- 1Division of Epilepsy, Department of Neurological Sciences, University of Nebraska Medical Center, Omaha, NE, United States
- 2Leon S. McGoogan Health Sciences Library, University of Nebraska Medical Center, Omaha, NE, United States
Background: Long-term sequelae of the new onset refractory status epilepticus (NORSE) include the development of epilepsy, cognitive deficits, and behavioral disturbances. The prevalence of these complications has been previously highlighted in case reports and case series: however, their full scope has not been comprehensively assessed.
Methods: We conducted a systematic review of the literature (PROSPERO ID CRD42022361142) regarding neurological and functional outcomes of NORSE at 30 days or longer following discharge from the hospital. A systematic review protocol was developed using guidance from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
Results: Of the 1,602 records for unique publications, 33 reports on adults and 52 reports on children met our inclusion criteria. They contained the description of 280 adults and 587 children of whom only 75.7 and 85% of patients, respectively had data on long-term follow-up. The mean age of adult and pediatric patients was 34.3 and 7.9 years, respectively; and the longest duration of follow up were 11 and 20 years, respectively. Seizure outcomes received major attention and were highlighted for 93.4 and 96.6% of the adult and pediatric NORSE patients, respectively. Seizures remained medically refractory in 41.1% of adults and 57.7% of children, while seizure freedom was achieved in only 26 and 23.3% of these patients, respectively. The long-term cognitive outcome data was provided for just 10.4% of the adult patients. In contrast, cognitive health data were supplied for 68.9% of the described children of whom 31.9% were moderately or severely disabled. Long-term functional outcomes assessed with various standardized scales were reported in 62.2 and 25.5% of the adults and children, respectively with majority of patients not being able to return to a pre-morbid level of functioning. New onset psychiatric disorders were reported in 3.3% of adults and 11.2% of children recovering from NORSE.
Conclusion: These findings concur with previous observations that the majority of adult and pediatric patients continue to experience recurrent seizures and suffer from refractory epilepsy. Moderate to severe cognitive disability, loss of functional independence, and psychiatric disorders represent a hallmark of chronic NORSE signifying the major public health importance of this disorder.
1. Introduction
New onset status epilepticus (NORSE) and its subcategory Febrile Infection Related Encephalopathy Syndrome (FIRES) have been described in adult and pediatric patients under various terms starting as early as 1950's. The term NORSE was coined by Wilder-Smith in 2005; the definition and clinical criteria were formalized in 2018 (1–3). NORSE encompasses various clinical presentations of de novo recurrent refractory seizures without evidence of acute structural, metabolic, or toxic causes (3). The true incidence of NORSE is unknown; however, it may constitute up to 20% of all cases of refractory status epilepticus (4, 5). NORSE most frequently occurs in previously healthy young adults and school-aged children; however, older individuals, including septuagenarians, have also been affected. In adult case series, a higher prevalence has been reported in women. This contrasts with pediatric case series where NORSE predominantly affects boys (1, 6–9). An etiology is identified in up to 50% of the adult patients with NORSE, most of whom suffer from primary or paraneoplastic autoimmune encephalitis (7). On the other hand, paraneoplastic and autoimmune NORSE is rare in children (10–12).
A prodromal phase has been reported in 60–100% of patients with NORSE. The prodrome precedes the onset of seizures and status epilepticus (SE) by 1–14 days (7–9, 13) and includes fever, a cardinal diagnostic criterion of FIRES, in 34–91% of patients (7–9). Other prodromal symptoms include headache, mild gastrointestinal or upper respiratory illness, and behavioral disturbances (7, 8). Electroencephalogram (EEG) abnormalities are present in all NORSE patients, and seizures are detected in more than 88% of patients undergoing continuous video EEG monitoring (7, 8). Brain magnetic resonance imaging (MRI) as well as laboratory examination of serum and cerebrospinal fluid (CSF) for the presence of autoantibodies or abnormal immunoglobulin indexes are routinely performed and are frequently abnormal (7–9).
Strides have been made in evidence-based care for NORSE patients. Criteria for the diagnosis of NORSE has been introduced and accepted by the neurology community (3). In addition, evidence- and experience-based recommendations for the management of patients with NORSE have been published by experts from the International NORSE Consensus Group (14, 15). While significant progress has been made in delineating the diagnostic and treatment approaches for NORSE, less emphasis has been placed on studying clinical outcomes, including the long-term sequalae, after the hospital discharge. Further, the literature focuses primarily on seizure outcomes, as refractory epilepsy represents the most significant disability in survivors of NORSE (1, 2, 6–12, 16–99). However, over two-thirds of patients experience moderate to severe cognitive disability following hospitalization or remain in a vegetative state (5, 6, 92, 93, 99). Reports concerning functional limitations after NORSE are sparse and include components of formal functional assessment or narrative descriptions of impaired academic performance or activities of daily living (1, 2, 6–12, 16–99). The emergence of psychiatric and behavioral disturbances after NORSE have also been described; however, the full spectrum of these complications have not been systematically assessed outside of the time of hospitalization.
In this study, we conducted a systematic review of literature on NORSE and compiled data on the neurological and psychiatric outcomes of NORSE at 30 or more days following hospitalization. The long-term outcomes of SE and refractory SE have been documented in recent systematic reviews where the patient symptoms were assessed starting as early as 30 days after the discharge (100–102). Consistent with previously set criteria (100–102), we considered to use a 30-day mark as an appropriate interval after which the outcomes of SE were considered “long-term.” Given that some of these complications may improve over time, we did not restrict the length of follow-up after hospitalization in these reports. We disaggregated the findings in adult and pediatric patients and summarized the key demographic and clinical features of these cohorts. The purpose of this systematic review is to tackle the special circumstance of de-novo SE and highlight the spectrum of neurocognitive disabilities in patient recovering from NORSE.
2. Methods
The systematic review protocol was developed using guidance from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (103) and registered in the International Prospective Register of Systematic Reviews (PROSPERO, Center for Reviews and Dissemination number CRD42022361142) (104).
2.1. Search strategy
Literature searches were initially carried out from May 30–June 1, 2021, and later updated with the final update on October 13, 2022. MEDLINE (EBSCOhost), CINAHL (EBSCOhost), APAPsycINFO (EBSCOhost), EMBASE (embase.com, version including 1974-present), Scopus, and the Cochrane Library (including the Cochrane Database of Systematic Reviews and The Cochrane Central Register of Controlled Trials, wiley.com) were searched from inception to the final search date. The search strategy was developed by a librarian (C.S.) in consultation with epileptologist with research interest in neuroimmunology and clinical subspecialty in autoimmune epilepsy (O.T.). Each database search included terms representing the “long-term follow-up” and “NORSE” concepts (see complete search strategies available at https://digitalcommons.unmc.edu/search/14). The “long-term follow-up” concept was represented by a variety of subject headings and keywords. Since none of the databases we used had a subject heading for the “NORSE” concept, this concept was represented by keywords and key phrases alone. The following alternate names and acronyms for NORSE were considered during search strategy development: new-onset refractory status epilepticus, NORSE, febrile infection-related epilepsy syndrome, FIRES, febrile illness-related epilepsy, fever-induced refractory epileptic encephalopathy, idiopathic catastrophic epileptic encephalopathy, severe refractory status epilepticus owing to presumed encephalitis, devastating epilepsy in school-age children, DESC, acute non-herpetic encephalitis with refractory repetitive partial seizures, acute encephalitis with refractory repetitive partial seizures, AERRPS, de novo cryptogenic refractory multifocal febrile status epilepticus. The search strategies were designed to retrieve records containing any of these names listed and to retrieve records containing any of the listed acronyms if the record in question also contained a word beginning with one of the following word trunks: epilep, convul, or seizur. Since no funds were available for translation, English- language filters were applied. Conference abstracts, editorials, and review articles were separated from other search results when filters allowed.
