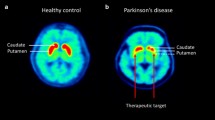
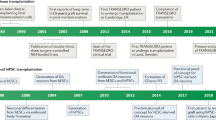
Abstract
Traditionally neural transplantation has had as its central tenet the replacement of missing neurons that have been lost because of neurodegenerative processes, as exemplified by diseases such as Parkinson disease (PD). However, the effectiveness and widespread application of this approach clinically has been limited, primarily because of the poor donor supply of human fetal neural tissue and the incomplete neurobiological understanding of the circuit reconstruction required to normalize function in these diseases. So, in PD the progress from promising neural transplantation in animal models to proof-of-principle, open-labeled clinical transplants, to randomized, placebo-controlled studies of neural transplantation has not been straightforward. The emergence of previously undescribed adverse effects and lack of significant functional advantage in recent clinical studies has been disappointing and has served to cast a new, and perhaps more realistic, perspective on this treatment approach. In fact, there have been calls by some involved in neural transplantation to return to the drawing board before pressing on with further clinical trials, and the return to basic experimentation. This therefore precipitates the question — is there a future for neural transplantation?
It is important to remember that there are a number of possible explanations for the disappointing results from the recent clinical trials in PD, ranging from the mode of transplantation to patient selection. Nevertheless, almost irrespective of these reasons for the current trial results, there have always been significant practical and ethical problems with using human fetal tissue, and so a number of alternative cell sources have been investigated. These alternative sources include stem cells, which are attractive for cell-based therapies because of their potential ease of isolation, propagation and manipulation, and their ability in some cases to migrate to areas of pathology and differentiate into specific and appropriate cell types. Furthermore, the availability of stem cells derived from non-embryonic sources (e.g. adult stem cells derived from the sub-ventricular zone) has removed some of the ethical limitations associated with the use of embryonic human tissue. These potentially beneficial aspects of stem cells means that there is a future for neural transplantation as a means of treating patients with a range of neurological disorders, although whether this will ever translate into a truly effective, widely available therapy remains unknown.




Similar content being viewed by others
References
Bucy PC. Cortical extirpation in the treatment of involuntary movements. Arch Neurol Psychiatr 1942; 21: 257–63
Putman TJ. Treatment of unilateral paralysis agitans by section of the lateral pyrmidal tract. Arch Neurol Psych 1940; 44: 950–76
Walker AE. Cerebral pedunculotomy for the relief of involuntary movements. J Nerv Ment Dis 1952; 116(6): 766–75
Cooper IS. The neurosurgical alleviation of parkinsonism. Am Surg 1956; 22(11): 1070–5
Hassler RRT. Indikationen und localisations metode der gezielten Hirnoperationen. Nevenartz 1954; 25(11): 441–7
Cooper IS. Surgical treatment of parkinsonism. Annu Rev Med 1965; 16: 309–30
Deep-Brain Stimulation for Parkinson’s Disease Study Group. Deep-brain stimulation of the subthalamic nucleus or the pars interna of the globus pallidus in Parkinson’s disease. N Engl J Med 2001; 345: 956–63
Hurelbrink CB, Barker RA. The potential of GDNF as a treatment for Parkinson’s disease. Exp Neurol 2004; 185: 1–6
Nutt JG, Burchiel KJ, Cornelia CL, et al. Randomized, double-blind trial of glial cell line-derived neurotrophic factor (GDNF) in PD. Neurology 2003; 60: 69–73
