Acute FPIES and DIES: is a G lacking?
- 1Department of Health Sciences, University of Florence, Florence, Italy
- 2Allergy Unit, Meyer Children’s Hospital IRCCS, Florence, Italy
Food-Protein Induced Enterocolitis Syndrome (FPIES) is a clinical entity that, in the last years, has become significantly more relevant; it has been the focus of an increasing number of publications in the scientific community. The first clinical reports suggestive of chronic FPIES are from 1960 to 1970, and they described the main presenting feature as protracted diarrhea in newborns (1–3).
In 1978 (4) diagnostic criteria for these children were proposed: (1) symptoms presenting at less than two months of age; (2) while receiving the causative formula, the infant has watery stools with mucus, blood, and leukocytes, and a peripheral polymorphonuclear leukocytosis; (3) diarrhea ceases and normal growth resumes when the offending antigen is eliminated; (4) the response to a challenge meets the criteria described.
Later, in 1986, these criteria were modified to incorporate acute symptoms like vomiting: (1) disappearance of the symptoms of vomiting and diarrhea and of diagnostic findings in the stool (blood and leucocytes) after all antigens are removed from the diet; (2) no other cause for the colitis is demonstrable; (3) symptoms do not recur, and weight gain is normal for one month on a low-antigen formula, such as breast milk or casein hydrolysate formula, as the only dietary source; (4) a challenge with milk or soy formula, or other offending food antigens, reproduces symptoms (5).
Sicherer, in 1998, included in the criteria of repetitive vomiting, the main symptom of acute FPIES in subjects aged younger than 9 months at initial diagnosis, and coined FPIES as a syndrome (6).
In 2013, Miceli Sopo (7) proposed to increase the age of presentation to less than two years and introduced a 2–4 hs latency between exposure to the incriminating food and elicitation of repetitive and important vomiting, pallor, hyporeactivity, and lethargy.
In 2015, Leonard divided the criteria for acute reactions into major and minor criteria and proposed that the criteria should not include an age limit for the age of onset. Repetitive vomiting or protracted diarrhea (1) was considered a major criterion, together with the absence of cutaneous and respiratory symptoms suggestive of an IgE-mediated allergy (2), removal of causative food results in resolution of symptoms (3), and re-exposure or a food challenge eliciting the typical symptoms (4). Minor criteria were added: hypotension (1), lethargy, pallor or hypotonia (2), negative skin-prick test and undetectable specific IgE level (3), absence of fever or hypothermia (<36°C) (4) (8).
In 2017, Nowak-Węgrzyn et al. (9) published the International Consensus Guidelines for the diagnosis and management of FPIES that, differently from previous authors, identified one major criterion and nine minor criteria, emphasizing the symptom of vomiting. The acute FPIES diagnosis is made if the major criterion and at least 3 minor criteria are met. Repetitive vomiting in the 1–4 h/s period after ingesting the suspected drug and the absence of classic IgE-mediated allergic skin or respiratory symptoms was considered the only major criterion. If only a single episode has occurred, a diagnostic Oral Food Challenge (OFC) should be strongly considered to confirm the diagnosis, especially because viral gastroenteritis is so common in this age group. Minor criteria were identified as: a second (or more) episode of repetitive vomiting after eating the same suspect food (1), repetitive vomiting episode 1–4 h/s after eating a different food (2), extreme lethargy with any suspected reaction (3), marked pallor with any suspected reaction (4), need for emergency department visit with any suspected reaction (5), need for intravenous fluid support with any suspected reaction (6), diarrhea in 24 hs (usually 5–10 hs) (7), hypotension (8), hypothermia (9).
In 2021, Vazquez-Ortiz and Infante proposed to add crampy abdominal pain, nausea (if vomiting is absent), and an increase in absolute neutrophil count >1,500/mm3 with normalization within 24 hs in the minor criteria to improve diagnostic accuracy (10).
Chronic FPIES is not as characterized as acute FPIES. It develops with regular/repeated ingestion of the triggering food (e.g., feeding an infant on cow's milk- or soy-based formula), presenting e.g., as chronic/intermittent emesis, watery diarrhea, and failure to thrive. Severe chronic FPIES can lead to dehydration and shock, potentially requiring bowel rest and intravenous fluids. However, for chronic FPIES, a clear set of criteria has not been defined yet. In 2015, Consensus Guidelines (9) defined the resolution of the symptoms within 3–10 days after the elimination of the offending food(s). Acute recurrence of symptoms when the food is reintroduced, the onset of vomiting in 1–4 h/s and diarrhea in 24 hs (usually lasting 5–10 hs) were reported as the most important criterion for chronic FPIES diagnosis. However, without confirmatory challenge, the diagnosis of chronic FPIES remains presumptive.