2.2. Study selection and quality assessment
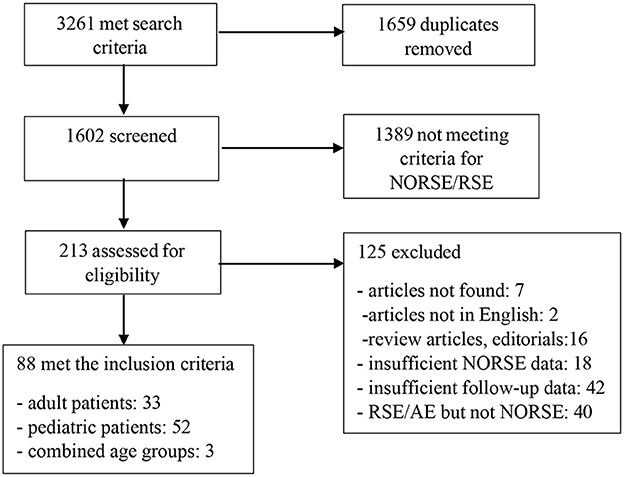
The database searches retrieved 3,261 total records (142 from CINAHL, 24 from the Cochrane Library, 637 from EMBASE, 911 from MEDLINE, 306 from PsycINFO and 1,241 from Scopus, Figure 1). All search results were imported into RefWorks and 1,659 duplicate records were removed using RefWorks' and Zotero duplicate detection tools. A total of 1,602 unique publications remained for title/abstract review (Figure 1). The titles and abstracts were reviewed by two independent neurologists (S.P. and N.G.) for the inclusion criteria and selected articles were chosen for full review. Disagreements between reviewers and inquiries by reviewers were resolved by another reviewer. Reports on other types of SE that did not fulfill the criteria of NORSE as well as those with outcomes reported at <30 days following the discharge were excluded. Case reports and case series were evaluated based on completeness and quality of reporting and were excluded if they did not provide the pertinent information (105).
2.3. Data extraction
Data extraction was performed using a standardized template. In addition to basic demographic data, the following outcomes were extracted: duration of follow-up, proportion of patients with seizures controlled with anti-seizure medications (ASM) or other treatments, proportion of patients with refractory seizures and recurrent SE, presence and severity of acquired cognitive disability with specific reference to memory impairment. In addition, functional outcomes assessed with Modified Rankin Scale (mRS), Clinical Global Impressions of Improvement Scale (CGI-I), Pediatric Cerebral Performance Score (PCPS), or Glasgow outcome score (GOS) at the last follow-up visit and proportion of patients with acquired neurological comorbidities, psychiatric complications, and learning disabilities were extracted.
2.4. Data analysis
Age was determined through weighted means, when possible. The sex distribution, proportions of patients with seizures, SE, cognitive failure, and other comorbidities were assessed when possible. Comparisons of results between the age groups were descriptive only and not statistically assessed. The summaries were disaggregated for the adult patients (19 years and older), the children, and the mixed age cohort. Given that the age range of patients in the individual reports was broad, the outcomes for various age groups could not be stratified. Similarly, given that the range of follow-up time was broad in the individual reports and these intervals were largely non-overlapping, the outcomes for the specific time intervals were not assessed.
3. Results
3.1. Demographic patient characteristics
From 1,602 publications identified by the search, 1,389 reports were excluded after the initial title and abstract screening as they did not meet criteria for NORSE/refractory status epilepticus (RSE), and 213 reports were selected for full manuscript review (Figure 1). Of these 213 articles, 33 studies in adults and 52 studies in children met the inclusion criteria (Figure 1). Three reports contained findings for adult and pediatric age groups. Collectively, the reports contained a description of 280 adult and 587 pediatric patients of which the data on the long-term seizure and cognitive outcomes were available for 212 (75.7 %) adult and 499 (85%) pediatric patients, respectively. In two studies that did not disaggregate findings by age (166 patients total), the data on long-term outcomes were available in 127 (76.5%) patients (51, 59).
In the adult group, the mean age of patients was 34.3 years, and the majority (77.6%) were female. The working diagnosis of NORSE was established in 146 (68.9%) patients, FIRES in 9 (4.3%) patients, new onset super refractory SE (NOSRSE) in 17 (8%) patients, RSE and superrefractory SE (SRSE) in 17 (8%) patients, autoimmune anti-NMDA receptor encephalitis in 2 (0.9%) patients, unspecified autoimmune encephalitis in 19 (8.9%) patients, and acute encephalitis with repetitive recurrent partial seizures (AERRPS) or presumed limbic encephalitis in 2 (0.9%) patients. Pertinent laboratory findings in these patients included the autoantibodies against NMDA receptors (28), GABAA receptors (1), GABAB receptors (2), voltage gated potassium channel complex (VGKC, 7), contactin-associated-protein (CASPR,1), type 1 anti-neuronal nuclear protein (ANNA-1, anti-Hu, 1) and GAD-65 proteins (8) in the serum or CSF; however, the tumor status of patients was not consistently recorded. In the large case series on NORSE, paraneoplastic antibodies in patients with confirmed malignancies included anti-Ro (1), anti- NMDA receptor (9), anti-VGKC (3), anti-Hu (3), anti-voltage gated calcium channel (VGCC, 2) antibodies and collapsing response mediator protein 5 (CRMP-5, 1) (5). The identification of infectious agents, including Epstein-Bar virus and other pathogens (Cytomegalovirus, Herpes simplex virus, human immunodeficiency virus, Mycoplasma pneumoniae, Treponema pallidum, Toxoplasma gondii, Varicella zoster virus, West Nile virus) were reported in 14 patients. Collectively, the etiology of NORSE appeared to be established in 37.7% of 106 patients described in articles meeting our inclusion criteria. Other findings in adult patients were signal abnormalities on the MRI (4) or mass spectroscopy (1) of the brain.
In pediatric studies, the mean age of patients was 7.9 years, and 42.5 % were female. The diagnosis of NORSE or FIRES were established in 149 (29.8%) and 237 (47.4 %) of patients, respectively. Other diagnoses included AERRPS in 35 (7%) patients, devastating encephalopathy in school age children (DESC) in 14 (2.8%) patients, hemiconvulsion-hemiplegia syndrome (HHE) in 35 (7%) patients, SE-related presumed encephalitis in 19 (3.8%) patients, Mycoplasma pneumoniae encephalitis in 5 (1%) patients, anti-NMDA receptor encephalitis in 3 (0.6%) patients, as well as anti-GAD encephalitis, steroid-responsive encephalopathy and associated autoimmune thyroiditis (SREAT) or SRSE in 1 patient each (0.2%). The pertinent findings in patients' evaluation included presence of autoantibodies in the serum and CSF specimens in 19 (3.2%) patients. The antibodies against NMDA receptors (2), GABAA receptors (1), AMPA-GluR3 receptors (1), and GAD-65 protein (6) as well as elevated serum and CSF anti-thyroid peroxidase (TPO) antibodies (1) were documented along with the increase of CSF cytokines such as tumor necrosis factor and interleukins 6 and 10 (1). Genetic deficiencies (5) and secondary hemophagocytic lymphohistiocytosis (3) were also recorded as the possible etiologies of NORSE. Brain MRI examinations were abnormal in 31 patients and revealed various degrees of cortical atrophy (10), signal changes in the claustrum (1), temporal cortical regions (2), and cerebellum (1) as well as bilateral hippocampal atrophy (1).
In two large reports that collectively included 127 adult and pediatric patients, the proportion of female patients was smaller than male (41.7%); but the age of participants was not consistently provided (51, 59). The autoimmune antibodies were not identified in serum and CSF of these patients. The cortical and hippocampal signal abnormalities on brain MRI were reported in 27 patients.
3.2. Seizure outcomes
The duration of follow-up in the adult group ranged from 30 days to 11 years. Seizure outcomes were reported in 198 out of 212 patients (93.4%, Supplementary Table 1). Of these, 10 (5.1%) patients become seizure-free and discontinued the ASMs, while 28 (14.1%) and 15 (7.6%) patients were seizure-free when receiving the ASMs alone or ASM in combination with other treatments, respectively. In the 10 patients who were seizure-free, the treatment status was not reported. Medically refractory epilepsy was diagnosed in 82 (41.4%) patients for whom other treatment approaches were tried, including immunotherapies [steroids, intravenous immunoglobulin (IVIG), tacrolimus, mycophenolate mofetil, rituximab, anakinra, plasma exchange, cyclophosphamide] in various combinations (25) as well ketogenic diet (5), modified Atkin's diet (3), neurostimulation (2) and focal cortical resections (5). Delayed recurrent SE was reported in two patients.
