Abstract
Background
Docetaxel has shown promising single-agent activity in non-small cell lung cancer (NSCLC) and its activity can be enhanced by the addition of platinum compounds. Several studies indicate that carboplatin may be as effective as cisplatin but with better tolerability.
Objective
A phase II study was performed to investigate the safety and efficacy of combination chemotherapy with docetaxel and carboplatin in patients with advanced NSCLC.
Patients and Methods
30 chemotherapy-naïve patients with stage IIIB and IV NSCLC were treated with docetaxel 90 mg/m2 over 1 hour, followed by carboplatin administered according to a target area under the curve of 5 mg/ml/min (Calvert’s formula). Treatment was repeated every 3 weeks for 6 cycles.
Results
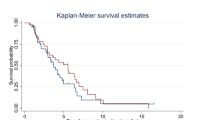
Myelosuppression was the predominant toxicity. Grade 3 or 4 granulocytopenia occurred in 77% of the patients. However, granulocyte colony-stimulating factor (G-CSF) was not utilised and neutropenic fever did not occur. Grade 3 or 4 nail disorder developed in 27% of the patients. Other non-haematological toxicities, including fluid retention, were mild to moderate. The objective response rate was 30% (two complete and seven partial responses). The median time to progression was 24 weeks and median survival 57 weeks. One-year survival was 56%.
Conclusions
The combination of docetaxel and carboplatin appears to be well tolerated and active in patients with advanced NSCLC.



Similar content being viewed by others
References
Non-Small Cell Lung Cancer Collaborative Group. Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. BMJ 1995; 311: 899–909
Cullen MH, Billingham LJ, Woodroffe CM, et al. Mitomycin, ifosfamide and cisplatin in unresectable non-small-cell lung cancer: effects on survival and quality of life. J Clin Oncol 1999; 17: 3188–94
Bunn PA Jr. Review of therapeutic trials of carboplatin in lung cancer. Semin Oncol 1989; 16: 27–33
Bunn PA Jr. The expanding role of cisplatin in the treatment of non-small cell lung cancer. Semin Oncol 1989; 16: 10–21
Ihde DC. Chemotherapy of lung cancer. N Engl J Med 1992; 327: 1434–41
Soquet PJ, Chauvin F, Boissel JP, et al. Polychemotherapy in advanced non-small cell lung cancer: a meta-analysis. Lancet 1993; 342: 19–21
Marino P, Pampallona S, Preatoni A, et al. Chemotherapy versus supportive care in advanced non-small cell lung cancer: results of a meta-analysis of the literature. Chest 1994; 106: 861–5
Sørensen BT, Strömgren A, Jakobsen P, et al. Dose-toxicity relationship of carboplatin in combination with cyclophos-phamide in ovarian cancer patients. Cancer Chemother Pharmacol 1991; 28: 39–401
Bonomi PD, Finkelstein DM, Ruckdeschel JC, et al. Combination chemotherapy versus single agents followed by combination chemotherapy in stage IV non-small cell lung cancer. A study of the Eastern Cooperative Oncology Group. J Clin Oncol 1989; 7: 1603–13
Kreisman H, Ginsberg S, Propert KJ, et al. Carboplatin or iproplarin in advanced non-small cell lung cancer: a Cancer and Leukemia Group B study. Cancer Treat Rep 1987; 71: 1049–52
Ringel I, Horwitz SB. Studies with RP 56976 (Taxotere): a semi-synthetic analog of Taxol. J Natl Cancer Inst 1991; 83: 288–91
Cerny T, Kaplan S, Pavlidis N, et al. Docetaxel (Taxotere) is active in non-small-cell lung cancer: a phase II trial of the EORTC Early Clinical Trials Group (ECTG). Br J Cancer 1994; 70: 384–7
Francis PA, Rigas JR, Kris MG, et al. Phase II trial of docetaxel in patients with stage III and IV non-small-cell lung cancer. J Clin Oncol 1994; 12: 1232–7
Miller VA, Rigas JR, Francis PA, et al. Phase II trial of a 75 mg/m2 dose of docetaxel with prednisone premedication for patients with advanced non-small cell lung cancer. Cancer 1995; 75: 968–72
Fossella FV, Lee JS, Murphy WK, et al. Phase II study of docetaxel for recurrent or metastatic non-small-cell lung cancer. J Clin Oncol 1994; 12: 1238–44
Burris H, Eckardt J, Fields S, et al. Phase II trials of Taxotere in patients with non small cell lung cancer [abstract no. 1116]. Proc Am Soc Clin Oncol 1993; 12: 335
Watanabe K, Yokoyama A, Furuse K, et al. Phase II trial of docetaxel in previously untreated non-small cell lung cancer [abstract no. 1095]. Proc Am Soc Clin Oncol 1994; 13
Fossella FV, Lee JS, Shin DM, et al. Phase II study of docetaxel for advanced or metastatic platinum-refractory non-small-cell lung cancer. J Clin Oncol 1995; 13: 645–51
Miller VA. Docetaxel in the management of advanced non-small cell lung cancer. Semin Oncol 1998; 25Suppl. 8: 15–9
Green MR, Kreisman H, Doll DC, et al. Carboplatin in non-small cell lung cancer: an update on the Cancer and Leukemia Group experience. Semin Oncol 1992; 19Suppl. 2: 44–7
