Abstract
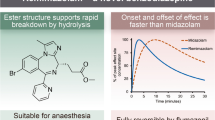
Remifentanil is a synthetic opioid that was developed in the early 1990s and introduced into clinical use in 1996. It is a methyl ester and is metabolised by nonspecific tissue and plasma esterases. Consequently, it is a drug that undergoes rapid elimination and has a reported terminal elimination half-life of between 10 and 35 minutes. Because there is no drug accumulation, the context-sensitive half-time remains constant; thus the pharmacokinetics of the drug do not change regardless of the duration of infusion. The organ-independent elimination of remifentanil, coupled with the fact that its clearance is greater in infants and neonates compared with older age groups, make its pharmacokinetic profile different from any other opioid. In addition, its unique metabolism confers predictability in its clinical use. Like other opioid μ, receptor agonists, remifentanil provides dose-dependent analgesia, while the adverse effects of this drug, e.g. respiratory depression, are also thought to be dose related. The incidence of nausea and vomiting appear similar to other opioids. Its rapid and consistent metabolism regardless of duration of infusion has made remifentanil an attractive analgesic/anaesthetic option for paediatric care providers.


Similar content being viewed by others
References
Anand KJ, Hickey PR. Halothane-morphine compared with high-dose sufentanil for anesthesia and postoperative analgesia in neonatal cardiac surgery. N Engl J Med 1992; 326(1): 1–9
Amin HM, Sopchak AM, Esposito BF, et al. Naloxone-induced and spontaneous reversal of depressed ventilatory responses to hypoxia during and after continuous infusion of remifentanil and alfentanil. J Pharmacol Exp Ther 1995; 274: 34–9
Davis PJ, Stiller RL, Wilson AS, et al. In vitro remifentanil metabolism: the effects of whole blood constituents and plasma butyrylcholinesterase. Anesth Analg 2002; 95: 1305–7
Hoke JF, Cunningham F, James MK, et al. Comparative pharmacokinetics and pharmacodynamics of remifentanil, its principle metabolite (GR990291) and alfentanil in dogs. J Pharmacol Exp Ther 1997; 281: 226–32
Egan TD, Lemmens HJM, Fiset P, et al. The pharmacokinetics of the new short-acting opioid remifentanil (GI87084B) in healthy adult male volunteers. Anesthesiology 1993; 79: 881–92
Egan TD, Minto CF, Hermann DJ, et al. Remifentanil versus alfentanil: comparative pharmacokinetics and pharmacodynamics in healthy adult male volunteers. Anesthesiology 1996; 84: 821–33
Glass PSA, Gan TJ, Howell S. A review of the pharmacokinetics and pharmacodynamics of remifentanil. Anesth Analg 1999; 89: S7–14
Glass PSA, Hardman D, Kamiyama Y, et al. Preliminary pharmacokinetics and pharmacodynamics of an ultra-shortacting opioid: remifentanil (GI87084B). Anesth Analg 1993; 77: 1031–40
Westmoreland CL, Hoke JF, Sebel PS, et al. Pharmacokinetics of remifentanil (GI87084B) and its major metabolite (GI90291) in patients undergoing elective inpatient surgery. Anesthesiology 1993; 79: 893–903
Shafer SL, Varvel JR. Pharmacokinetics, pharmacodynamics, and rational opioid selection. Anesthesiology 1991; 74: 53–63
Kapila A, Glass PSA, Jacobs JR, et al. Measured context-sensitive half times of remifentanil and alfentanil. Anesthesiology 1995; 83: 968–75
Hughes MA, Glass PSA, Jacobs JR. Context-sensitive half-time in multicompartment pharmacokinetic models for intravenous anesthetic drugs. Anesthesiology 1992; 76: 334–41
Dershwitz M, Hoke JF, Rosow CE, et al. Pharmacokinetics and pharmacodynamics of remifentanil in volunteer subjects with severe liver disease. Anesthesiology 1996; 84: 812–20
Hoke F, Shlugman D, Dershwitz M, et al. Pharmacokinetics and pharmacodynamics of remifentanil in persons with renal failure compared with healthy volunteers. Anesthesiology 1997; 87: 533–41
Dahaba AA, Oettl K, von Klobucar F, et al. End-stage renal failure reduces central clearance and prolongs the elimination half life of remifentanil. Can J Anaesth 2002; 49: 369–74
Michelsen LG, Holford NHG, Lu W, et al. The pharmacokinetics of remifentanil in patients undergoing coronary artery bypass grafting with cardiopulmonary bypass. Anesth Analg 2001; 93: 1100–5
Russell D, Royston D, Rees PH, et al. Effect of temperature and cardiopulmonary bypass on the pharmacokinetics of remifentanil. Br J Anaesth 1997; 79: 546–59
Ross AK, Davis PJ, del Dear G, et al. Pharmacokinetics of remifentanil in anesthetized pediatric patients undergoing elective surgery or diagnostic procedures. Anesth Analg 2001; 93: 1393–401
Davis PJ, Wilson AS, Siewers RD, et al. The effects of cardiopulmonary bypass on remifentanil kinetics in children undergoing atrial septal defect repair. Anesth Analg 1999; 89: 904–8
Wee LH, Moriarty A, Cranston A, et al. Remifentanil infusion for major abdominal surgery in small infants. Paediatr Anaesth 1999; 9: 415–8