In the pediatric group, the duration of follow-up ranged from 30 days to 20 years. Seizure outcomes were reported for 482 (96.6%) patients (Supplementary Table 2). Forty-seven patients (9.8%) were seizure free without ASMs, while 66 (13.7%) patients achieved seizure control with ASMs alone or when treated with ASMs combined with ketogenic diet. The treatment status in an additional 27 patients was not recorded, although they were reported to be seizure free. Medically refractory epilepsy was documented in 278 (57.7%) patients of whom 34 patients required therapies beyond ASMs, including steroids (3), IVIG (5), tocilizumab (2), anakinra (2), and ketogenic diet (12). Neuromodulation (7) and focal resections (2) were also listed among the treatments for refractory seizures. Recurrent SE was reported in 8 patients.
3.3. Cognitive outcomes
Cognitive outcomes at 30 days or later following the hospital discharge were reported for 22 (10.4%) of the adult patients included in the reviewed literature; most of these patients were described in case reports (Supplementary Table 1). Most of the studies assessed cognitive outcomes subjectively. All available data on neuropsychological testing, including the formal test scores are provided in Supplementary Tables 2, 3. At the last follow-up, 3 patients (13.6%) had normal cognition, 2 (9.1%) had mild cognitive impairment, and 4 (18.2%) and 2 (9.1%) had moderate or severe degrees of cognitive impairment, respectively. Three patients (13.6%) remained in a vegetative state; however, the time of assessment (10 months) was only documented for one patient. Memory impairment was reported in 12 patients (54.5%). Four (33.3%) of these had moderate or severe impairment. Specific reference to working or visual memory impairments were made in 2 patients. Mild naming deficits (1) and persistent impairment of processing speed and verbal memory function (1) were also reported at the last follow-up.
Reports concerning pediatric patients elaborated on cognitive outcomes after hospital discharge more frequently than reports concerning adults. The assessment of cognitive status was documented in 68.9% of all 344 included pediatric patients with NORSE (Supplementary Tables 2, 3). Seventy-eight patients (22.7%) have experienced complete cognitive recovery. Mild impairment was diagnosed in 42 children (12.2%), while moderate and severe loss were noted in 60 (17.4%) and 50 (14.5%) of children, respectively. Thirty patients (8.7%) have remained in a vegetative state. The degree of intellectual disability was not specified in 94 (27.3%) children. References to specific memory impairment were made in reports concerning 19 patients of whom 8 had a severe memory loss. Delayed motor, social, and verbal development were noted in 1 child described in a case report.
3.4. Functional outcomes and activities of daily living
In adult reports, mRS scores and activities of daily living data were included in 123 (58%) and 13 (6.1%) of patients, respectively (Supplementary Table 1). In 37 (30%) patients, the specific values of mRS were provided and were as follows: 23 patients (30.1 %) with scores of 0–1, 27 (22.5%) with scores of 2–3, and 24 (19.5%) with scores of 4–6. Eight patients had no formal assessment but were noted to return to baseline or had good recovery and have remained autonomous in their day-to-day functioning. Three patients had not regained consciousness. The GOS was reported in 9 (4.2%) patients of whom 2, 1, and 6 had scores of 5, 3, and 1, respectively (Supplementary Table 1). A report of 14 patients had only narrative characterization of the activities of daily living. Eight of these 14 patients were described as being independent and 6 patients as needing assistance. Other assessments included mentioning of a patient's ability to resume previous academic activities (1) and another patient's referral to a supervised nursing facility (1).
Pediatric patients' functional outcomes as assessed with mRS, PCPS, CGI-I, pediatric GOS, and GOS were reported in 61 (12.2%), 63 (12.6%), 5 (1%), 18 (3.6%), and 16 (3.2%) patients, respectively (Supplementary Table 2). There were 6, 2, and 6 patients with the mRS scores of 0–1, 2–3, and 4–6, respectively. Sixty-three patients were assessed with PCPS. Favorable outcomes, defined as score ≤ 2, were noted in 12 (19.1%) children while unfavorable outcomes (i.e., score >3) were noted in 51 (80.9%) patients. The outcomes assessed with CGI-I were distributed as follows: favorable (i.e., score of 2–3) in 3 patients, and without change (i.e., score 4) in 2 patients. The subdomains of communication and autonomy were reported in 5 patients. Pediatric GOS was used in two studies to assess outcomes with a score of 1 and 4 corresponding to the good recovery and vegetative state, respectively. There were 4, 3, 7, and 4 patients with scores of 1, 2, 3, and 4, respectively. Three other studies utilized GOS with scores that were defined differently such that 5 was consistent with good recovery and 2 was consistent with vegetative state. Scores of 5, 4, 3, and 2 were reported in 7, 4, 1, and 4 patients, respectively. Of 87 patients, for whom the activities of daily living were characterized, 55 (63.2%) were independent and 32 (36.8%) required assistance. In the narrative descriptions of the functional status, there were reports of resuming premorbid academic activities (3), having some academic difficulties (14), requiring special education (1), developing learning disabilities (10), having attention deficits and executive dysfunction (2), and developing severe developmental delay (5).
3.5. Acquired psychiatric comorbidities and neurological deficits
The emergence of new onset psychiatric disorders in adult survivors of NORSE were reported in only 7 (3.3%) patients (Supplementary Table 1). These manifestations included schizophrenia (1), attention deficit disorder (2), Capgras syndrome (1), psychomotor agitation (1), and personality changes (2). Various neurological deficits were reported in 6 patients and included bilateral lower extremity weakness and mild ataxia (1), right hemiparesis and dysphasia (1), mild receptive dysphasia (1), and moderate language impairment (1) as well as unspecified gait disturbance (1).
The description of psychiatric complications of NORSE were more common in the pediatric literature and were provided for 56 (11.2%) of all included children (Supplementary Table 2). Reports concerning 56 of the pediatric patients mentioned behavioral disturbances of various severity: 13 developed aggression, 2 suffered from emotional lability, 4 had apathy, and 1 had conduct disorder. Mild or severe attention deficit hyperactivity disorder and various manifestations of attention impairment were diagnosed in 9 patients and an autism spectrum disorder was reported in 1 patient. One child attempted suicide. Various neurological sequalae of NORSE were described in 108 (21.6%) children. Specifically, the long-term motor deficits included hemiplegia (35, 32.4%), unspecified motor impairments (36, 33.3%) and unilateral tongue weakness (1, 0.01%). Other impairments included visual field deficits (1), peripheral neuropathy (2), ataxia (3), choreoathetoid movements (1), and tremor (3). Language deficits were reported in 20 (18.5%) patients. One of these children improved after the initiation of responsive neurostimulation.
4. Discussion
In the present study, we systematically reviewed and summarized the literature on the long-term neurological and psychiatric outcomes of NORSE in adult and pediatric patients who survived longer than 30 days after hospital discharge. We found that seizure status is assessed in over 90% of patients who had the data on the long-term outcomes. However, the cognitive outcomes were only included in one tenth of these reports in adults and nearly two-thirds of the reports concerning children. Functional outcomes were included in more than 60% of studies of adults and more than 25 % of studies of children. Unfortunately, the functional outcomes were measured using four different outcome scales limiting our ability to synthesize study results into an understanding of the overall scope of associated disability. New onset psychiatric disorders were under-reported and were only included in a small proportion of the reports. Overall, these findings reflect the lack of standardization for the reporting of outcomes, particularly for reporting symptoms other than seizures, and may also reflect a gap in the care of these patients after the initial hospital encounters.
4.1. Seizure outcomes
Consistent with previous observations, in this systematic review, we found that majority of adults and children with NORSE will continue to have seizures 30 days or later after the hospital discharge. Of those who continue to have seizures, 41.1% of adults and 57.7% of children will remain refractory to either conventional ASDs used alone or in combination with immunotherapies, ketogenic diet, and neurostimulation. The pathogenesis of recurrent seizures in NORSE is not clear. Several proposed mechanisms of uncontrolled seizure generation during the acute phase of NORSE included aberrant signaling in the interleukin (IL)-1 and toll-like receptor (TLR)-mediated pathways, overactivation of the NLRP3 inflammasome as well as functional or genetic deficiency of IL-1 receptor antagonist activity (48, 67, 106–113). These mechanisms can also be involved in late seizure recurrence in survivors of NORSE. Of note, chronic epilepsy in cryptogenic NORSE develops without a latent period which is distinct from the post-infectious epilepsies associated with viral or bacterial pathogens (10, 114).
