Summary
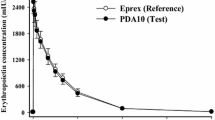
After long term therapy with epoetin (recombinant human erythropoietin) 17 patients with end-stage renal disease (ESRD) were studied in 3 groups to assess pharmacokinetics during the intertreatment interval and during haemofiltration and dialysis treatment. Epoetin was measured by radioimmunoassay. After an intravenous bolus of epoetin 150 U/kg bodyweight, the half-life was 7.7h, steady-state volume of distribution was 0.066 L/kg and total plasma clearance was 5.4 ml/min. The mean steady-state serum concentration during multiple-dose administration was 656 U/L. The drug was not eliminated by haemofiltration or dialysis. Long term treatment of ESRD patients with epoetin does not significantly alter the pharmacokinetic profile of the drug. Epoetin dosage adjustment or substitution after haemofiltration and dialysis is not necessary.
Similar content being viewed by others
References
Akahane K, Tojo A, Fukamachi H, Kitamura T, Saito T, et al. Binding of iodinated erythropoietin to rat bone marrow cells under normal and anemic conditions. Experimental Hematology 17: 177–182, 1989
Brockmöller J, Köchling J, Scigalla P, Weber W, Roots I, et al. Pharmacokinetics and pharmacodynamics of recombinant human erythropoietin. European Journal of Pharmacology 183: 2391–2392, 1990
Brockmöller J, Köchling J, Weber W, Looby M, Roots I, et al. The pharmacokinetics and pharmacydynamics of recombinant human erythropoietin in haemodialysis patients. British Journal of Clinical Pharmacology 34: 499–508, 1992
Cotes PM, Pippard MJ, Reid CDL, Winearls CG, Oliver DO, et al. Characterization of the anaemia of chronic renal failure and the mode of its correction by a preparation of human erythropoietin (r-HuEPO). An investigation of the pharmacokinetics of intravenous erythropoietin and its effects on erythrokinetics. Quarterly Journal of Medicine 70: 113–137, 1989
Davis JM, Arakawa T, Strickland TW, Yphantis DA. Characterization of recombinant human erythropoietin produced in Chinese hamster ovary cells. Biochemistry 26: 2633–2638, 1987
DeSpain M, Cheung A. Binding of recombinant erythropoietin (EPO) to hemodialysis membranes. Kidney International 37: 293, 1990
Eckardt K-U, Kurtz A, Hirth P, Scigalla P, Wieczorek L, et al. Evaluation of the stability of human erythropoietin in samples for radioimmunoassay. Klinische Wochenschrift 66: 241–245, 1988
Egrie JC, Eschbach JW, McGuire T, Adamson JW. Pharmacokinetics of recombinant human erythropoietin (rHuEpo) administered to hemodialysis (HD) patients. Kidney International 33: 262, 1988
Eschbach JW, Egrie JC, Downing MR, Browne JK, Adamson JW. Correction of the anemia of end-stage renal disease with recombinant human erythropoietin. Results of a combined phase I and II clinical trial. New England Journal of Medicine 316: 73–78, 1987
Flaharty KK, Caro J, Erslev A, Whalen JJ, Morris M, et al. Pharmacokinetics and erythropoietic response to human recombinant erythropoietin in healthy men. Clinical Pharmacology and Therapeutics 47: 557–564, 1990
Gibson TP. Influence of renal disease on pharmacokinetics. In Evans et al. (Eds) Applied pharmacokinetics: principles of therapeutic drug monitoring, pp. 32–56, San Francisco, Applied Therapeutics, 1980
Halstenson CE, Macres M, Katz SA, Schnieders JR, Watanab M, et al. Comparative pharmacokinetics and pharmacodynamics of epoetin alfa and epoetin beta. Clinical Pharmacology and Therapeutics 50: 702–712, 1991
Heinzel G, Woloszczak R, Thomann P. (Eds) TopFit (Version 2.0): pharmacokinetic and pharmacodynamic data analysis system for the PC, G Fischer, Stuttgart/Jena/New York, 1993