Gill SS, Patel NK, Hotton GR, et al. Direct brain infusion of glial cell line-derived neurotrophic factor in Parkinson disease. Nat Med 2003; 9: 589–95
Perlow MJ, Freed WJ, Hoffer BJ, et al. Brain grafts reduce motor abnormalities produced by destruction of nigrostriatal dopamine system. Science 1979; 204: 643–7
Dunnett SB, Björklund A, Stenevi U, et al. Behavioral recovery following transplantation of substantia nigra in rats subjected to 6-OHDA lesions of the nigrostriatal pathway: 2. Bilateral lesions. Brain Res 1981; 229: 457–70
Kordower JH, Freeman TB, Snow BJ, et al. Neuropathological evidence of graft survival and striatal reinnervation after the transplantation of fetal mesencephalic tissue in a patient with Parkinson’s disease. N Engl J Med 1995; 332: 1118–24
Bjorklund A, Stenevi U. Reconstruction of the nigrostriatal dopamine pathway by intracerebral nigral transplants. Brain Res 1979; 177: 555–60
Piccini P, Lindvall O, Bjorklund A, et al. Delayed recovery of movement-related cortical function in Parkinson’s disease after striatal dopaminergic grafts. Ann Neurol 2000; 48: 689–95
Piccini P, Brooks DJ, Bjorklund A, et al. Dopamine release from nigral transplants visualized in vivo in a Parkinson’s patient. Nat Neurosci 1999; 2: 1137–40
Brundin P, Pogarell O, Hagell P, et al. Bilateral caudate and putamen grafts of embryonic mesencephalic tissue treated with lazaroids in Parkinson’s disease. Brain 2000; 123: 1380–90
Widner H, Tetrad J, Rehncrona S, et al. Bilateral fetal mesencephalic grafting in two patients with parkinsonism induced by 1-me-thyl-4 -phenyl-l, 2,3,6-tetrahydropyridine (MPTP). N Engl J Med 1992; 327: 1556–63
Hauser RA, Freeman TB, Snow BJ, et al. Long-term evaluation of bilateral fetal nigral transplantation in Parkinson disease. Arch Neurol 1999; 56: 179–87
Hagell P, Schräg A, Piccini P, et al. Sequential bilateral transplantation in Parkinson’s disease; effects of the second graft. Brain 1999; 122: 1121–32
Isacson O, Bjorklund L, Pemaute RS. Parkinson’s disease: interpretations of transplantation study are erroneous. Nat Neurosci 2001; 4: 553
Freed CR, Greene PE, Breeze RE, et al. Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N Engl J Med 2001; 344: 710–9
Isacson O. The production and use of cells as therapeutic agents in neurodegenerative diseases. Lancet Neurol 2003; 2: 417–24
Olanow CW, Goetz CG, Kordower JH, et al. A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson’s disease. Ann Neurol 2003; 54: 403–14
Hurelbrink CB, Armstrong RJ, Barker RA, et al. Hibernated human fetal striatal tissue: successful transplantation in a rat model of Huntington’s disease. Cell Transplant 2000; 9: 743–9
Whone AL, Moore RY, Piccini PP, et al. Plasticity of the nigropallidal pathway in Parkinson’s disease. Ann Neurol 2003; 53: 206–13
Olanow CW. Transplantation for Parkinson’s disease: Pros, cons, and where do we go from here? Mov Disord 2002; 17: S15
Kirik D, Annett LE, Burger C, et al. Nigrostriatal alpha-synucleinopathy induced by viral vector-mediated overexpression of human alpha-synuclein: a new primate model of Parkinson’s disease. Proc Natl Acad Sci U S A 2003; 100: 2884–9
Phelps CJ, Koike C, Vaught TD, et al. Production of alpha 1,3-galactosyltransferase-deficient pigs. Science 2003; 299: 411–4
Cozzi E, Tucker AW, Langford GA, et al. Characterization of pigs transgenic for human decay-accelerating factor. Transplantation 1997; 64: 1383–92
Zhou C, Mclnnes E, Parsons N, et al. Production and characterisation of a pig line transgenic for human membrane cofactor protein. Xenotransplantation 2002; 9: 183–90
Lai L, Kolber-Simonds D, Park KW, et al. Production of alpha-l, 3-galactosyl-transferase knockout pigs by nuclear transfer cloning. Science 2002; 295: 1089–92