4.2. Cognitive outcomes
Our findings add to those of previous studies that showed only a small proportion of patients have achieved their pre-morbid cognitive function after they were discharged from the hospital (9). Nearly one-third of adults or children continue to suffer from moderate and severe intellectual disability. In a case series of 14 pediatric patients with NORSE, all children attended special education in the later course, and seven patients had severe cognitive failure. The primary impairment involved deficits in frontal lobe function and was manifesting as the lack of motor and speech initiative, major slowness, perseveration, and poor attention (9, 10). While there were no specific patterns in neuropathological findings in NORSE, gliosis, laminar cortical necrosis, and diffuse cortical atrophy are the shared common features (1, 6, 99, 115, 116). Moreover, various degrees of persistent inflammation and structural changes such as mesial temporal sclerosis may contribute to the severity of cognitive phenotype.
Chronic cognitive disability after NORSE represents the major public health problem. Since many patients are previously healthy, severe cognitive impairment or vegetative states after NORSE are devastating. A need exists for comprehensive chronic care that includes cognitive rehabilitation for patients and respite for caregivers. The mechanism of cognitive failure in NORSE is unclear, but it is likely linked to the severity and duration of seizures (91). In subtypes of autoimmune encephalitis that can manifest as NORSE (e.g., anti-NMDA receptor encephalitis), antibodies were found to be directly pathogenic for memory failure and seizures. However, the pathogenesis of epileptic encephalopathy in other antibody-mediated or cryptogenic NORSE is not clear (7, 117, 118). The lack of the uniformed objective measurements of cognitive function noted in the reviewed literature represents a major shortcoming of this research. More systematic and comprehensive objective testing of survivals is needed in future studies.
4.3. Functional outcomes and activities of daily living
We found that a comprehensive approach in documenting the functional outcomes in survivors of NORSE were lacking. Further, only one-fourth of the children and 60% of adults in our study had their functional outcomes assessed and reported. While the mRS was the most common outcome scale applied in adult patients, there were three additional outcomes scales used in pediatric patients. Such inconsistent data availability in outcomes particularly for the pediatric patients introduce a potential bias and limits definitive conclusions. In the recent systematic analysis of functional outcomes in autoimmune encephalitis, it was established that mRS had poor sensitivity for cognitive disability and mood disturbances in encephalitis at follow-up (119). There was an additional focus on the academic performance and other aspects of social functioning in children, which were consistently underreported in the adult literature. Given that many patients with NORSE have now survived for several decades, a more standardized approach in categorization of their functional abilities is needed to monitor their recovery and develop guidelines for individualized rehabilitation.
4.4. Acquired psychiatric comorbidities and neurological deficits
Psychiatric comorbidities, including recurrent psychosis are frequently encountered in severe epileptic encephalopathies (120); however, the prevalence of mental illness in association with recent refractory SE is unknown. Multiple factors could contribute to the development of psychosis and depression in patients recovering from NORSE, including the individual vulnerability to the effects of ASDs or immunotherapy as well as prolonged brain hypoxia (121). The presence of specific autoimmune antibodies (such as anti-AMPA or anti-NMDA receptor antibodies) in NORSE with established etiologies can guide the anticipation of chronic psychiatric sequalae (122). The accounts of new onset psychiatric disorders in only 3.3 and 11.2% of the adults and pediatric patients with chronic NORSE likely reflects underreporting and insufficient attention to these comorbidities on the part of neurologists involved in care of these patients.
4.5. Limitations
Our study has several limitations. We found that the majority of reports on long-term outcomes of NORSE are focused on seizure status while other domains of the neurological and psychiatric health were assessed and commented on inconsistently. This likely represents a reporting bias and underreporting in various relevant health domains for the most severely disabled patients (e.g., those in vegetative state) and those whose seizures were under better control. This limits our conclusions regarding the prevalence of multiple manifestations of chronic NORSE other than seizures. Given the retrospective and observational design of most reviewed studies, it is unclear whether the severity of SE and its refractoriness can be linked to any of the reported outcomes. This impedes our progress in understanding the mechanisms of cognitive failure and development of psychiatric comorbidities in NORSE. Given that the analysis in specific age categories in pediatric patients was not feasible, the outcomes of SE at different stages of brain development and the effects of age-related compensatory abilities have not been accounted for. Likewise, the inability to disaggregate the data into the specific duration of follow-up precluded the analysis of outcomes at different stages of recovery from NORSE. Lastly, we acknowledge the selection and information bias that was not specifically assessed in this systematic review.
5. Conclusions
We found that most patients with chronic NORSE continue to experience recurrent seizures, and seizure treatment and reporting remain the main focus of the literature. Documentation of cognitive disability, loss of functional independence, and onset psychiatric manifestations have been inconsistent and should be interpreted with caution given the methodological limitations. While challenging to implement due to the rarity and geographic dispersion of NORSE, future prospective studies may help provide high-quality evidence to guide the management and rehabilitation of these patients.
Author contributions
OT: conceptualizing the study, drafting of the manuscript for content, and major role in the analysis and interpretation of data. SP: main role in the acquisition and analysis of data. CS: main role in the acquisition and analysis of data as well as study design. YP: acquisition and analysis of data. NG: drafting of the manuscript and main role in the acquisition and analysis of data as well as study design. All authors contributed to the article and approved the submitted version.
Funding
OT received salary and research support from the NIH P20GM130447 Cognitive Neuroscience and Development of Aging (CONDA) Award and Nebraska Stem Cell Grant (NE DHHS, LB606).
Acknowledgments
The authors would like to thank Robin Taylor for her excellent editorial assistance.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2023.1095061/full#supplementary-material
References
1. Wilder-Smith E, Lim ECH, Teoh HL, Sharma VK, Tan JJH, Chan BPL, et al. The NORSE (new-onset refractory status epilepticus) syndrome: defining a disease entity. Ann Acad Med Singap. (2005) 34:417–20.
2. Ismail FY, Kossoff EH. AERRPS, DESC, NORSE, FIRES: multi-labeling or distinct epileptic entities? Epilepsia. (2011) 52:e185–9. doi: 10.1111/j.1528-1167.2011.03293.x
3. Hirsch LJ, Gaspard N, van Baalen A, Nabbout R, Demeret S, Loddenkemper T, et al. Proposed consensus definitions for new-onset refractory status epilepticus (NORSE), febrile infection-related epilepsy syndrome (FIRES), and related conditions. Epilepsia. (2018) 59:739–44. doi: 10.1111/epi.14016
4. Gaspard N. Unusual causes of status epilepticus. In:Drislane FW, Kaplan MB, , editors. Status Epilepticus: A Clinical Perspective. Cham: Springer International Publishing (2018). p. 87–110.