Davis PJ, Finkel J, Orr R, et al. A randomized, double-blinded study of remifentanil versus fentanyl for tonsillectomy and adenoidectomy surgery in pediatric ambulatory surgical patients. Anesth Analg 2000; 90: 863–71
Chiaretti A, Pietrini D, Piastra M, et al. Safety and efficacy of remifentanil in craniosynostosis repair in children less than 1 year old. Pediatr Neurosurg 2000; 33: 83–8
Chambers N, Lopez T, Thomas J, et al. Remifentanil and the tunneling phase of paediatric ventriculoperitoneal shunt insertion: a double-blind, randomized, prospective study. Anaesthesia 2002; 57: 133–9
Friesen RH, Veit AS, Archibald DJ, et al. A comparison of remifentanil and fentanyl for fast track paediatric cardiac anaesthesia. Paediatr Anaesth 2003; 13: 122–5
Dönmez A, Kizilkan A, Berksun H, et al. One center’s experience with remifentanil infusions for pediatric cardiac catheterization. J Cardiothorac Vasc Anesth 2001; 15: 736–9
Litman RS. Conscious sedation with remifentanil during painful medical procedures. J Pain Symptom Manage 2000; 19: 468–71
Keidan I, Berkenstadt H, Sidi A, et al. Propofol/remifentanil versus propofol alone for bone marrow aspiration in paediatric haemato-oncological patients. Paediatr Anaesth 2001; 11: 297–301
German JW, Aneja R, Heard C, et al. Continuous remifentanil for pediatric neurosurgery patients. Pediatr Neurosurg 2000; 33: 227–9
Minto CF, Schnider TW, Egan TD, et al. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil. Anesthesiology 1997; 86: 10–23
Dershwitz M, Randel GI, Rosow CE, et al. Initial clinical experience with remifentanil, a new opioid metabolized by esterases. Anesth Analg 1995; 81: 619–23
Lang E, Kapila A, Shlugman D, et al. Reduction of isoflurane minimal alveolar concentration by remifentanil. Anesthesiology 1996; 85: 721–8
Westmoreland CL, Sebel PS, Gropper A. Fentanyl of alfentanil decreases the minimal alveolar anesthetic concentration of isoflurane in surgical patients. Anesth Analg 1994; 78: 23–8
Robinson DN, O’Brien K, Kumar R, et al. Tracheal intubation without neuromuscular blockade in children: a comparison of propofol combined either with alfentanil or remifentanil. Paediatr Anaesth 1998; 8: 467–71
Klemola UM, Hiller A. Tracheal intubation after induction of anesthesia in children with propofol: remifentanil or propofolrocuronium. Can J Anesth 2000; 47(9): 854–9
Davis PJ, Lerman J, Suresh S, et al. A randomized multicenter study of remifentanil compared with alfentanil, isoflurane, or propofol in anesthetized pediatrie patients undergoing elective strabismus surgery. Anesth Analg 1997; 84: 982–9
Muñoz H, Corínez L, Altermatt F, et al. Remifentanil requirements during sevoflurane administration to block somatic and cardiovascular responses to skin incision in children and adults. Anesthesiology 2002; 97: 1142–5
Prys-Roberts C, Lerman J, Murat I, et al. Comparison of remifentanil versus regional anaesthesia in children anaesthetized with isoflurane/ nitrous oxide. Anaesthesia 2000; 55: 870–6
Davis PJ, Galinkin J, McGowan FX, et al. A randomized multicenter study of remifentanil compared with halothane in neonates and infants undergoing pyloromyotomy: I. Emergence and recovery profiles. Anesth Analg 2001; 93: 1380–6
Galinkin JL, Davis PJ, McGowan FX, et al. A randomized multicenter study of remifentanil compared with halothane in neonates and infants undergoing pyloromyotomy: II. Perioperative breathing patterns in neonates and infants with pyloric stenosis. Anesth Analg 2001; 93: 1387–92
Pinsker MC, Carroll NV. Quality of emergence from anesthesia and incidence of vomiting with remifentanil in a pediatrie population. Anesth Analg 1999; 89: 71–4
Rüsch D, Happe W, Wulf H. Is TIVA with remifentanil/propofol superior to sevoflurane/N2O regarding PONV in children undergoing strabismus surgery? Der Anaesthesist 1999; 48: 80–8
Eltzschig HK, Schroeder TH, Eissler BJ, et al. The effect of remifentanil or fentanyl on postoperative vomiting and pain in children undergoing strabismus surgery. Anesth Analg 2002; 94: 1173–7
Haber GW, Litman RS. Generalized tonic-clonic activity after remifentanil administration. Anesth Analg 2001; 93: 1532–3
Strong WE, Matson M. Probable seizure after alfentanil. Anesth Analg 1989; 68: 692–3
Safwat AM, Daniel D. Grand mal seizure after fentanyl administration [letter]. Anesthesiology 1983; 59: 78
Brian JE, Seifen AB. Tonic-clonic activity after sufentanil. Anesth Analg 1987; 66: 481–3
Blair JM, Hill DA. Probable seizure after remifentanil in a 4-year-old boy. Anaesthesia 2000; 55: 489–518
Acknowledgements
Dr Peter J. Davis has undertaken research/consultancy work for Abbott Laboratories, Baxter Health Care Corporation and GlaxoSmithKline. No sources of funding were used to assist in the preparation of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davis, P.J., Cladis, F.P. The Use of Ultra-Short-Acting Opioids in Paediatric Anaesthesia. Clin Pharmacokinet 44, 787–796 (2005). https://doi.org/10.2165/00003088-200544080-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200544080-00002