Hinchliffe SJ, Rushmere NK, Hanna SM, et al. Molecular cloning and functional characterization of the pig analogue of CD59: relevance to xenotransplantation. J Immunol 1998; 160: 3924–32
Freeman TB, Wojak JC, Brandeis L, et al. Cross-species intracerebral grafting of embryonic swine dopaminergic neurons. Prog Brain Res 1988; 78: 473–7
Armstrong RJ, Hurelbrink CB, Tyers P, et al. The potential for circuit reconstruction by expanded neural precursor cells explored through porcine xenografts in a rat model of Parkinson’s disease. Exp Neurol 2002; 175: 98–111
Winkler C, Kirik D, Bjorklund A, et al. Transplantation in the rat model of Parkinson’s disease: ectopic versus homotopic graft placement. Prog Brain Res 2000; 127: 233–65
Wictorin K, Bjorklund A. Axon outgrowth from grafts of human embryonic spinal cord in the lesioned adult rat spinal cord. Neuroreport 1992; 3: 1045–8
Wictorin K, Brundin P, Sauer H, et al. Long distance directed axonal growth from human dopaminergic mesencephalic neuroblasts implanted along the nigrostriatal pathway in 6-hydroxydopamine lesioned adult rats. J Comp Neurol 1992; 323: 475–94
Isacson O, Deacon TW, Pakzaban P, et al. Transplanted xenogeneic neural cells in neurodegenerative disease models exhibit remarkable axonal target specificity and distinct growth patterns of glial and axonal fibres. Nat Med 1995; 1: 1189–94
Huffaker TK, Boss BD, Morgan AS, et al. Xenografting of fetal pig ventral mesencephalon corrects motor asymmetry in the rat model of Parkinson’s disease. Exp Brain Res 1989; 77: 329–36
Galpern WR, Bums LH, Deacon TW, et al. Xenotransplantation of porcine fetal ventral mesencephalon in a rat model of Parkinson’s disease: functional recovery and graft morphology. Exp Neurol 1996; 140: 1–13
Wictorin K, Brundin P, Gustavii B, et al. Reformation of long axon pathways in adult rat central nervous system by human forebrain neuroblasts. Nature 1990; 347: 556–8
Deacon TW, Pakzaban P, Burns LH, et al. Cytoarchitectonic development, axonglia relationships, and long distance axon growth of porcine striatal xenografts in rats. Exp Neurol 1994; 130: 151–67
Garcia AR, Deacon TW, Dinsmore J, et al. Extensive axonal and glial fiber growth from fetal porcine cortical xenografts in the adult rat cortex. Cell Transplant 1995; 4: 515–27
Hurelbrink CB, Armstrong RJ, Dunnett SB, et al. Neural cells from primary human striatal xenografts migrate extensively in the adult rat CNS. Eur J Neurosci 2002; 15: 1255–66
Fink JS, Schumacher JM, Ellias SL, et al. Porcine xenografts in Parkinson’s disease and Huntington’s disease patients: preliminary results. Cell Transplant 2000 Mar–Apr; 9(2): 273–8
Schumacher JM, Ellias SA, Palmer EP, et al. Transplantation of embryonic porcine mesencephalic tissue in patients with PD. Neurology 2000 Mar 14; 54(5): 1042–50
Zhang SC, Wernig M, Duncan ID, et al. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol 2001; 19: 1129–33
Bjorklund LM, Sanchez-Pernaute R, Chung S, et al. Embryonic stem cells develop into functional dopaminergic neurons after transplantation in a Parkinson rat model. Proc Natl Acad Sci U S A 2002; 99: 2344–9
Freed CR. Will embryonic stem cells be a useful source of dopamine neurons for transplant into patients with Parkinson’s disease? Proc Natl Acad Sci U S A 2002; 99: 1755–7
Kim JH, Auerbach JM, Rodriguez-Gomez JA, et al. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson’s disease. Nature 2002; 418: 50–6
Reynolds BA, Weiss S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 1992; 255: 1707–10
Armstrong RJ, Harrower TP, Hurelbrink CB, et al. Porcine neural xenografts in the immunocompetent rat; immune response following grafting of expanded neural precursor cells. Neuroscience 2001; 106: 201–16
Svendsen CN, ter Borg MG, Armstrong RJE, et al. A new method for the rapid and long term growth of human neural precursor cells. J Neurosci Methods 1998; 85: 141–53
