Abstract
Background: Multiple sclerosis (MS) is a chronic neurological disease that affects 240 per 100 000 Canadians. Of these patients, 10–80% (average 70%) experience pain. Sativex® is a cannabis-based drug recently approved for neuropathic pain.
Objectives: In this study, we determine individuals’ preferences between two treatment options as well as the willingness to pay (WTP) for Sativex®, expressed as the amount they would pay in insurance premiums to have access to that treatment.
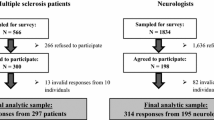
Methods: The WTP instrument comprised a decision board as a visual aid, and a questionnaire. A decision board helps clinicians standardize the presentation of treatment information. In this study, the decision board described two treatment options: a three-drug combination (gabapentin, amytriptyline, acetaminophen [paracetamol] {i.e. pills}) and the three-drug combination plus Sativex® (i.e. ‘pills and oral spray’). Information on efficacy and adverse effects was taken from trial data; wording was guided by a panel of neurologists and tested for clarity on lay people. The instrument was administered to 500 participants from Canada’s general population using the bidding game approach. Descriptive statistics were calculated.
Results: Mean (SD) age of participants was 39 (13) years, with a female: male distribution of 56: 44. The decision board was presented in both English (85%) and French (15%). Of 500 interviewees, 253 (50.6%) chose the ‘pills and oral spray’. Mean monthly WTP for the insurance premium for those who chose the ‘pills and oral spray’ was $Can8 (SD ± 15, median 4, range 0–200).
Conclusions: Assuming that 51% of the general population are willing to pay additional premiums as reported in this study, the premiums collected would cover the cost of Sativex® for all Canadian MS patients experiencing pain, with a surplus.



Similar content being viewed by others
Notes
The sample size was based on significance and a desired precision level of 5%. The calculation was based on the assumption that equal proportions of participants would select each of the three preference options (i.e. treatment option 1, treatment option 2 and indifferent). Furthermore, we assumed that ≥90% of the data collected would be usable. Therefore, we estimated that the minimum required sample size was 450 participants in total. We ended up collecting data from 500 participants.
References
Bruck G, Stadelmann C. Inflammation and degeneration in multiple sclerosis. Neurol Sci 2003; 24 Suppl. 5: S265–7
Multiple Sclerosis Society of Canada. MS information: frequently asked questions. Toronto (ON): Multiple Sclerosis Society of Canada, 2004 [online]. Available from URL: http://www.mssociety.ca/en/information/faq.htm#1 [Accessed 2006 Nov 15]
Beck C, Metz L, Svenson L, et al. Regional variation of multiple sclerosis prevalence in Canada. Multiple Sclerosis 2005; 11: 516–9
Brichetto G, Messmer Uccelli M, Mancardi G, et al. Symptomatic medication use in multiple sclerosis. Mult Scler 2003; 9: 458–60
Archibald C, McGrath P, Ritvo P. Pain prevalence, severity and impact in a clinical sample of multiple sclerosis patients. Pain 1994; 58: 89–93
Solaro C, Lunardi G, Mancardi G. Pain and MS. Int MS J 2003; 10 (1): 14–9
Burks J. A review of current medical aspects of multiple sclerosis. J Neurol Rehabil 1992; 6: 131–9
Indaco A, Iachetta C, Nappi C, et al. Chronic and acute pain syndromes in patients with multiple sclerosis. Acta Neurol Scand 1994; 16: 97–102
Moulin D. Pain in central and peripheral demyelinating disorders. Neurol Clin 1998; 16: 889–97
Maloni H. Multiple sclerosis and pain. Hackensack (NJ): The Consortium of Multiple Sclerosis Centers, 2004 [online]. Available from URL: http://www.mscare.org/cmsc/CMSC-Multiple-Sclerosis-and-Pain.html [Accessed 2006 Nov 15]
Piwko C, Desjardins OB, Bereza BG, et al. Pain due to multiple sclerosis: analysis of the prevalence and economic burden in Canada. Pain Res Manag 2007; 12 (4): 259–65
Pollman W, Feneberg W, Steinbrecher A, et al. Therapy of pain syndromes in multiple sclerosis: an overview with evidence-based recommendations. Fortschr Neurol Psychiatr 2005; 73: 268–85