Hypoalbuminemia and poor weight gain can predict chronic cow’s milk induced FPIES in young infants with chronic gastrointestinal symptoms (9).
Vomiting is the most common symptom of pediatric FPIES in cohort studies (11, 12) meanwhile, in the adult form case series of FPIES, abdominal pain and diarrhea were more frequently reported, but vomiting is still often present (13) (Table 1).
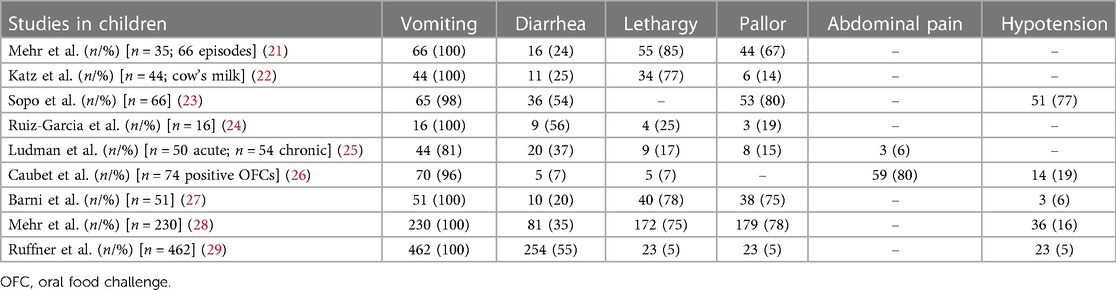
Table 1. Most common symptoms of food-protein induced enterocolitis syndrome in several published studies in children.
So far, there are no specific laboratory tests for the diagnosis of this disease, and only OFCs have shown to be useful in its diagnosis (14). Also, compared to IgE-mediated diseases, adrenaline does not represent the first line of treatment, not showing an improvement in patients’ symptoms or disease resolution. Ondansetron (a serotonin 5-HT3 receptor antagonist used to treat nausea and vomiting, often after chemotherapy) is reported as a successful treatment for vomiting, abdominal pain, and lethargy during FPIES (15, 16).
Gastric involvement is therefore considered essential for the diagnosis of a possible acute FPIES but has not been included in the acronym, which is why we think that the term Food-Protein Induced Gastro Enterocolitis Syndrome (FPIGES) may be considered by the scientific community.
In 2014, Novembre et al. first described an amoxicillin-induced adverse reaction with the same characteristics as FPIES, named drug-induced enterocolitis syndrome (DIES) (17).
In 2019, the diagnostic criteria for patients with possible DIES were proposed (18).
DIES shares with FPIES the clinical presentation of vomiting in the 1–4 h/s period after ingestion of the suspected drug and the absence of classic IgE-mediated allergic skin or respiratory symptoms as the major diagnostic criterion. Minor criteria were identified: a second episode of repetitive vomiting after ingestion of the same drug (1), repetitive vomiting episode 1–4 h/s after ingestion of a different drug (2), extreme lethargy (3), marked pallor (4), need for emergency department visit (5), need for intravenous fluid support (6), diarrhea in 24 hs (usually 5–10 hs) after ingested drug (7), hypotension (8), hypothermia (9). The diagnosis of DIES requires that a patient meet the major criteria and at least 3 minor criteria. If only a single episode has occurred, a diagnostic OFC should be strongly considered to confirm the diagnosis.
In the reported cases of DIES, vomiting in the 1–4 h/s period after ingestion of the suspected drug is the only symptom always present (19, 20). Gastric involvement is therefore considered crucial for the diagnosis of possible DIES but has not been included in the acronym, which is why we think that the term Drug-induced Gastro Enterocolitis Syndrome (DIGES) may be considered by the scientific community.
In conclusion, in our opinion, the terms FPIGES and DIGES, underlining the classic gastric involvement in FPIES and DIES, may be considered by the scientific community. This may lead to a better description of these clinical entities to correctly recognize these underdiagnosed syndromes and guide their treatment.