Svendsen CN, Fawcett JW, Bentlage C, et al. Increased survival of rat EGF-generated CNS progenitor cells using B27 supplemented medium. Exp Brain Res 1995; 102: 407–14
Ahmed S, Reynolds BA, Weiss S. BDNF enhances the differentiation but not the survival of CNS stem cell-derived neuronal precursors. J Neurosci 1995; 15: 5765–78
Arsenijevic Y, Weiss S. Insulin-like growth factor-I is a differentiation factor for postmitotic CNS stem cell-derived neuronal precursors: distinct actions from those of brain-derived neurotrophic factor. J Neurosci 1998; 18: 2118–28
Studer L, Tabar V, McKay RDG. Transplantation of expanded mesencephalic precursors leads to recovery in parkinsonion rats. Nat Neurosci 1998; 1: 290–5
Doetsch F, Alvarez-Buylla A. Network of tangential pathways for neuronal migration in adult mammalian brain. Proc Natl Acad Sci U S A 1996; 93: 14895–900
Lois C, Alvarez-Buylla A. Proliferating subventricular zone cells in the adult mammalian forebrain can differentiate into neurons and glia. Proc Natl Acad Sci U S A 1993; 90: 2074–7
Lois C, Alvarez-Buylla A. Long-distance neuronal migration in the adult mammalian brain. Science 1994; 264: 1145–8
Altman J. Autoradiographic and histological studies of postnatal neurogenesis: IV. Cell proliferation and migration in the anterior forebrain, with special reference to persisting neurogenesis in the olfactory bulb. J Comp Neurol 1969; 137: 433–57
Pencea V, Bingaman KD, Freedman LJ, et al. Neurogenesis in the subventricular zone and rostral migratory stream of the neonatal and adult primate forebrain. Exp Neurol 2001; 172: 1–16
Kornack DR, Rakic P. The generation, migration, and differentiation of olfactory neurons in the adult primate brain. Proc Natl Acad Sci U S A 2001; 98: 4752–7
Palmer TD, Takahashi J, Gage FH. The adult rat hippocampus contains primordial neural stem cells. Mol Cell Neurosci 1997; 8: 389–404
Suhonen JO, Peterson DA, Ray J, et al. Differentiation of adult hippocampus-derived progenitors into olfactory neurons in vivo. Nature 1996; 383: 624–7
Song H, Stevens CF, Gage FH. Astroglia induce neurogenesis from adult neural stem cells. Nature 2002; 417: 39–44
Eriksson PS, Perfilieva E, Bjork-Eriksson T, et al. Neurogenesis in the adult human hippocampus. Nat Med 1998; 4: 1313–7
Armstrong RJ, Barker RA. Neurodegeneration; a failure of neuroregeneration? Lancet 2001; 358: 1174–6
Levesque MF, Neuman T. Autologous transplantation of adult human neural stem cells and differentiated dopaminergic neurons for Parkinson’s disease; long term post-operative clinical and functional metabolic results [abstract]. Proceedings of the 8th International Meeting on Neural Transplantation and Repair. Exp Neurol 2002; 175: 7.1
Nunes MC, Roy NS, Keyoung HM, et al. Identification and isolation of multi-potential neural progenitor cells from the subcortical white matter of the adult human brain. Nat Med 2003; 9: 439–47
Zhao M, Momma S, Delfani K, et al. Evidence for neurogenesis in the adult mammalian substantia nigra. Proc Natl Acad Sci U S A 2003; 100: 7925–30
Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 2002; 418: 41–9
Ying QL, Nichols J, Evans EP, et al. Changing potency by spontaneous fusion. Nature 2002; 416: 545–8
Terada N, Hamazaki T, Oka M, et al. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 2002; 416: 542–5
Shults CW. Coenzyme Q(10)in neurodegenerative diseases. Curr Med Chem 2003; 10: 1917–21
Schilling G, Coonfield ML, Ross CA, et al. Coenzyme Q10 and remacemide hydrochloride ameliorate motor deficits in a Huntington’s disease transgenic mouse model. Neurosci Lett 2001; 315: 149–53
Huntington Study Group. A randomized, placebo-controlled trial of coenzyme Q10 and remacemide in Huntington’s disease. Neurology 2001; 57: 397–404