5. Gaspard N, Hirsch LJ, Sculier C, Loddenkemper T, van Baalen A, Lancrenon J, et al. New-onset refractory status epilepticus (NORSE) and febrile infection–related epilepsy syndrome (FIRES): state of the art and perspectives. Epilepsia. (2018) 59:745–52. doi: 10.1111/epi.14022
6. Costello DJ, Kilbride RD, Cole AJ. Cryptogenic new onset refractory status epilepticus (NORSE) in adults-infectious or not? J Neurol Sci. (2009) 277:26–31. doi: 10.1016/j.jns.2008.10.007
7. Gaspard N, Foreman BP, Alvarez V, Kang CC, Probasco JC, Jongeling AC, et al. New-onset refractory status epilepticus. Neurology. (2015) 85:1604–13. doi: 10.1212/WNL.0000000000001940
8. Iizuka T, Kanazawa N, Kaneko J, Tominaga N, Nonoda Y, Hara A, et al. Cryptogenic NORSE. Neurol Neuroimmunol Neuroinflamm. (2017) 4:e396. doi: 10.1212/NXI.0000000000000396
9. Kramer U, Chi C, Lin K, Specchio N, Sahin M, Olson H, et al. Febrile infection-related epilepsy syndrome (FIRES): Pathogenesis, treatment, and outcome: a multicenter study on 77 children. Epilepsia. (2011) 52:1956–65. doi: 10.1111/j.1528-1167.2011.03250.x
10. Mikaeloff Y, Jambaqué I, Hertz-Pannier L, Zamfirescu A, Adamsbaum C, Plouin P, et al. Devastating epileptic encephalopathy in school-aged children (DESC): a pseudo encephalitis. Epilepsy Res. (2006) 69:67–79. doi: 10.1016/j.eplepsyres.2006.01.002
11. Caraballo RH, Reyes G, Avaria MFL, Buompadre MC, Gonzalez M, Fortini S, et al. Febrile infection-related epilepsy syndrome: a study of 12 patients. Seizure. (2013) 22:553–9. doi: 10.1016/j.seizure.2013.04.005
12. Patil SB, Roy AG, Vinayan KP. Clinical profile and treatment outcome of febrile infection-related epilepsy syndrome in South Indian children. Ann Indian Acad Neurol. (2016) 19:188–94. doi: 10.4103/0972-2327.173305
13. Van Baalen A, Vezzani A, Häusler M, Kluger G. Febrile infection-related epilepsy syndrome: clinical review and hypotheses of epileptogenesis. Neuropediatrics. (2017) 48:5–18. doi: 10.1055/s-0036-1597271
14. Wickstrom R, Taraschenko O, Dilena R, Payne ET, Specchio N, Nabbout R, et al. International consensus recommendations for management of new onset refractory status epilepticus including febrile infection-related epilepsy syndrome: statements and supporting evidence. Epilepsia. (2022) 63:2840–64. doi: 10.1111/epi.17397
15. Wickström R, Taraschenko O, Dilena R, Payne ET, Specchio N, Nabbout R, et al. International consensus recommendations for management of new onset refractory status epilepticus (NORSE) including febrile infection-related epilepsy syndrome (FIRES): Summary and clinical tools. Epilepsia. (2022) 63:2827–39. doi: 10.1111/epi.17391
16. Acampora R, Quiroga Subirana P, Durante L, Tonziello R, Aversano G, Lieto M, et al. A case of febrile infection-related epilepsy syndrome (FIRES) in young adult: still a diagnostic and therapeutic challenge. Neurol Sci. (2022) 43:4555–8. doi: 10.1007/s10072-022-06106-8
17. Al-Chalabi M, Amsdell J, Iftikhar S, Hollingshead C, Rashid M, Sheikh A. New onset refractory status epilepticus secondary to HIV CNS viral escape syndrome: case report. Seizure. (2022) 94:112–4. doi: 10.1016/j.seizure.2021.11.027
18. Aledo-Serrano A, Hariramani R, Gonzalez-Martinez A, Álvarez-Troncoso J, Toledano R, Bayat A, et al. Anakinra and tocilizumab in the chronic phase of febrile infection-related epilepsy syndrome (FIRES): effectiveness and safety from a case-series. Seizure. (2022) 100:51–5. doi: 10.1016/j.seizure.2022.06.012
19. Aydemir S, Kandula P. High dose cannabidiol (CBD) in the treatment of new-onset refractory status epilepticus (NORSE). Seizure. (2022) 94:126–8. doi: 10.1016/j.seizure.2021.11.020
20. Basso M, Gastaldi M, Leonardi V, Izzo G, Olivotto S, Ferrario S, et al. Connections Between febrile infection-related epilepsy syndrome and autoimmune encephalitis. a case report of a child with new anti-neuronal antibodies. Front Pediatr. (2022) 10:908518. doi: 10.3389/fped.2022.908518
21. Jain V, Konanki R, Chaitra R, Srivastava K, Sharma R. Enteral lorazepam is a promising weaning strategy for midazolam-responsive febrile infection-related epilepsy syndrome (FIRES): a case series. Disord. (2022) 24:667–76. doi: 10.1684/epd.2022.1438
22. Luo T, Wang Y, Lu G, Zhou Y, Wang Y. Vagus nerve stimulation for super-refractory status epilepticus in febrile infection–related epilepsy syndrome: a pediatric case report and literature review. Child's Nerv Syst. (2022) 38:1401–4. doi: 10.1007/s00381-021-05410-6
23. Nawfal O, Toufaili H, Dib G, Dirani M, Beydoun A. New-onset refractory status epilepticus as an early manifestation of multisystem inflammatory syndrome in adults after COVID-19. Epilepsia. (2022) 63:e51–6. doi: 10.1111/epi.17231
24. Obara K, Ono T. Ketogenic diet for a young adult patient with chronic-phase febrile infection-related epilepsy syndrome. Cureus. (2022) 14:e22099. doi: 10.7759/cureus.22099
25. Perulli M, Cicala G, Turrini I, Musto E, Quintiliani M, Gambardella ML, et al. Fighting autoinflammation in FIRES: the role of interleukins and early immunomodulation. Epi Beh Rep. (2022) 18:100531. doi: 10.1016/j.ebr.2022.100531
26. Sivathanu D, Kewalramani D, Kumar Manokaran R. Favorable response to classic ketogenic diet in a child with anti-GAD 65 antibody mediated super refractory status epilepticus. Epi Beh Rep. (2022) 19:100557. doi: 10.1016/j.ebr.2022.100557
27. Acar T, Boncuk S, Acar B, Aras YG. Complete recovery in cryptogenic norse with early immunotherapy: a case report. Turk Noroloji Derg. (2021) 27:79–81. doi: 10.4274/tnd.2020.01879
28. Baba S, Okanishi T, Ohsugi K, Suzumura R, Niimi K, Shimizu S, et al. Possible role of high-dose barbiturates and early administration of parenteral ketogenic diet for reducing development of chronic epilepsy in febrile infection-related epilepsy syndrome: a case report. Neuropediatrics. (2021) 52:133–7. doi: 10.1055/s-0040-1716903
29. Choi C, Ma S, Ma KK, Leung H, Mok VC. Super-refractory status epilepticus in autoimmune encephalitis treated with interleukin-1 receptor antagonist, anakinra. Epileptic Disord. (2021) 23:500–5. doi: 10.1684/epd.2021.1283
30. Donnelly JP, Kasatwar N, Hafeez S, Seifi A, Gilbert A, Barthol C, et al. Resolution of cryptogenic new onset refractory status epilepticus with tocilizumab. Epi Beh Rep. (2021) 15:100431. doi: 10.1016/j.ebr.2021.100431
31. Horino A, Kuki I, Inoue T, Nukui M, Okazaki S, Kawawaki H, et al. Intrathecal dexamethasone therapy for febrile infection-related epilepsy syndrome. Ann Clin Transl Neurol. (2021) 8:645–55. doi: 10.1002/acn3.51308
32. Karunaratne K, Ahrabian D, Monoghan B, Campion T, Yousry T, Lunn MP, et al. Bortezomib for anti-NMDAR encephalitis following daclizumab treatment in a patient with multiple sclerosis. BMJ Neurol Open. (2021) 3:e000096. doi: 10.1136/bmjno-2020-000096
33. Nath M, Shah YD, Theroux LM, Petrides G, Karkare S, Sanghani SN, et al. A role for electroconvulsive therapy in the management of new onset refractory status epilepticus (NORSE) in a young child. Neurol India. (2021) 69:1374–9. doi: 10.4103/0028-3886.329559
34. Wickstrom R, Taraschenko O, Dilena R, Payne ET, Specchio N, Nabbout R, et al. Clinical presentation of new onset refractory status epilepticus in children (the pSERG cohort). Epilepsia. (2021) 62:1629. doi: 10.1111/epi.16950
35. Wang X, Wan J, Wei Z, Song C, Kang X, Du F, et al. Status epilepticus in patients with anti-NMDAR encephalitis requiring intensive care: a follow-up study. Neurocrit Care. (2022) 36:192–201. doi: 10.1007/s12028-021-01283-4
36. Wu J, Lan X, Yan L, Hu Y, Hong S, Jiang L, et al. A retrospective study of 92 children with new-onset refractory status epilepticus. Epilepsy Behav. (2021) 125:108413. doi: 10.1016/j.yebeh.2021.108413
37. Gugger JJ, Husari K, Probasco JC, Cervenka MC. New-onset refractory status epilepticus: a retrospective cohort study. Seizure. (2020) 74:41–8. doi: 10.1016/j.seizure.2019.12.002