Author contributions
EN conceptualized the work. EN, MG, FC, GL, LS, SB and FM collected the data and drafted the manuscript. EN, MG, FC, GL, LS, SB and FM analyzed the data and critically revised the manuscript. All authors contributed to the article and approved the submitted version.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
2. Powell GK. Enterocolitis in low-birth-weight infants associated with milk and soy protein intolerance. J Pediatr. (1976) 88(5):840–4. doi: 10.1016/S0022-3476(76)81128-6
3. Ikola RA. Severe intestinal reaction following ingestion of rice. Arch Pediatr Adolesc Med. (1963) 105(3):281. doi: 10.1001/archpedi.1963.02080040283010
4. Powell GK. Milk- and soy-induced enterocolitis of infancy. J Pediatr. (1978) 93(4):553–60. doi: 10.1016/S0022-3476(78)80887-7
5. Powell GK. Food protein-induced enterocolitis of infancy: differential diagnosis and management. Compr Ther. (1986) 12(2):28–37.3956146
6. Sicherer SH, Eigenmann PA, Sampson HA. Clinical features of food protein–induced enterocolitis syndrome. J Pediatr. (1998) 133(2):214–9. doi: 10.1016/S0022-3476(98)70222-7
7. Miceli Sopo S, Greco M, Monaco S, Tripodi S, Calvani M. Food protein-induced enterocolitis syndrome, from practice to theory. Expert Rev Clin Immunol. (2013) 9(8):707–15. doi: 10.1586/1744666X.2013.814418
8. Leonard SA, Nowak-Węgrzyn A. Food protein–induced enterocolitis syndrome. Pediatr Clin North Am. (2015) 62(6):1463–77. doi: 10.1016/j.pcl.2015.07.011
9. Nowak-Węgrzyn A, Chehade M, Groetch ME, Spergel JM, Wood RA, Allen K, et al. International consensus guidelines for the diagnosis and management of food protein–induced enterocolitis syndrome: executive summary—workgroup report of the adverse reactions to foods committee, American academy of allergy, asthma & immunology. J Allergy Clin Immunol. (2017) 139(4):1111–1126.e4. doi: 10.1016/j.jaci.2016.12.966
10. Vazquez-Ortiz M, Infante S. Diagnostic criteria for food protein–induced enterocolitis syndrome. Ann Allergy Asthma Immunol. (2021) 126(5):458–9. doi: 10.1016/j.anai.2021.01.031
11. Wong S, Duan L, Galper A, Atkinson A, Upton J, Eiwegger T. Food protein-induced enterocolitis syndrome in a tertiary pediatric center: safety of guideline-conforming food challenges. Allergy Asthma Clin Immunol. (2022) 18(1):54. doi: 10.1186/s13223-022-00694-y
12. Stiefel G, Alviani C, Afzal NA, Byrne A, du Toit G, DunnGalvin A, et al. Food protein-induced enterocolitis syndrome in the British Isles. Arch Dis Child. (2022) 107(2):123–7. doi: 10.1136/archdischild-2020-320924
13. González-Delgado P, Muriel J, Jiménez T, Cameo JI, Palazón-Bru A, Fernández J. Food protein–induced enterocolitis syndrome in adulthood: clinical characteristics, prognosis, and risk factors. J Allergy Clin Immunol Pract. (2022) 10(9):2397–403. doi: 10.1016/j.jaip.2022.05.006
14. Barni S, Vazquez-Ortiz M, Giovannini M, Liccioli G, Sarti L, Cianferoni A, Mori F. Diagnosing food protein-induced enterocolitis syndrome. Clin Exp Allergy. (2021) 51(1):14–28. doi: 10.1111/cea.13767
15. Miceli Sopo S, Battista A, Greco M, Monaco S. Ondansetron for food protein-induced enterocolitis syndrome. Int Arch Allergy Immunol. (2014) 164(2):137–9. doi: 10.1159/000363384
16. Holbrook T, Keet CA, Frischmeyer-Guerrerio PA, Wood RA. Use of ondansetron for food protein–induced enterocolitis syndrome. J Allergy Clin Immunol. (2013) 132(5):1219–20. doi: 10.1016/j.jaci.2013.06.021
17. Novembre E, Mori F, Barni S, Pucci N. Drug-induced enterocolitis syndrome (DIES). Pediatr Allergy Immunol. (2014) 25(4):415–6. doi: 10.1111/pai.12225
18. van Thuijl AOJ, Landzaat LJ, Liem O, Emons JAM, Arends NJT. Drug-induced enterocolitis syndrome (DIES). Ann Allergy Asthma Immunol. (2019) 122(5):538–9. doi: 10.1016/j.anai.2019.02.004