Kopyov OV, Jacques S, Lieberman A, et al. Safety of intrastriatal neurotransplantation for Huntington’s disease patients. Exp Neurol 1998; 149: 97–108
Hauser RA, Furtado S, Cimino CR, et al. Bilateral human fetal striatal transplantation in Huntington’s disease. Neurology 2002; 58: 687–95
Rosser AE, Barker RA, Harrower T, et al. Unilateral transplantation of human primary fetal tissue in four patients with Huntington’s disease: NEST-UK safety report ISRCTN no 36485475. J Neurol Neurosurg Psychiatry 2002; 73: 678–85
Bachoud-Levi A, Bourdet C, Brugieres P, et al. Safety and tolerability assessment of intrastriatal neural allografts in five patients with Huntington’s disease. Exp Neurol 2000; 161: 194–202
Freeman TB, Cicchetti F, Hauser RA, et al. Transplanted fetal striatum in Huntington’s disease: phenotypic development and lack of pathology. Proc Natl Acad Sci U S A 2000; 97: 13877–82
Bachoud-Levi AC, Remy P, Nguyen JP, et al. Motor and cognitive improvements in patients with Huntington’s disease after neural transplantation. Lancet 2000; 356: 1975–9
Jessell TM. Neuronal specification in the spinal cord: inductive signals and transcriptional codes. Nat Rev Genet 2000; 1: 20–9
Wichterle H, Lieberam I, Porter JA, et al. Directed differentiation of embryonic stem cells into motor neurons. Cell 2002; 110: 385–97
Wu P, Tarasenko YI, Gu Y, et al. Region-specific generation of cholinergic neurons from fetal human neural stem cells grafted in adult rat. Nat Neurosci 2002; 5: 1271–8
Archer DR, Cuddon PA, Lipsitz D, et al. Myelination of the canine central nervous system by glial cell transplantation: a model for repair of human myelin disease. Nat Med 1997; 3: 54–9
Groves AK, Barnett SC, Franklin RJ, et al. Repair of demyelinated lesions by transplantation of purified O-2A progenitor cells. Nature 1993; 362: 453–5
Imaizumi T, Lankford KL, Burton WV, et al. Xenotransplantation of transgenic pig olfactory ensheathing cells promotes axonal regeneration in rat spinal cord. Nat Biotechnol 2000; 18: 949–53
Jefferson S, Jacques T, Kiernan BW, et al. Inhibition of oligodendrocyte precursor motility by oligodendrocyte processes: implications for transplantation-based approaches to multiple sclerosis. Mult Scler 1997; 3: 162–7
Franklin RJ, Blakemore WF. To what extent is oligodendrocyte progenitor migration a limiting factor in the remyelination of multiple sclerosis lesions? Mult Scler 1997; 3: 84–7
O’Leary MT, Blakemore WF. Oligodendrocyte precursors survive poorly and do not migrate following transplantation into the normal adult central nervous system. J Neurosci Res 1997; 48: 159–67
Pluchino S, Quattrini A, Brambilla E, et al. Injection of adult neurospheres induces recovery in a chronic model of multiple sclerosis. Nature 2003; 422: 688–94
Sugaya K, Brannen CL. Stem cell strategies for neuroreplacement therapy in Alzheimer’s disease. Med Hypotheses 2001; 57: 697–700
Winkler J, Thal LJ, Gage FH, et al. Cholinergic strategies for Alzheimer’s disease. J Mol Med 1998; 76: 555–67
Gage FH, Chen KS. Neural transplants: prospects for Alzheimer’s disease. Curr Opin Neurol Neurosurg 1992; 5: 94–9
Wenning GK, Tison F, Scherfler C, et al. Towards neurotransplantation in multiple system atrophy: clinical rationale, pathophysiological basis, and preliminary experimental evidence. Cell Transplant 2000; 9: 279–88
Waldner R, Puschban Z, Scherfler C, et al. No functional effects of embryonic neuronal grafts on motor deficits in a 3-nitropropionic acid rat model of advanced striatonigral degeneration (multiple system atrophy). Neuroscience 2001; 102: 581–92
Lu P, Jones LL, Snyder EY, et al. Neural stem cells constitutively secrete neurotrophic factors and promote extensive host axonal growth after spinal cord injury. Exp Neurol 2003; 181: 115–29
Sankar V, Muthusamy R. Role of human amniotic epithelial cell transplantation in spinal cord injury repair research. Neuroscience 2003; 118: 11–7
