor for the detection of salmonella. J. Microbiol. Methods 2014, 98, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Yang, C.Y.; Sun, R.L.; Cheng, Y.F.; Kao, W.C.; Yang, P.C. Rapid single cell detection of staphylococcus aureus by aptamer-conjugated gold nanoparticles. Sci. Rep. 2013, 3, 1863. [Google Scholar] [CrossRef] [PubMed]
- Veigas, B.; Jacob, J.M.; Costa, M.N.; Santos, D.S.; Viveiros, M.; Inacio, J.; Martins, R.; Barquinha, P.; Fortunato, E.; Baptista, P.V. Gold on paper-paper platform for Au-nanoprobe TB detection. Lab Chip 2012, 12, 4802–4808. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Tang, S.; Storhoff, J.; Marla, S.; Bao, Y.P.; Wang, X.; Wong, E.Y.; Ragupathy, V.; Ye, Z.; Hewlett, I.K. Multiplexed, rapid detection of H5N1 using a pcr-free nanoparticle-based genomic microarray assay. BMC Biotechnol. 2010, 10, 74. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Z.; Kim, S.; Cho, W.; Lee, S.Y. Optical detection of nanoparticle-enhanced human papillomavirus genotyping microarrays. Biomed. Opt. Express 2013, 4, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Chudobova, D.; Cihalova, K.; Skalickova, S.; Zitka, J.; Rodrigo, M.A.; Milosavljevic, V.; Hynek, D.; Kopel, P.; Vesely, R.; Adam, V.; et al. 3D-printed chip for detection of methicillin-resistant staphylococcus aureus labeled with gold nanoparticles. Electrophoresis 2015, 36, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Low, K.F.; Rijiravanich, P.; Singh, K.K.; Surareungchai, W.; Yean, C.Y. An electrochemical genosensing assay based on magnetic beads and gold nanoparticle-loaded latex microspheres for vibrio cholerae detection. J. Biomed. Nanotechnol. 2015, 11, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.R.; Kim, J.; Suzuki, T.; Lee, J.; Park, E.Y. Detection of influenza virus using peroxidase-mimic of gold nanoparticles. Biotechnol. Bioeng. 2016. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Yu, Z.; Liu, D.; Peng, T.; Liu, K.; Wang, S.; Xiong, Y.; Wei, H.; Xu, H.; Lai, W. Dual gold nanoparticle lateflow immunoassay for sensitive detection of Escherichia coli O157:H7. Anal. Chim. Acta 2015, 876, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.M.; Huang, R.; Dong, C.X.; Tang, L.J.; Gui, R.; Jiang, J.H. Plasmonic elisa for the ultrasensitive detection of treponema pallidum. Biosens. Bioelectron. 2014, 58, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Zhan, L.; Zhen, S.J.; Wan, X.Y.; Gao, P.F.; Huang, C.Z. A sensitive surface-enhanced raman scattering enzyme-catalyzed immunoassay of respiratory syncytial virus. Talanta 2016, 148, 308–312. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Duan, N.; Li, J.; Ye, J.; Ma, S.; Le, G. Ultrasensitive chemiluminescent immunoassay of salmonella with silver enhancement of nanogold labels. Luminescence 2011, 26, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Hewlett, I. Nanoparticle-based immunoassays for sensitive and early detection of HIV-1 capsid (p24) antigen. J. Infect. Dis. 2010, 201 (Suppl. S1), S59–S64. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Xie, Z.; Xie, Z.; Luo, S.; Xie, L.; Huang, L.; Fan, Q.; Zhang, Y.; Wang, S.; Zeng, T. Silver nanoparticles coated graphene electrochemical sensor for the ultrasensitive analysis of avian influenza virus H7. Anal. Chim. Acta 2016, 913, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Miu, T.; Xu, H.; Duan, N.; Ding, X.; Li, S. Sensitive immunoassay of listeria monocytogenes with highly fluorescent bioconjugated silica nanoparticles probe. J. Microbiol. Methods 2010, 83, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wu, X.; Gan, M.; Xu, F.; He, L.; Yang, D.; Xu, H.; Shah, N.P.; Wei, H. Rapid detection of staphylococcus aureus in dairy and meat foods by combination of capture with silica-coated magnetic nanoparticles and thermophilic helicase-dependent isothermal amplification. J. Dairy Sci. 2015, 98, 1563–1570. [Google Scholar] [CrossRef] [PubMed]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef] [PubMed]