38. Husari KS, Labiner K, Huang R, Said RR. New-onset refractory status epilepticus in children: etiologies, treatments, and outcomes. Pediatr Crit Care Med. (2020) 21:59–66. doi: 10.1097/PCC.0000000000002108
39. Jaafar F, Haddad L, Koleilat N, Sharara-Chami R, Shbarou R. Super refractory status epilepticus secondary to anti-GAD antibody encephalitis successfully treated with aggressive immunotherapy. Epi Beh Rep. (2020) 14:100396. doi: 10.1016/j.ebr.2020.100396
40. Kim HJ, Lee S, Kim H, Kim SJ, Jeon S, Koo YS. The timelines of MRI findings related to outcomes in adult patients with new-onset refractory status epilepticus. Epilepsia. (2020) 61:1735–48. doi: 10.1111/epi.16620
41. Matthews E, Alkhachroum A, Massad N, Letchinger R, Doyle K, Claassen J, et al. New-onset super-refractory status epilepticus: a case series of 26 patients. Neurology. (2020) 95:e2280–5. doi: 10.1212/WNL.0000000000010787
42. Stredny CM, Case S, Sansevere AJ, Son M, Henderson L, Gorman MP. Interleukin-6 blockade with tocilizumab in Anakinra-refractory febrile infection-related epilepsy syndrome (FIRES). Child Neurol Open. (2020) 7. doi: 10.1177/2329048X20979253
43. Theroux L, Shah Y, Cukier Y, Rodgers S, Karkare S, Bonda D, et al. Improved seizure burden and cognitive performance in a child treated with responsive neurostimulation (RNS) following febrile infection related epilepsy syndrome (FIRES). Epileptic Disord. (2020) 22:811–6. doi: 10.1684/epd.2020.1224
44. Al-Khateeb M, Adem F, Moqbel A, Baz S. Schizophrenia following new-onset refractory status epilepticus secondary to antiphospholipid syndrome. Neurosciences. (2019) 24:240–4. doi: 10.17712/nsj.2018.3.20180014
45. Fatuzzo D, Giuliano L, Mainieri G, Sortino G, Sofia V, Zappia M. Febrile infection-related epilepsy syndrome (FIRES) in an adult patient: an early neuroradiological finding. Neurol Sci. (2019) 40:2407–10. doi: 10.1007/s10072-019-03925-0
46. Lam S, Lu W, Weng W, Fan P, Lee W. The short-term and long-term outcome of febrile infection-related epilepsy syndrome in children. Epilepsy Behav. (2019) 95:117–23. doi: 10.1016/j.yebeh.2019.02.033
47. Peng P, Peng J, Yin F, Deng X, Chen C, He F, et al. Ketogenic diet as a treatment for super-refractory status epilepticus in febrile infection-related epilepsy syndrome. Front Neurol. (2019) 10:423. doi: 10.3389/fneur.2019.00423
48. Westbrook C, Subramaniam T, Seagren RM, Tarula E, Co D, Furstenberg-Knauff M, et al. Febrile infection-related epilepsy syndrome treated successfully with Anakinra in a 21-year-old woman. Wisc Med J. (2019) 118:135–9.
49. Albakaye M, Belaïdi H, Lahjouji F, Errguig L, Kuate C, Maiga Y, et al. Clinical aspects, neuroimaging, and electroencephalography of 35 cases of hemiconvulsion-hemiplegia syndrome. Epilepsy Behav. (2018) 80:184–90. doi: 10.1016/j.yebeh.2017.12.018
50. Caputo D, Iorio R, Vigevano F, Fusco L. Febrile infection-related epilepsy syndrome (FIRES) with super-refractory status epilepticus revealing autoimmune encephalitis due to GABAAR antibodies. Eur J Paediatr Neurol. (2018) 22:182–5. doi: 10.1016/j.ejpn.2017.11.005
51. Chen W, Su Y, Jiang M, Liu G, Tian F, Ren G. Status epilepticus associated with acute encephalitis: long-term follow-up of functional and cognitive outcomes in 72 patients. Eur J Neurol. (2018) 25:1228–34. doi: 10.1111/ene.13678
52. Farias-Moeller R, LaFrance-Corey R, Bartolini L, Wells EM, Baker M, Doslea A, et al. Fueling the FIRES: hemophagocytic lymphohistiocytosis in febrile infection-related epilepsy syndrome. Epilepsia. (2018) 59:1753–63. doi: 10.1111/epi.14524
53. Jafarpour S, Hodgeman RM, De Marchi Capeletto C, de Lima MTA, Kapur K, Tasker RC, et al. New-onset status epilepticus in pediatric patients: causes, characteristics, and outcomes. Pediatr Neurol. (2018) 80:61–9. doi: 10.1016/j.pediatrneurol.2017.11.016
54. Lee H, Chi C. Febrile infection-related epilepsy syndrome (FIRES): therapeutic complications, long-term neurological and neuroimaging follow-up. Seizure. (2018) 56:53–9. doi: 10.1016/j.seizure.2018.02.003
55. Lin J, Wang Y, Lan S, Chan O, Hsia S, Chou M, et al. Combination of intravenous immunoglobulin and steroid pulse therapy improves outcomes of febrile refractory status epilepticus. Epilepsy Res. (2018) 142:100–5. doi: 10.1016/j.eplepsyres.2018.03.017
56. Okanishi T, Fujimoto A, Hashimoto R, Nishimura M, Kanai S, Ogawa M, et al. Epileptic spasms secondary to acute cerebral and cerebellar encephalitis. Brain Dev. (2018) 40:218–21. doi: 10.1016/j.braindev.2017.11.006
57. Shin JW, Koo YS, Kim YS, Kim DW, Kim KK, Lee SY, et al. Clinical characterization of unknown/cryptogenic status epilepticus suspected as encephalitis: a multicenter cohort study. J Neuroimmunol. (2018) 315:1–8. doi: 10.1016/j.jneuroim.2017.12.004
58. Steriade C, Moosa ANV, Hantus S, Prayson RA, Alexopoulos A, Rae-Grant A. Electroclinical features of seizures associated with autoimmune encephalitis. Seizure. (2018) 60:198–204. doi: 10.1016/j.seizure.2018.06.021
59. Yuan F, Yang F, Jia R, Li W, Jiang Y, Zhao J, et al. Multimodal predictions of super-refractory status epilepticus and outcome in status epilepticus due to acute encephalitis. Front Neurol. (2018) 99:832 doi: 10.3389/fneur.2018.00832
60. Alparslan C, Kamit-Can F, Anil AB, Olgaç-Dündar N, Çavuşoglu D, Göç Z. Febrile infection-related epilepsy syndrome (FIRES) treated with immunomodulation in an 8-year-old boy and review of the literature. Turk J Pediatr. (2017) 59:463–6. doi: 10.24953/turkjped.2017.04.014
61. Basha MM, Suchdev K, Dhakar M, Kupsky WJ, Mittal S, Shah AK. Acute resective surgery for the treatment of refractory status epilepticus. Neurocrit Care. (2017) 27:370–80. doi: 10.1007/s12028-017-0381-z
62. Gofshteyn JS, Wilfong A, Devinsky O, Bluvstein J, Charuta J, Ciliberto MA, et al. Cannabidiol as a potential treatment for febrile infection-related epilepsy syndrome (FIRES) in the acute and chronic phases. J Child Neurol. (2017) 32:35–40. doi: 10.1177/0883073816669450
63. Morita M, Fujimoto A, Okanishi T, Nishimura M, Sato K, Kanai S, et al. Vagus nerve stimulation therapy improved refractory epilepsy secondary to acute encephalitis with refractory, repetitive partial seizures (AERRPS). Interdiscip Neurosurg. (2017) 9:76–9. doi: 10.1016/j.inat.2017.03.007
64. Patel NA, Jerry JM, Jimenez XF, Hantus ST. New-onset refractory status epilepticus associated with the use of synthetic cannabinoids. Psychosomatics. (2017) 58:180–6. doi: 10.1016/j.psym.2016.10.006
65. Dillien P, Ferrao Santos S, Van Pesch V, Suin V, Lamoral S, Hantson P. New-onset refractory status epilepticus: more investigations, more questions. Case Rep Neurol. (2016) 8:127–33. doi: 10.1159/000447295
66. Gagnon M, Savard M, Mourabit Amari K. Refractory status epilepticus and autoimmune encephalitis with GABAAR and GAD65 antibodies: a case report. Seizure. (2016) 37:25–7. doi: 10.1016/j.seizure.2016.02.006
67. Kenney-Jung D, Vezzani A, Kahoud RJ, LaFrance-Corey R, Ho M, Muskardin TW, et al. Febrile infection-related epilepsy syndrome treated with Anakinra; biosciences(United States). Ann Neurol. (2016) 80:939–45. doi: 10.1002/ana.24806
68. Rivas-Coppola M, Shah N, Choudhri AF, Morgan R, Wheless JW. Chronological evolution of magnetic resonance imaging findings in children with febrile infection-related epilepsy syndrome. Pediatr Neurol. (2016) 55:22–9. doi: 10.1016/j.pediatrneurol.2015.09.003
69. Sato Y, Numata-Uematsu Y, Uematsu M, Kikuchi A, Nakayama T, Kakisaka Y, et al. Acute encephalitis with refractory, repetitive partial seizures: Pathological findings and a new therapeutic approach using tacrolimus. Brain Dev. (2016) 38:772–6. doi: 10.1016/j.braindev.2016.02.006
70. Uchida T, Takayanagi M, Kitamura T, Nishio T, Numata Y, Endo W, et al. High-dose phenobarbital with intermittent short-acting barbiturates for acute encephalitis with refractory, repetitive partial seizures. Pediatr Int. (2016) 58:750–3. doi: 10.1111/ped.12934