Harvey RL, Chopp M. The therapeutic effects of cellular therapy for functional recovery after brain injury. Phys Med Rehabil Clin N Am 2003; 14: S143–51
McDonald JW, Howard MJ. Repairing the damaged spinal cord: a summary of our early success with embryonic stem cell transplantation and remyelination. Prog Brain Res 2002; 137: 299–309
Hains BC, Johnson KM, Eaton MJ, et al. Serotonergic neural precursor cell grafts attenuate bilateral hyperexcitability of dorsal horn neurons after spinal hemisection in rat. Neuroscience 2003; 116: 1097–110
Riess P, Zhang C, Saatman KE, et al. Transplanted neural stem cells survive, differentiate, and improve neurological motor function after experimental traumatic brain injury. Neurosurgery 2002; 51: 1043–52
Ogawa Y, Sawamoto K, Miyata T, et al. Transplantation of in vitro-expanded fetal neural progenitor cells: results in neurogenesis and functional recovery after spinal cord contusion injury in adult rats. J Neurosci Res 2002; 69: 925–33
Jain S, Mathur R, Sharma R. Foetal amygdalar transplantation facilitates recovery of retention deficit in CeA lesioned rats. Indian J Exp Biol 2000; 38: 1014–9
Sinden JD, Rashid-Doubell F, Kershaw TR, et al. Recovery of spatial learning by grafts of a conditionally immortalized hippocampal neuroepithelial cell line into the ischaemia-lesioned hippocampus. Neuroscience 1997; 81: 599–608
Tarricone BJ, Simon JR, Li YJ, et al. Neural grafting of cholinergic neurons in the hippocampal formation. Behav Brain Res 1996; 74: 25–44
Patel SN, Clayton NS, Krebs JR. Hippocampal tissue transplants reverse lesion-induced spatial memory deficits in zebra finches (Taeniopygia guttata). J Neurosci 1997; 17: 3861–9
Li YJ, Simon JR, Low WC. Intrahippocampal grafts of cholinergic-rich striatal tissue ameliorate spatial memory deficits in rats with fornix lesions. Brain Res Bull 1992; 29: 147–55
Willing AE, Lixian J, Milliken M, et al. Intravenous versus intrastriatal cord blood administration in a rodent model of stroke. J Neurosci Res 2003; 73: 296–307
Chopp M, Li Y. Treatment of neural injury with marrow stromal cells. Lancet Neurol 2002; 1: 92–100
Savitz SI, Rosenbaum DM, Dinsmore JH, et al. Cell transplantation for stroke. Ann Neurol 2002; 52: 266–75
Savitz SL, Malhotra S, Gupta G, et al. Cell transplants offer promise for stroke recovery. J Cardiovasc Nurs 2003; 18: 57–61
Amar AP, Zlokovic BV, Apuzzo ML. Endovascular restorative neurosurgery: a novel concept for molecular and cellular therapy of the nervous system. Neurosurgery 2003; 52: 402–12
Modo M, Rezaie P, Heuschling P, et al. Transplantation of neural stem cells in a rat model of stroke: assessment of short-term graft survival and acute host immu-nological response. Brain Res 2002; 958: 70–82
Li Y, Chen J, Chen XG, et al. Human marrow stromal cell therapy for stroke in rat: neurotrophins and functional recovery. Neurology 2002; 59: 514–23
Nelson PT, Kondziolka D, Wechsler L, et al. Clonal human (hNT) neuron grafts for stroke therapy; neuropathology in a patient 27 months after implantation. Am J Pathol 2002; 160: 1201–6
Kondziolka D, Wechsler L, Achim C. Neural transplantation for stroke. J Clin Neurosci 2002; 9: 225–30
Kondziolka D, Wechsler L, Goldstein S, et al. Transplantation of cultured human neuronal cells for patients with stroke. Neurology 2000 Aug 22: 55(4); 565–9
Fukunaga A, Uchida K, Hara K, et al. Differentiation and angiogenesis of central nervous system stem cells implanted with mesenchyme into ischemic rat brain. Cell Transplant 1999; 8: 435–41
Grabowski M, Christofferson RH, Brundin P, et al. Vascularization of fetal neocortical grafts implanted in brain infarcts in spontaneously hypertensive rats. Neuroscience 1992; 51: 673–82