- Beyth, N.; Houri-Haddad, Y.; Domb, A.; Khan, W.; Hazan, R. Alternative antimicrobial approach: Nano-antimicrobial materials. Evid. Based Complement. Altern. Med. 2015, 2015, 246012. [Google Scholar] [CrossRef] [PubMed]
- Yah, C.S.; Simate, G.S. Nanoparticles as potential new generation broad spectrum antimicrobial agents. Daru 2015, 23, 43. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.M.; Hess, K.L.; Gearhart, J.M.; Geiss, K.T.; Schlager, J.J. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol. In Vitro 2005, 19, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Serpooshan, V. Silver-coated engineered magnetic nanoparticles are promising for the success in the fight against antibacterial resistance threat. ACS Nano 2012, 6, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Landis, R.F.; Rotello, V.M. Nanoparticle-based antimicrobials: Surface functionality is critical. F1000Research 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Pelgrift, R.Y.; Friedman, A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013, 65, 1803–1815. [Google Scholar] [CrossRef] [PubMed]
- Hameed, A.S.; Karthikeyan, C.; Ahamed, A.P.; Thajuddin, N.; Alharbi, N.S.; Alharbi, S.A.; Ravi, G. In vitro antibacterial activity of ZnO and Nd doped ZnO nanoparticles against ESBL producing Escherichia coli and Klebsiella pneumoniae. Sci. Rep. 2016, 6, 24312. [Google Scholar] [CrossRef] [PubMed]
- Dastjerdi, R.; Montazer, M. A review on the application of inorganic nano-structured materials in the modification of textiles: Focus on anti-microbial properties. Colloids Surf. B Biointerfaces 2010, 79, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Grumezescu, A.M.; Gestal, M.C.; Holban, A.M.; Grumezescu, V.; Vasile, B.S.; Mogoanta, L.; Iordache, F.; Bleotu, C.; Mogosanu, G.D. Biocompatible Fe3O4 increases the efficacy of amoxicillin delivery against gram-positive and gram-negative bacteria. Molecules 2014, 19, 5013–5027. [Google Scholar] [CrossRef] [PubMed]
- Gestal, M.C.; Holban, A.M. Advances in nanotechnology as an alternative against superbugs. JSM Chem. 2014, 2, 1–5. [Google Scholar]
- Saidykhan, L.; Abu Bakar, M.Z.; Rukayadi, Y.; Kura, A.U.; Latifah, S.Y. Development of nanoantibiotic delivery system using cockle shell-derived aragonite nanoparticles for treatment of osteomyelitis. Int. J. Nanomed. 2016, 11, 661–673. [Google Scholar] [CrossRef] [PubMed]
- Dhanalakshmi, V.; Nimal, T.R.; Sabitha, M.; Biswas, R.; Jayakumar, R. Skin and muscle permeating antibacterial nanoparticles for treating staphylococcus aureus infected wounds. J. Biomed. Mater. Res. B Appl. Biomater. 2016, 104, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Bolocan, A.; Mihaiescu, D.E.; Andronescu, E.; Voicu, G.; Grumezescu, A.M.; Ficai, A.; Vasile, B.S.; Bleotu, C.; Chifiriuc, M.C.; Pop, C.S. Biocompatible hydrodispersible magnetite nanoparticles used as antibiotic drug carriers. Rom. J. Morphol. Embryol. 2015, 56, 365–370. [Google Scholar] [PubMed]
- Min, J.; Choi, K.Y.; Dreaden, E.C.; Padera, R.F.; Braatz, R.D.; Spector, M.; Hammond, P.T. Designer dual therapy nanolayered implant coatings eradicate biofilms and accelerate bone tissue repair. ACS Nano 2016, 10, 4441–4450. [Google Scholar] [CrossRef] [PubMed]