71. Capizzi G, Vittorini R, Torta F, Davico C, Rainò E, Conio A, et al. Lidocaine treatment in refractory status epilepticus resulting from febrile infection-related epilepsy syndrome: a case report and follow-up. Neuropediatrics. (2015) 46:65–8. doi: 10.1055/s-0034-1389898
72. Tian L, Li Y, Xue X, Wu M, Liu F, Hao X, et al. Super-refractory status epilepticus in West China. Acta Neurol Scand. (2015) 132:1–6. doi: 10.1111/ane.12336
73. Barros P, Brito H, Ferreira PC, Ramalheira J, Lopes J, Rangel R, et al. Resective surgery in the treatment of super-refractory partial status epilepticus secondary to NMDAR antibody encephalitis. Eur J Paediatr Neurol. (2014) 18:449–52. doi: 10.1016/j.ejpn.2014.01.013
74. Hainsworth JB, Shishido A, Theeler BJ, Carroll CG, Fasano RE. Treatment responsive GABA(B)-receptor limbic encephalitis presenting as new-onset super-refractory status epilepticus (NORSE) in a deployed U. S soldier Epileptic Disord. (2014) 16:486–93. doi: 10.1684/epd.2014.0702
75. Hilberath JM, Schmidt H, Wolf GK. Steroid-responsive encephalopathy associated with autoimmune thyroiditis (SREAT): case report of reversible coma and status epilepticus in an adolescent patient and review of the literature. Eur J Pediatr. (2014) 173:1263–73. doi: 10.1007/s00431-014-2391-6
76. Marques IB, Teotónio R, Cunha C, Bento C, Sales F. Anti-NMDA receptor encephalitis presenting with total insomnia—a case report. J Neurol Sci. (2014) 336:276–80. doi: 10.1016/j.jns.2013.10.034
77. Matsuzono K, Kurata T, Deguchi S, Yamashita T, Deguchi K, Abe K. Ketogenic diet therapy is effective in encephalitis with refractory seizures. Neurol Res. (2014) 36:906–10. doi: 10.1179/1743132814Y.0000000371
78. Singh RK, Joshi SM, Potter DM, Leber SM, Carlson MD, Shellhaas RA. Cognitive outcomes in febrile infection-related epilepsy syndrome treated with the ketogenic diet. Pediatrics. (2014) 134:e1431–5. doi: 10.1542/peds.2013-3106
79. Ueda R, Saito Y, Ohno K, Maruta K, Matsunami K, Saiki Y, et al. Effect of levetiracetam in acute encephalitis with refractory, repetitive partial seizures during acute and chronic phase. Brain Dev. (2015) 37:471–7. doi: 10.1016/j.braindev.2014.08.003
80. Finné Lenoir X, Sindic C, Van Pesch V, El Sankari S, De Tourtchaninoff M, Denays R, et al. Anti-N-methyl-D-aspartate receptor encephalitis with favorable outcome despite prolonged status epilepticus. Neurocrit Care. (2013) 18:89–92. doi: 10.1007/s12028-012-9788-8
81. Juhász C, Buth A, Chugani DC, Kupsky WJ, Chugani HT, Shah AK, et al. Successful surgical treatment of an inflammatory lesion associated with new-onset refractory status epilepticus. Neurosurg Focus. (2013) 34:E5. doi: 10.3171/2013.3.FOCUS1336
82. Kumar G, Mittal S, Moudgil SS, Kupsky WJ, Shah AK. Histopathological evidence that hippocampal atrophy following status epilepticus is a result of neuronal necrosis. J Neurol Sci. (2013) 334:186–91. doi: 10.1016/j.jns.2013.08.016
83. Puoti G, Elefante A, Saracino D, Capasso A, Cotrufo R, Belluomo Anello C. New-onset refractory status epilepticus mimicking herpes virus encephalitis. Case Rep Neurol. (2013) 5:162–7. doi: 10.1159/000355273
84. Verma R, Raut TP, Giri P, Praharaj HN. New onset refractory status epilepticus (NORSE) as the heralding manifestation of herpes simplex encephalitis. BMJ Case Rep. (2013). doi: 10.1136/bcr-2013-009466
85. Gordon Boyd J, Debicki DB, Young GB. Temporal lobe epilepsy after refractory status epilepticus: an illustrative case and review of the literature. Epilepsy Res Treat. (2012) 2012:209701. doi: 10.1155/2012/209701
86. Howell KB, Katanyuwong K, MacKay MT, Bailey CA, Scheffer IE, Freeman JL, et al. Long-term follow-up of febrile infection-related epilepsy syndrome. Epilepsia. (2012) 53:101–10. doi: 10.1111/j.1528-1167.2011.03350.x
87. Lin J, Lin K, Hsia S, Wang H. Therapeutic hypothermia for febrile infection-related epilepsy syndrome in two patients. Pediatr Neurol. (2012) 47:448–50. doi: 10.1016/j.pediatrneurol.2012.08.013
88. Lin J, Lin K, Hsia S, Wang H, Chou I, Lin Y. Antiglutamic acid decarboxylase antibodies in children with encephalitis and status epilepticus. Pediatr Neurol. (2012) 47:252–8. doi: 10.1016/j.pediatrneurol.2012.06.013
89. Okumura A, Komatsu M, Abe S, Kitamura T, Matsui K, Ikeno M, et al. Amplitude-integrated electroencephalography in patients with acute encephalopathy with refractory, repetitive partial seizures. Brain and Development. (2011) 33:77–82. doi: 10.1016/j.braindev.2009.12.009
90. Johnson N, Henry C, Fessler AJ, Dalmau J. Anti-NMDA receptor encephalitis causing prolonged nonconvulsive status epilepticus. Neurology. (2010) 75:1480–2. doi: 10.1212/WNL.0b013e3181f8831a
91. Lin J, Lin K, Hsia S, Wu C, Chou I, Wang H. Analysis of status epilepticus with mycoplasma pneumoniae encephalitis. Pediatr Neurol. (2010) 43:41–5. doi: 10.1016/j.pediatrneurol.2010.02.017
92. Nabbout R, Mazzuca M, Hubert P, Peudennier S, Allaire C, Flurin V, et al. Efficacy of ketogenic diet in severe refractory status epilepticus initiating fever induced refractory epileptic encephalopathy in school age children (FIRES). Epilepsia. (2010) 51:2033. doi: 10.1111/j.1528-1167.2010.02703.x
93. Sakuma H, Awaya Y, Shiomi M, Yamanouchi H, Takahashi Y, Saito Y, et al. Acute encephalitis with refractory, repetitive partial seizures (AERRPS): a peculiar form of childhood encephalitis. Acta Neurol Scand. (2010) 121:251–6. doi: 10.1111/j.1600-0404.2009.01198.x
94. Lin J, Lin K, Wang H, Hsia S, Wu C. Effect of topiramate, in combination with lidocaine, and phenobarbital, in acute encephalitis with refractory repetitive partial seizures. Brain Dev. (2009) 31:605–11. doi: 10.1016/j.braindev.2008.09.010
95. Lin J, Lin K, Wang H, Hsia S, Wu C. Analysis of status epilepticus related presumed encephalitis in children. Eur J Paediatr Neurol. (2008) 12:32–7. doi: 10.1016/j.ejpn.2007.05.007
96. Shyu C, Lee H, Chi C, Chen C. Acute encephalitis with refractory, repetitive partial seizures. Brain Dev. (2008) 30:356–61. doi: 10.1016/j.braindev.2007.10.008
97. Saito Y, Maegaki Y, Okamoto R, Ogura K, Togawa M, Nanba Y, et al. Acute encephalitis with refractory, repetitive partial seizures: case reports of this unusual post-encephalitic epilepsy. Brain Dev. (2007) 29:147–56. doi: 10.1016/j.braindev.2006.08.005
98. Kramer U, Shorer Z, Ben-Zeev B, Lerman-Sagie T, Goldberg-Stern H, Lahat E. Severe refractory status epilepticus owing to presumed encephalitis. J Child Neurol. (2005) 20:184–7. doi: 10.1177/08830738050200030301
99. Baxter P, Clarke A, Cross H, Harding B, Hicks E, Livingston J, et al. Idiopathic catastrophic epileptic encephalopathy presenting with acute onset intractable status. Seizure. (2003) 12:379–87. doi: 10.1016/S1059-1311(02)00340-0
100. Raspall-Chaure M, Chin RFM, Neville BG, Scott RC. Outcome of paediatric convulsive status epilepticus: a systematic review. Lancet Neurol. (2006) 5:769–79. doi: 10.1016/S1474-4422(06)70546-4
101. Sculier C, Gaínza-Lein M, Sánchez Fernández I, Loddenkemper T. Long-term outcomes of status epilepticus: a critical assessment. Epilepsia. (2018) 59:155–69. doi: 10.1111/epi.14515
102. DeLorenzo RJ, Hauser WA, Towne AR, Boggs JG, Pellock JM, Penberthy L, et al. A prospective, population-based epidemiologic study of status epilepticus in Richmond, Virginia. Neurology. (1996) 46:1029. doi: 10.1212/WNL.46.4.1029
103. Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. (2015) 4:1. doi: 10.1186/2046-4053-4-1
104. Taraschenko O, Pavuluri S, Schmidt CM, Pulluru Y, Gupta N. Seizure Burden Neuropsychological Outcomes of New-Onset Refractory Status Epilepticus (NORSE): A Systematic Review. (2022). Available online at: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=361142 (accessed January 7, 2023).