Health Canada. Drugs and health products: approval of SATIVEX® with conditions. Fact sheet. Ottawa (ON): Health Canada, 2008 [online]. Available from URL: http://www.hc-sc.gc.ca/dhp-mps/prodpharma/notices-avis/conditions/sativex_fs_fd_091289-eng.php [Accessed 2008 Dec 17]
Russo E. Sativex® cannabis based medicine maintains improvements in sleep quality in patients with multiple sclerosis and neuropathic pain [poster]. Wiltshire: GW Pharmaceuticals [online]. Available from URL: http://www.safeaccess.ca/research/pdf/Russo_AAN_Poster.pdf [Accessed 2006 Nov 15]
Gafni A. Willingness-to-pay as a measure of benefits: relevant questions in the context of public decision making about health care programmes. Med Care 1991; 29: 1246–52
O’Brien B, Gafni A. When do the ‘dollars’ make sense? Toward a conceptual framework for contingent valuation studies in health care. Med Dec Mak 1996; 16: 288–302
Gafni A. Willingness-to-pay: what’s in a name? Pharmacoeconomics 1998; 14 (5): 465–70
Gafni A. Willingness-to-pay (WTP) in the context of an economic evaluation of healthcare programs: theory and practice. Am J Manage Care 1997; 3 Suppl. 5: S21–32
O’Brien B, Goeree R, Gafni A, et al. Assessing the value of a new pharmaceutical: a feasibility study of contingent valuation in managed care. Med Care 1998; 36: 370–84
Matthews D, Rocchi A, Gafni A. Putting your money where your mouth is: willingness to pay for dental gel. Pharmacoeconomics 2002; 20 (4): 245–55
Levine M, Gafni A, Markham B, et al. A bedside decision instrument to elicit a patient’s preference concerning adjuvant chemotherapy for breast cancer. Ann Int Med 1992; 117: 53–8
Whelan T, Levine M, Willan A, et al. Effect of a decision aid on knowledge and treatment decision making for breast cancer surgery: a randomized trial. JAMA 2004; 292: 435–41
Rog D, Nurmikko T, Friede T, et al. Randomized controlled trial of cannabis based medicine in central pain due to multiple sclerosis. Neurology 2005; 65: 812–9
Miles M, Huberman A. Examples of strategies in qualitative inquiry: qualitative data analysis. Thousand Oaks (CA): Sage Publications, 1994: 27–30
StatsCan. Population by mother tongue, by province and territory (2001 census). Canada: Statistics Canada, 2006 [online]. Available from URL: http://www.40.statcan.ca/i01/cst01/demo31a.htm?sdi=population%20mother%20tongue [Accessed 2006 Nov 15]
StatsCan. Population by sex and age group, by province and territory. Canada: Statistics Canada, 2007 [online]. Available from URL: http://www40.statcan.gc.ca/i01/cst01/demo31a-eng.htm [Accessed 2006 Nov 15]
StatsCan. Labour force characteristics. Canada: Statistics Canada, 2006 [online]. Available from URL: http://www40.statcan.ca/i01/cst01/econ10.htm. [Accessed 2006 Nov 15]
Drummond M, O’Brien B, Stoddart G, et al. Cost-benefit analysis. In: Drummond MF, Sculpher MJ, Torrance GW, et al., editors. Methods for the economic evaluation of health care programmes. New York: Oxford University Press Inc, 2005: 203–31
Acknowledgements
The authors would like to acknowledge Dr Amiram Gafni, from McMaster University for scientific input in selected parts of the study, as well as Dr Adriana Costei and Rima Aziziyeh for their technical contribution. The authors would also like to thank Dr Virginia Devonshire from the University of British Columbia, Dr Mark Freedman from the Ottawa Hospital, Dr Allan Gordon from the Wasser Pain Management Centre at the Mount Sinai Hospital in Toronto and Dr Melanie Ursell from St. Michael’s Hospital for their clinical input in developing the decision boards.
This study was sponsored by Bayer Inc., and conducted by PharmIdeas under contract to Bayer Inc. B. Jaszewski is employed by Bayer Inc. Thomas Einarson was, at the time of analysis, a principal of PharmIdeas and has previously consulted with Bayer. Michael Iskedjian is a principal of PharmIdeas.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iskedjian, M., Desjardins, O., Piwko, C. et al. Willingness to Pay for a Treatment for Pain in Multiple Sclerosis. Pharmacoeconomics 27, 149–158 (2009). https://doi.org/10.2165/00019053-200927020-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-200927020-00005