Hill BT, Whelen RD, Shellard SA, et al. Differential cytotoxic effects of docetaxel in a range of mammalian tumour cell lines and certain drug resistant cell lines in vitro. Invest New Drugs 1994; 12: 169–82
Kelland LR, Abel G. Comparative cytotoxicity of Taxol and Taxotere against cisplatin-sensitive and resistant human ovarian carcinoma cell lines. Cancer Chemother Pharmacol 1995; 30: 444–50
Gandara DR, Voles E, Green M, et al. Activity of docetaxel in platinum-treated non-small-cell lung cancer: results of a phase II multicenter trial. J Clin Oncol 2000; 18: 131–5
Georgoulias V, Androulakis N, Dimopoulos AM, et al. First-line treatment of advanced non-small-cell lung cancer with docetaxel and cisplatin: a multicenter phase II study. Ann Oncol 1998; 9: 331–4
Le Chevalier T, Monnier A, Douillard JY, et al. Docetaxel (Taxotere) plus cisplatin: an active and well tolerated combination in patients with advanced non-small-cell lung cancer. Eur J Cancer 1998; 34: 2032–6
Zalcberg J, Millward M, Bishop J, et al. Phase II study of docetaxel and cisplatin in advanced non-small-cell lung cancer. J Clin Oncol 1998; 16: 1948–53
Lee FH, Cabetta R, Usackk BF, et al. New platinum complexes in clinical trials. Cancer Treat Rev 1983; 10: 43–5
Belani CP, Hadeed V, Ramanathan RK, et al. Docetaxel and carboplatin: a phase I and pharmacokinetic trial for advanced non-hematologic malignancies [abstract no. 771]. Proc Am Soc Clin Oncol 1997; 16: 220a
Belani CP, Einzig A, Bonomi P, et al. Multi-institutional phase II trial of docetaxel and carboplatin combination in patients with stage IIIB and IV non-small cell lung cancer [abstract no. 52]. Lung Cancer 1997; 18: 16
Capazzoli MJ, Belani CP, Einzig A, et al. Multi-institutional phase II trial of docetaxel and carboplatin combination in patients (pts) with stage IIIB and IV non-small cell lung cancer (NSCLC) [abstract no. 1845]. Proc Am Soc Clin Oncol 1998; 17: 479a
Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982; 5: 649–55
Calvert AH, Newell DR, Gumbrell LA, et al. Carboplatin dosage: Prospective evaluation of a simple formula based on renal function. J Clin Oncol 1989; 7: 1748–56
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31–41
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–81
Sandler A. First-line combination chemotherapy for advanced non-small cell lung cancer: The Eastern Cooperative Oncology Group and Southwest Oncology Group Experience. Semin Oncol 1999; 26Suppl. 15: 44–51
Belani CP, Bonomi P, Dobbs T, et al. Multicenter phase II trial of docetaxel and cisplatin combination in patients with non-small cell lung cancer [abstract no. 1660]. Proc Am Soc Clin Oncol 1997; 16
Faderl B, Pawel JV, Krauβ C, et al. Phase 2 study of docetaxel (D) and cisplatin (CISPT) in a circadian timing as first line chemotherapy (CT) in advanced non small cell lung cancer (NSCLC) [abstractno. 1856]. Proc Am Soc Clin Oncol 1998; 17
Cole JT, Gralla RJ, Rittenberg CN, et al. Defining the dose of docetaxel (Taxotere) in combination chemotherapy of non-small cell lung cancer: preserving efficacy with lower dose regimens [abstract no. 1671]. Proc Am Soc Clin Oncol 1997; 16
Gatzemeier U, Rosell R, Belticher D, et al. Randomized Pan-European trial comparing paclitaxel/carboplatin versus paclitaxel/cisplatin in advanced non-small cell lung cancer [abstract no. 973]. Abstracts and Proceedings of ECCO 10. Sept 12–16, 1999; Vienna, Austria
Langer CJ, Leighton JC, Comis RL, et al. Paclitaxel and carboplatin in combination in the treatment of advanced non-small-cell lung cancer: a phase II toxicity, response, and survival analysis. J Clin Oncol 1995; 13: 1860–70
McKeage MJ. Comparative adverse effect profiles of platinum drugs. Drug Safety 1995; 13: 228–44
Siddiqui N, Boddy AV, Thomas HD, et al. A clinical and pharmacokinetic study of the combination of carboplatin and paclitaxel for epithelial ovarian cancer. Br J Cancer 1997; 75: 287–94
Vaughn D, Mick R, Ratajczak J, et al. Investigating the platelet sparing mechanism of paclitaxel/carboplatin chemotherapy [abstract no. 897]. Proc Am Soc Clin Oncol 1997; 16
Acknowledgements
This study was supported in part by a grant from Aventis (formerly Rhône-Poulenc Rorer), Germany GmbH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schuette, W., Bork, I., Wollschläger, B. et al. Combination Chemotherapy with Docetaxel and Carboplatin for Advanced Non-Small Cell Lung Cancer. Clin. Drug Investig. 21, 161–168 (2001). https://doi.org/10.2165/00044011-200121030-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200121030-00001