- Milo, S.; Thet, N.T.; Liu, D.; Nzakizwanayo, J.; Jones, B.V.; Jenkins, A.T. An in-situ infection detection sensor coating for urinary catheters. Biosens. Bioelectron. 2016, 81, 166–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diab, R.; Khameneh, B.; Joubert, O.; Duval, R. Insights in nanoparticle-bacterium interactions: New frontiers to bypass bacterial resistance to antibiotics. Curr. Pharm. Des. 2015, 21, 4095–4105. [Google Scholar] [CrossRef] [PubMed]


| Type of Nanoparticles | Based on | Main Characteristic | Use | Representative Uses | Ref. | |
|---|---|---|---|---|---|---|
| Polymers | Synthetic | PLG | Biocompatibility & biodegradability | entrap antigen for delivery (carrier) to certain cells and sustain Ag release according to their biodegradation rate | Toxoplasmosis | [33] |
| HIV | [34] | |||||
| PLGA | S. aureus | [35] | ||||
| TB | [36] | |||||
| Brucella abortus | [37] | |||||
| Antrhrax | [20] | |||||
| Plasmodium vivax | ||||||
| HBV | ||||||
| PGE | Influenza Virus | [38] | ||||
| HIV | [39] | |||||
| Polystyrene | Can be conjugated with a variety of Ag or surface-modified with various functional groups | P. malariae | [40] | |||
| Polyester Bio-Beds | Vaccine delivery system low cost & biocompatibility | TB | [41] | |||
| Natural | Inulin: ADVAX™ | Activator of complement alternative pathway, potent adjuvant. | adjuvant | Antrax | [42] | |
| Listeria monocytogenes | [43] | |||||
| Influenza virus | [44] | |||||
| SARS-CoV | [45] | |||||
| HBV | [25] | |||||
| HIV, JVE-WNV | [46] | |||||
| Alginate | Biocompatibility, biodegradability & nontoxic | K. pneumoniae | [47] | |||
| P. aeruginosa | [48] | |||||
| Pullulan | Influenza virus | [49] | ||||
| Difteria | [50] | |||||
| Chitosan | ||||||
| Easily modified | E. coli O157:H7 | [51] | ||||
| P. aeruginosa | [52] | |||||
| Influenza virus | [38,53] | |||||
| HBV | [54] | |||||
| Filariasis | [55] | |||||
| Dengue | [56] | |||||
| Hydrogel | Pullulan | Flexible mesh size Large surface area for multivalent conjugation: high capacity for Ag | hydrophilic 3D polymer network. | S. pneumoniae | [57] | |
| Chitostan | C. botulinum | [20] | ||||
| S. pneumonie | [58] | |||||
| Papilomavirus | [59] | |||||
| NDV | [60] | |||||
| Dengue | [61] | |||||
| Liposomes | LPD | phospholipids | Biodegradable & nontoxic encapsulate Ag and form virosomes | adjuvant | P. malarie | [40] |
| Influenza Virus (INFLEXAL® V) | [62] | |||||
| HAV (Epaxal®) | ||||||
| HIV | [63] | |||||
| ICMV | adjuvant carrier | P. vivax | [40] | |||
| Emulsions | Oil-in-water/water-in-oil | MF59™ | Mixed with Ag & transport | adjuvant | S. aureus | [64] |
| Influenza Virus (FLUAD®, AFLUNOV®, FOCETRIA®, OPTAFLU®) | [62] | |||||
| HAV (Epaxal®) | ||||||
| AS03/AS04 | Inluenza Virus (Pandemrix®, Prepandrix®) | [62] | ||||
| HBV (Fendrix®) | ||||||
| Papilomavirus (Cervarix®) | ||||||
| Montanide™ | S. aureus | [65] | ||||
| Plasmodium spp. | [66] | |||||
| L. amazonensis | [67] | |||||
| Inorganic | AuNP | Au/gold | APC cytokine production can be induced according to shape and size | Adjuvant recognition, absorption of specific biomolecules, improvement of interaction with cells & enhancement of cellular uptake | Pneumococo | [68] |
| L. monocytogenes | [43] | |||||
| Burkholderia mallei | [69] | |||||
| Yersinia pestis | [70] | |||||
| P. falciparum | [71] | |||||
| GNP | HIV | [72] | ||||
| CNT | Carbon | Good biocompatibility Synthesized into a variety of nanotubes and mesoporous spheres multiple copies of protein and peptide Ag | P. vivax | [73] | ||
| SiNP | Si | Biocompatible | E. coli 0111 | [74] | ||