105. Murad MH, Sultan S, Haffar S, Bazerbachi F. Methodological quality and synthesis of case series and case reports. BMJ Evid Based Med. (2018) 23:60–3. doi: 10.1136/bmjebm-2017-110853
106. Clarkson BDS, LaFrance-Corey R, Kahoud RJ, Farias-Moeller R, Payne ET, Howe CL. Functional deficiency in endogenous interleukin-1 receptor antagonist in patients with febrile infection-related epilepsy syndrome. Ann Neurol. (2019) 85:526–37. doi: 10.1002/ana.25439
107. Dilena R, Mauri E, Aronica E, Bernasconi P, Bana C, Cappelletti C, et al. Therapeutic effect of Anakinra in the relapsing chronic phase of febrile infection–related epilepsy syndrome. Epilepsia Open. (2019) 4:344–50. doi: 10.1002/epi4.12317
108. Hsieh M, Lin J, Hsia S, Huang J, Yeh K, Chang K, et al. Diminished toll-like receptor response in febrile infection-related epilepsy syndrome (FIRES). Biomed J. (2020) 43:293–304. doi: 10.1016/j.bj.2020.05.007
109. Yue J, Wei YJ, Yang XL, Liu SY, Yang H. Zhang C. NLRP3 inflammasome and endoplasmic reticulum stress in the epileptogenic zone in temporal lobe epilepsy: molecular insights into their interdependence. Neuropathol Appl Neurobiol. (2020) 46:770–85. doi: 10.1111/nan.12621
110. Cristina de Brito Toscano E, Leandro Marciano Vieira É, Boni Rocha Dias B, Vidigal Caliari M, Paula Gonçalves A, Varela Giannetti A, et al. NLRP3 and NLRP1 inflammasomes are up-regulated in patients with mesial temporal lobe epilepsy and may contribute to overexpression of caspase-1 and IL-β in sclerotic hippocampi. Brain Res. (2021) 1752:147230. doi: 10.1016/j.brainres.2020.147230
111. Sakuma H, Tanuma N, Kuki I, Takahashi Y, Shiomi M, Hayashi M. Intrathecal overproduction of proinflammatory cytokines and chemokines in febrile infection-related refractory status epilepticus. J Neurol Neurosurg Psychiatr. (2015) 86:820. doi: 10.1136/jnnp-2014-309388
112. Jun J, Lee S, Kim R, Chu K, Lee SK. Tocilizumab treatment for new onset refractory status epilepticus. Ann Neurol. (2018) 84:940–5. doi: 10.1002/ana.25374
113. Kothur K, Bandodkar S, Wienholt L, Chu S, Pope A, Gill D, et al. Etiology is the key determinant of neuroinflammation in epilepsy: elevation of cerebrospinal fluid cytokines and chemokines in febrile infection-related epilepsy syndrome and febrile status epilepticus. Epilepsia. (2019) 60:1678–88. doi: 10.1111/epi.16275
114. Nabbout R, Vezzani A, Dulac O, Chiron C. Acute encephalopathy with inflammation-mediated status epilepticus; 21163447. Lancet Neurol. (2011) 10:99–108. doi: 10.1016/S1474-4422(10)70214-3
115. Van Baalen A, Häusler M, Boor R, Rohr A, Sperner J, Kurlemann G, et al. Febrile infection–related epilepsy syndrome (FIRES): a nonencephalitic encephalopathy in childhood. Epilepsia. (2010) 51:1323–8. doi: 10.1111/j.1528-1167.2010.02535.x
116. Boyd JG, Taylor S, Rossiter JP, Islam O, Spiller A, Brunet DG. New-onset refractory status epilepticus with restricted DWI and neuronophagia in the pulvinar. Neurology. (2010) 74:1003. doi: 10.1212/WNL.0b013e3181d5dc4f
117. Taraschenko O, Fox HS, Pittock SJ, Zekeridou A, Gafurova M, Eldridge E, et al. A mouse model of seizures in anti–N-methyl-d-aspartate receptor encephalitis. Epilepsia. (2019) 60:452–63. doi: 10.1111/epi.14662
118. Planagumà J, Leypoldt F, Mannara F, Gutiérrez-Cuesta J, Martín-García E, Aguilar E, et al. Human N-methyl D-aspartate receptor antibodies alter memory and behaviour in mice. Brain. (2015) 138:94–109. doi: 10.1093/brain/awu310
119. Halliday A, Duncan A, Cheung M, Boston RC, Apiwattanakul M, Camacho X, et al. Second-line immunotherapy and functional outcomes in autoimmune encephalitis: a systematic review and individual patient data meta-analysis. Epilepsia. (2022) 63:2214–24. doi: 10.1111/epi.17327
120. Qin P, Xu H, Laursen TM, Vestergaard M, Mortensen PB. Risk for schizophrenia and schizophrenia-like psychosis among patients with epilepsy: population based cohort study. BMJ. (2005) 331:23. doi: 10.1136/bmj.38488.462037.8F
121. Akanuma N, Adachi N, Fenwick P, Ito M, Okazaki M, Hara K, et al. Individual vulnerabilities to psychosis after antiepileptic drug administration. BMJ Neurol Open. (2020) 2:e000036. doi: 10.1136/bmjno-2019-000036
Keywords: chronic NORSE, febrile infection epilepsy-related syndrome (FIRES), refractory seizures, encephalopathy, cognitive failure, mood disturbances, functional outcomes, seizure outcomes
Citation: Taraschenko O, Pavuluri S, Schmidt CM, Pulluru YR and Gupta N (2023) Seizure burden and neuropsychological outcomes of new-onset refractory status epilepticus: Systematic review. Front. Neurol. 14:1095061. doi: 10.3389/fneur.2023.1095061
Received: 10 November 2022; Accepted: 02 January 2023;
Published: 24 January 2023.
Edited by:
Aljoscha Thomschewski, Paracelsus Medical University, AustriaReviewed by:
Yvonne Höller, University of Akureyri, IcelandLunliya Thampratankul, Mahidol University, Thailand
Copyright © 2023 Taraschenko, Pavuluri, Schmidt, Pulluru and Gupta. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Olga Taraschenko,  olha.taraschenko@unmc.edu
olha.taraschenko@unmc.edu
 Olga Taraschenko
Olga Taraschenko Spriha Pavuluri1
Spriha Pavuluri1 Yashwanth Reddy Pulluru
Yashwanth Reddy Pulluru