Abstract
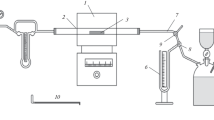
In lean coal and lignite, sulfur is predominantly present as organic compounds. According to experiments with a QMC-230 mass spectrometer and an IR absorption analyzer, the removal of sulfur in the reduction of iron-ore concentrates is hindered by the adsorption of sulfur-bearing gases and its absorption by the reduced iron to form FeS, which is not reduced by carbon or hydrogen. Considerable sulfur removal is only observed within a calcium-oxide overburden, by the reaction CaO + H2S = H2O + CaS.
Similar content being viewed by others
References
Amdur, A.M., Potapov, A.M., Raznitsina, A.L., and Munkhtuul, L., Kinetics of the Reduction of Iron-Ore Concentrates by Coal, Izv. Vyssh. Uchebn. Zaved., Chern. Metall., 2012, no. 8, pp. 17–19.
Kozhevnikov, I.Yu., Beskoksovaya metallurgiya zheleza (Coke-Free Metallurgy of Iron), Moscow: Metallurgiya, 1970.
Nikol’skii, B.P., Grigorov, O.N., and Pozin, M.E., Spravochnik khimiya (Handbook of Chemistry), Leningrad-Moscow: Khimiya, 1966.
Gagarin, S.G., Forms of Sulfur and Nitrogen in Coal’s Organic Mass, Koks Khim., 2003, no. 7, pp. 31–38.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.M. Amdur, D.V. Blagin, V.V. Pavlov, L. Munkhtuul, 2013, published in Koks i Khimiya, 2013, No. 3, pp. 2–6.
About this article
Cite this article
Amdur, A.M., Blagin, D.V., Pavlov, V.V. et al. Removal of sulfur in the reduction of iron-ore concentrates by coal. Coke Chem. 56, 81–84 (2013). https://doi.org/10.3103/S1068364X13030034
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068364X13030034