19. Mori F, Liccioli G, Fuchs O, Barni S, Giovannini M, Sarti L, Novembre E, Caubet JC. Drug-induced enterocolitis syndrome: similarities and differences compared with food protein-induced enterocolitis syndrome. Pediatr Allergy Immunol. (2021) 32(6):1165–72. doi: 10.1111/pai.13491
20. Eyraud C, Biermé P, Adam M, Braun C. Drug-induced enterocolitis syndrome: a rare, severe, non-IgE-mediated immediate drug allergy. Case report and literature review. Arch Pediatr. (2023) 30(1):67–70. doi: 10.1016/j.arcped.2022.11.007
21. Mehr S, Kakakios A, Frith K, et al.. Food protein-induced enterocolitis syndrome: 16-year experience. Pediatrics. (2009) 123:e459-64.19188266
22. Katz Y, Goldberg MR, Rajuan N, et al.. The prevalence and natural course of food protein-induced enterocolitis syndrome to cow's milk: a large-scale, prospective population-based study. J Allergy Clin Immunol. (2011) 127:647-53.e1-3.21377033
23. Sopo SM, Giorgio V, Dello Iacono I, et al.. A multicentre retrospective study of 66 Italian children with food protein-induced enterocolitis syndrome: different management for different phenotypes. Clin Exp Allergy. (2012) 42:1257-65.22805473
24. Ruiz-García M, Díez CE, García SS, et al.. Diagnosis and natural history of food protein-induced enterocolitis syndrome in children from a tertiary hospital in central Spain. J Investig Allergol Clin Immunol. (2014) 24:354-6.
25. Ludman S, Harmon M, Whiting D, et al.. Clinical presentation and referral characteristics of food protein-induced enterocolitis syndrome in the United Kingdom. Ann Allergy Asthma Immunol. (2014) 113:290-4.25065570
26. Caubet JC, Ford LS, Sickles L, et al.. Clinical features and resolution of food protein-induced enterocolitis syndrome: 10-year experience. J Allergy Clin Immunol. (2014) 134:382-9.24880634
27. Barni S, Sarti L, Mori F, Liotti L, Pucci N, Novembre EA modified oral food challenge in children with food protein-induced enterocolitis syndrome. Clin Exp Allergy. (2019) 49(12):1633-6. doi: 10.1111/cea.13477. Epub 2019 Sep 13. PMID: 31386243.31386243
28. Mehr S, Frith K, Barnes EH, Campbell DE FPIES Study Group. Food protein-induced enterocolitis syndrome in Australia: A population-based study, 2012-2014. J Allergy Clin Immunol. (2017) 140(5):1323-30. doi: 10.1016/j.jaci.2017.03.027. Epub 2017 Apr 18. PMID: 28427879.28427879
29. Ruffner MA, Ruymann K, Barni S, Cianferoni A, Brown-Whitehorn T, Spergel JM. Food protein-induced enterocolitis syndrome: insights from review of a large referral population. J Allergy Clin Immunol Pract. (2013) 1(4):343-9. doi: 10.1016/j.jaip.2013.05.011. Epub 2013 Jun 28. PMID: 24565539.24565539
Keywords: definition, FPIES, rare allergic diseases, DIES, pediatrics
Citation: Novembre E, Giovannini M, Catamerò F, Liccioli G, Sarti L, Barni S and Mori F (2023) Acute FPIES and DIES: is a G lacking?. Front. Pediatr. 11:1185196. doi: 10.3389/fped.2023.1185196
Received: 13 March 2023; Accepted: 8 May 2023;
Published: 20 June 2023.
Edited by:
Marzia Duse, Sapienza University of Rome, ItalyReviewed by:
Maurizio Mennini, Bambino Gesù Children’s Hospital (IRCCS), Italy© 2023 Novembre, Giovannini, Catamerò, Liccioli, Sarti, Barni and Mori. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Francesco Catamerò francesco.catamero@unifi.it
†These authors share last authorship
‡ORCID Mattia Giovannini orcid.org/0000-0001-9568-6882 Francesco Catamerò orcid.org/0009-0001-5633-9025 Giulia Liccioli orcid.org/0000-0002-5216-0423 Lucrezia Sarti orcid.org/0000-0001-8055-3788 Simona Barni orcid.org/0000-0001-5598-2740 Francesca Mori orcid.org/0000-0001-7483-0128
 Elio Novembre
Elio Novembre Mattia Giovannini
Mattia Giovannini Francesco Catamerò
Francesco Catamerò Giulia Liccioli
Giulia Liccioli Lucrezia Sarti2,‡
Lucrezia Sarti2,‡  Simona Barni
Simona Barni Francesca Mori
Francesca Mori