Kampf D, Eckardt KU, Fischer HC, Schmalisch C, Ehmer B, et al. Pharmacokinetics of recombinant human erythropoietin in dialysis patients after single and multiple subcutaneous administrations. Nephron 61: 393–398, 1992
Kindler J, Eckardt K-U, Ehmer B, Jandeleit K, Kurtz A, et al. Single-dose pharmacokinetics of recombinant human erythropoietin in patients with various degrees of renal failure. Nephrology Dialysis Transplantation 4: 345–349, 1989
Lim VS, DeGowin RL, Zavala D, Kirchner PT, Abels R, et al. Recombinant human erythropoietin treatment in pre-dialysis patients: a double-blind placebo-controlled trial. Annals of Internal Medicine 110: 108–114, 1989
Maxwell AP, Douglas JF, Afrasiabi M, Lappin TRJ, Bridges JM. Erythropoietin pharmacokinetics and red cell metabolism in haemodialysis patients. Nephrology Dialysis Transplantation 4: 476, 1989
Mufson RA, Gesner TG. Binding and internalization of recombinant human erythropoietin in murine erythroid precursor cells. Blood 69: 1485–1490, 1987
Muirhead N, Keown PA, Slaughter D, Mazaheri R, Jevnikar AM, et al. Recombinant human erythropoietin in the anaemia of chronic renal failure: a pharmacokinetic study. Nephrology Dialysis Transplantation 3: 499, 1988
Neumayer HH, Brockmöller J, Fritschka E, Roots I, Scigalla P, et al. Pharmacokinetics of recombinant human erythropoietin after sc administration and in long-term iv treatment in patients on maintenance haemodialysis. In Baldamus et al. (Eds) Erythropoietin: from molecular structure to clinical application, Contributions to Nephrology, Vol. 76, pp. 131–142, Karger, Basel, 1989
Nielsen OJ. Pharmacokinetics of recombinant human erythropoietin in chronic haemodialysis patients. Pharmacology and Toxicology 66: 83–86, 1990
Salmonson T, Danielson BG, Grahnén A, Wikström B. Pharmacokinetics of intravenous recombinant human erythropoietin in patients with chronic renal failure. Journal of Internal Medicine 228: 53–57, 1990a
Salmonson T, Wikström B, Danielson BG. Pharmacokinetics of intravenous recombinant erythropoietin (r-EPO) prior to and during long-term treatment in haemodialysis patients. Nephrology Dialysis Transplantation 5: 742, 1990b
Sawada K, Krantz SB, Sawyer ST, Civin CI. Quantitation of specific binding of erythropoietin to human erythroid colony-forming cells. Journal of Cellular Physiology 137: 337–345, 1988
Stockenhuber F, Loibl U, Gottsauner-Wolf M, Jahn C, Manker W, et al. Pharmacokinetics and dose response after intravenous and subcutaneous administration of recombinant erythropoietin in patients on regular haemodialysis treatment or continuous ambulatory peritoneal dialysis. Nephron 59: 399–402, 1991
Takeda F, Momose T, Ubukata E, Morio K, Sano M, et al. Pharmacokinetics of recombinant human erythropoietin in patients with chronic renal failure. XIth International Congress of Nephrology 1990, Tokyo, Abstract No. 293 A, 1990
Winearls CG, Oliver DO, Pippard MJ, Reid C, Downing MR, et al. Effect of human erythropoietin derived from recombinant DNA on the anaemia of patients maintained by chronic haemodialysis. Lancet 2: 1175–1178, 1986
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gladziwa, U., Klotz, U., Bäumer, K. et al. Pharmacokinetics of Epoetin (Recombinant Human Erythropoietin) after Long Term Therapy in Patients Undergoing Haemodialysis and Haemofiltration. Clin-Pharmacokinet 25, 145–153 (1993). https://doi.org/10.2165/00003088-199325020-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199325020-00007