| Influenza Virus | [75] | |||||
| HBV | [76] | |||||
| calcium | Ca | Excellent biocompatibility & non-toxic for DNA vaccines and mucosal immunity | Enterovirus 71 | [77] | ||
| NDV | [78] | |||||
| HIV | [79] | |||||
| ISCOM | Quil A, cholesterol, phospholipids & protein Ag | Trap the Ag by apolar interactions mucosal immunity | Adjuvant | S. aureus | [80] | |
| P. malarie | [40] | |||||
| Chagas disease | [81] | |||||
| Tetanus | [82] | |||||
| Influenza Virus | [20] | |||||
| HSV | ||||||
| HBV | ||||||
| HIV | ||||||
| VLP | Self-assembly biocompatible capsid protein | Evolved viral structure & delivery platform | Induce potent immune responses | Papilomavirus (Cervarix®, GARDA SIL®) | [62] | |
| HBV (Engerix®, RECOMBIVAX®HB) | ||||||
| HIV, Influenza Virus, Marburg, Ébola | [83] | |||||
| E. coli, | [32] | |||||
| P. falciparum | ||||||
| Norovirus, HEV (Hecolin) | [84] | |||||
| VZV | [85] | |||||
| HVC | [86] | |||||
| Enterovirus | [87] | |||||
| Chikunguya Virus | [88] | |||||
| S. pneumoniae | ||||||
| Self-assembling systems | ferritin | Fe | Attempt to drive higher levels of protein quaternary structuring | Adjuvant | Influenza Virus, VEB, HCV, HIV | [32] |
| MVP | Protein | HIV | [89] | |||
| SAPNs | Peptides | Ability to repetitively present Ag Better biophysical & immunological properties | Strong immunogenic effect (of cellular vaccines) Purity & high specificity immune responses. | P. malarie | [90] | |
| Toxoplasmosis | [91] | |||||
| Influenza Virus | [92] | |||||
| HIV | [93] | |||||
| HCV | [85] | |||||
| Nanoparticle | Based on | Detection/Identification by | Detection | Limit of Detection | Ref. |
|---|---|---|---|---|---|
| Surface-Enhanced Raman scattering Spectroscopy (SERS) | |||||
| AgNPs | Label-free near infrared surface-enhanced Raman scattering (NIR-SERS) | Spectrum | MRSA, Listeria spp., E. coli & P. aeruginosa | 103 CFU/mL | [123] |
| Au-cotted-NPs | Surface-enhanced Raman scattering spectroscopy (SERS) | Legionella spp. | [124] | ||
| Vancomycin coatted Ag-Au-NPs | S. epidermidis, B. megaterium, E. coli & Salmonella enterica | 102 CFU/mL | [125] | ||
| Surface Plasmon resonance (SPR). NanoProbes | |||||
| AuNPs | Differential stabilization of Au-nanoprobes in presence of DNA targets following salt induced aggregation | Colorimeric detection from red to blue | TB | 0.75 μg 2 h | [11] |
| Different interaction between ssdna and dsdna at the surface of aunps, based on the aggregation of unmodified aunps | Bacillus. anthracis | [126] | |||
| Cross-linking approach, where the target DNA acts as a linker between two different Au nanoprobes; based on aunp aggregation | Colorimetric detection from red to blue | MRSA | 66 pg/μL (<105 CFU/mL) | [11] | |
| Interaction aunps–dsdna & the addition of thiolated probes specific to the inva gene in the Salmonella genomic DNA aggregates aunps | Colorimetric detection from red to violet | S. enterica | 37 fM | [127] | |
| The ability of ssdna oligo-targeters to stabilize the colloidal aunps preventing their salt-induced aggregation. | Colorimetric detection | Acinetobacter baumani | 0.8125 ng/μL | [128] | |
| Non-cross-linking method results from the differential aggregation profiles of Au-nanoprobes induced by increased ionic strength in the presence or absence of the specific target sequence | Colorimetric detection (SPR band: 525–650 nm) | MTBC and Plasmodium | [129] | ||
| MDRTB | [130] | ||||
| Multichannel fluorescence sensor | Ratiometric response according to three-color RGB output | BIOFILMS: Amycolatopsis azurea, B. licheniformic, B. megaterium, E. coli, P. aeruginosa | [131] | ||