Loscher W, Ebert U, Lehmann H, et al. Seizure suppression in kindling epilepsy by grafts of fetal GABAergic neurons in rat substantia nigra. J Neurosci Res 1998; 51: 196–209
Barry DI, Wanscher B, Kragh J, et al. Grafts of fetal locus coeruleus neurons in rat amygdala-piriform cortex suppress seizure development in hippocampal kindling. Exp Neurol 1989; 106: 125–32
Fine A, Meldrum BS, Patel S. Modulation of experimentally induced epilepsy by intracerebral grafts of fetal GABAergic neurons. Neuropsychologia 1990; 28: 627–34
Bengzon J, Brundin P, Kalen P, et al. Host regulation of noradrenaline release from grafts of seizure-suppressant locus coeruleus neurons. Exp Neurol 1991; 111: 49–54
Holmes GL, Thompson JL, Huh K, et al. Effect of neural transplants on seizure frequency and kindling in immature rats following kainic acid. Brain Res Dev Brain Res 1991; 64: 47–56
Turner DA, Shetty AK. Clinical prospects for neural grafting therapy for hippocampal lesions and epilepsy. Neurosurgery 2003; 52: 632–44
Cejas PJ, Martinez M, Karmally S, et al. Lumbar transplant of neurons genetically modified to secrete brain-derived neurotrophic factor attenuates allodynia and hyperalgesia after sciatic nerve constriction. Pain 2000; 86: 195–210
Eaton MJ, Karmally S, Martinez MA, et al. Lumbar transplant of neurons genetically modified to secrete galanin reverse pain-like behaviors after partial sciatic nerve injury. J Peripher Nerv Syst 1999; 4: 245–57
Eaton MJ, Santiago DI, Dancausse HA, et al. Lumbar transplants of immortalized serotonergic neurons alleviate chronic neuropathic pain. Pain 1997; 72: 59–69
Eaton MJ, Plunkett JA, Martinez MA, et al. Transplants of neuronal cells bioengineered to synthesize GABA alleviate chronic neuropathic pain. Cell Transplant 1999; 8: 87–101
Pappas GD, Lazorthes Y, Bes JC, et al. Relief of intractable cancer pain by human chromaffin cell transplants: experience at two medical centers. Neurol Res 1997; 19: 71–7
Ehtesham M, Kabos P, Gutierrez MA, et al. Induction of glioblastoma apoptosis using neural stem cell-mediated delivery of tumor necrosis factor-related apoptosis-inducing ligand. Cancer Res 2002; 62: 7170–4
Ehtesham M, Kabos P, Kabosova A, et al. The use of interleukin 12-secreting neural stem cells for the treatment of intracranial glioma. Cancer Res 2002; 62: 5657–63
Noble M. Can neural stem cells be used to track down and destroy migratory brain tumor cells while also providing a means of repairing tumor-associated damage? Proc Natl Acad Sci U S A 2000; 97: 12393–5
Aboody KS, Brown A, Rainov NG, et al. Neural stem cells display extensive tropism for pathology in adult brain: evidence from intracranial gliomas. Proc Natl Acad Sci U S A 2000; 97: 12846–51
Park KI, Ourednik J, Ourednik V, et al. Global gene and cell replacement strategies via stem cells. Gene Ther 2002; 9: 613–24
Snyder EY, Taylor RM, Wolfe JH. Neural progenitor cell engraftment corrects lysosomal storage throughout the MPS VII mouse brain. Nature 1995; 374: 367–70
Lacorazza HD, Flax JD, Snyder EY, et al. Expression of human beta-hex-osaminidase alpha-subunit gene (the gene defect of Tay-Sachs disease) in mouse brains upon engraftment of transduced progenitor cells. Nat Med 1996; 2: 424–9
Acknowledgements
Dr Harrower was a Wellcome Clinical Training Fellow and was additionally supported by the Sackler Foundation, the D R Macintosh trust, and received a Brain Scholarship from the Guarantors of Brain. The Parkinson’s Disease Society (PDS) and the Medical Research Council of the United Kingdom supported some of our work cited in this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harrower, T.P., Barker, R.A. Is There a Future for Neural Transplantation?. BioDrugs 18, 141–153 (2004). https://doi.org/10.2165/00063030-200418030-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-200418030-00001