| Fast lateral flow immunoassay (FLFI) approach combined with rapid “one step” lysis | Colorimetric detection | E. coli | 5 × 104 CFU/mL 25 min | [132] | |
| Aptamer-conjugated-AuNPs | aptamer–DNA duplex formed by the hybridization reaction between the capture probe and the aptamer probe, which induces a clear enzymatic catalysis of the oxidation of methionine by hydrogen peroxide | Biosensor | C. difficile | 1 nM | [133] |
| glassy carbon electrode modified with graphene oxide and AuNPs | Electrochemical impedance spectrum | Salmonella | 3 CFU/mL | [134] | |
| cell-based SELEX (Systematic Evolution of Ligands by Exponential Enrichment), and dissociation constants and binding specificity | Resonance light-scattering–detection system | S. aureus | 10 CFU/mL 1.5 h | [135] | |
| AuNPs paper-paper | Non-cross-linking assay wax-printed microplate paper platform | Colorimetric detection from red to blue | TB | 30 μg/mL 2 h | [136] |
| Microarrays | |||||
| AuNPs | Multiple capture and intermediate oligos to detect a target in multiple regions | Silver signal by scanomatric detection | Influenza Virus H5N1 | <105 copies of transcribed RNA; 2.5 h | [137] |
| Ag-Au core shell NPs | Nanoparticle-based microarrays using a photodiode sensor | SEM images | HPV | 0.05 pmol/μL | [138] |
| Magnetic nps | |||||
| AuMNPs | Non cross-linking aggregation phenomenon: specific interaction between meca gene with the gold | Colorimetric detection (λ = 530 nm) | MRSA | [139] | |
| Electrochemical geno-sensing assay onto the latex microspheres | AuNPs signal | Vibrio cholerae | 2 CFU/mL | [140] | |
| Fept@Vanco | Trapping gram-positive bacteria, based on interaction between the heptapeptide backbone of vancomycin and the d-alanyl-d-alanine dipeptide from the cell wall | MALDI-TOF | Staphylococcus spp., VRE & E. coli | 100 CFU/mL | [116] |
| Immunoassay | |||||
| Au-NPs | AuNPs bound to anti-human IgG | Colorimetric immunoassay | Influenza Virus | 10 pg/mL | [141] |
| FLFI combined with ELISA | E. coli 0157:H7 | 103 CFU/mL | [142] | ||
| Plasmonic ELISA (ELISA with enzyme-mediated SPR of AuNPs) | SIFILIS | 0.98pg/mL | [143] | ||
| Ag-NPs | SERS enzyme-catalyzed immunoassay | RAMAN Intensity | SRV | 0.05 pg/mL | [144] |
| ELISA, antigen-antibody immunoreaction | Chemiluminescence | Salmonella spp. | 50–100 CFU/mL | [145] | |
| Eu(III)-NPs | Fluorescence signal | HIV-1 p24 | <0.1 pg/mL | [146] | |
| AgNPs-G | gold electrode coated with AuNPs-G, whose is modified with H7-monoclonal antibodies | Electrochemical immunosensor | Aviar Influenza Virus H7 | 1.6 pg/mL | [147] |
| FSNPs | highly fluorescent bioconjugated nanoparticles probe | Fluorescence signal | L. monocitogenes | 50 CFU/mL | [148] |
| Fluorescence | |||||
| Si-MNPs | high specificity for dsDNA and bright fluorescence upon intercalation into dsDNA. | Nucleic-acid dye SYBR Green I signal (Intensity) | S. aureus | 50 CFU/mL | [149] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres-Sangiao, E.; Holban, A.M.; Gestal, M.C. Advanced Nanobiomaterials: Vaccines, Diagnosis and Treatment of Infectious Diseases. Molecules 2016, 21, 867. https://doi.org/10.3390/molecules21070867
Torres-Sangiao E, Holban AM, Gestal MC. Advanced Nanobiomaterials: Vaccines, Diagnosis and Treatment of Infectious Diseases. Molecules. 2016; 21(7):867. https://doi.org/10.3390/molecules21070867
Chicago/Turabian StyleTorres-Sangiao, Eva, Alina Maria Holban, and Monica Cartelle Gestal. 2016. "Advanced Nanobiomaterials: Vaccines, Diagnosis and Treatment of Infectious Diseases" Molecules 21, no. 7: 867. https://doi.org/10.3390/molecules21070867