Abstract
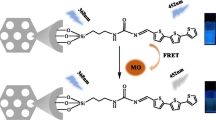
An inorganic-organic silica material (SBA-15-1), prepared by immobilization of the naphthalimide derivative within the channels of the mesoporous silica material SBA-15, is characterized by several spectroscopic methods. SBA-15-1 can be used as a chemical sensor for detecting and removing Hg2+ in a heterogeneous system. The fluorescence enhancement of SBA-15-1 was attributed to the formation of a complex between SBA-15-1 and Hg2+ by a 1:1 complex ratio with the photo-induced electron transfer (PET) being forbidden. The sensor can be applied to the quantification of Hg2+ with a linear range covering from 1.0 × 10−7 to 1.0 × 10−5 M under the neutral condition. Most importantly, the fluorescence changes of the sensor are remarkably specific for Hg2+ in the presence of other metal ions. Moreover, the response of the sensor toward Hg2+ is fast and chemically reversible. In addition, the sensor has been used for the determination of Hg2+ in environmental samples with satisfactory results.
Similar content being viewed by others
References
United Nations Environment Programme. Chemicals, Global Mercury Assessment, UNEP Chemicals, 2002, Geneva, Switzerland, 100.
D. W. Boening, Chemosphere, 2000, 40, 1335.
W. Zheng, M. Aschner, and J.-F. Ghersi-Egea, Toxicol. Appl. Pharmacol., 2003, 192, 1.
J. Mutter, J. Naumann, R. Schneider, H. Walach, and B. Haley, Neuroendocrinol. Lett., 2005, 26, 439.
A. P. de Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher, and T. E. Rice, Chem. Rev., 1997, 97, 1515.
C. Bargossi, M. C. Fiorini, M. Montalti, L. Prodi, and N. Zaccheroni, Coord. Chem. Rev., 2000, 208, 17.
G. Aragay, J. Pons, and A. Merkoci, Chem. Rev., 2011, 111, 3433.
D. T. Quang and J. S. Kim, Chem. Rev., 2010, 110, 6280.
E. M. Nolan and S. J. Lippard, Chem. Rev., 2008, 108, 3443.
Nuriman, B. Kuswandi, and W. Verboom, Anal. Chim. Acta, 2009, 655, 75.
A. Thakur, S. Sardar, and S. Ghosh, Inorg. Chem., 2011, 50, 7066.
M. H. Yang, P. Thirupathi, and K. H. Lee, Org. Lett., 2011, 13, 5028.
V. Bhalla , M. Kumar, P. R. Sharma, and T. Kaur, Inorg. Chem., 2012, 51, 2150.
Y.-J. Gong, X.-B. Zhang, C.-C. Zhang, A.-L. Luo, T. Fu, W. Tan, G.-L. Shen, and R.-Q. Yu, Anal. Chem., 2012, 84, 10777.
S. J. Lee, S. S. Lee, M. S. Lah, J. Hong, and J. H. Jung, Chem. Commun., 2006, 4539.
H. Y. Lee, D. R. Bae, J. C. Park, H. Song, W. S. Han, and J. H. Jung, Angew. Chem., Int. Ed., 2009, 48, 1239.
Z. Jin, X.-B. Zhang, D.-X. Xie, Y.-J. Gong, J. Zhang, X. Chen, G.-L. Shen, and R.-Q. Yu, Anal. Chem., 2010, 82, 6343.
Z. Jin, D.-X. Xie, X.-B. Zhang, Y.-J. Gong, and W. Tan, Anal. Chem., 2012, 84, 4253.
A. E. Kadib, R. Chimenton, A. Sachse, F. Fajula, A. Galarneau, and B. Coq, Angew. Chem., Int. Ed., 2009, 48, 4969.
I. Agirrezabal-Telleria, J. Requies, M. B. Güemez, and P. L. Arias, Appl. Catal., B, 2014, 145, 34.
J. A. Melero, G. Morales, J. Iglesias, M. Paniagua, B. Hernández, and S. Penedo, Appl. Catal., A, 2013, 466, 116.
J. Mondal, M. Nandi, A. Modak, and A. Bhaumik, J. Mol. Catal. A: Chem., 2012, 363 – 364, 254.
M. Vallet-Reqi, F. Balas, and D. Arcos, Angew. Chem., Int. Ed., 2007, 46, 7548.
J. Pang, Y. Luan, X. Yang, Y. Jiang, L. Zhao, Y. Zong, and Z. Li. Mini, Rev. Med. Chem., 2012, 12, 775.
T. Balaji, M. Sasidharan, and H. Matsunaga, Analyst, 2005, 130, 1162.
Y. Wang, B. Li, L. Zhang, L. Liu, Q. Zuo, and P. Li, New J. Chem., 2010, 34, 1946.
Z. Dong, X. Tian, Y. Chen, J. Hou, and J. Ma, RSC Adv., 2013, 3, 2227.
D. Wu, Z. Wang, G. Wu, and W. Huang, Mater. Chem. Phys., 2012, 137, 428.
Q. Meng, X. Zhang, C. He, P. Zhou, W. Su, and C. Duan, Talanta, 2011, 84, 53.
P. Zarabadi-Poor, A. Badiei, A. A. Yousefi, and J. Barroso-Flores, J. Phys. Chem. C, 2013, 117, 9281.
L. Gao, J. Q. Wang, L. Huang, X. X. Fan, J. H. Zhu, Y. Wang, and Z. G. Zou, Inorg. Chem., 2007, 46, 10287.
Q. Meng, X. Zhang, C. He, G. He, P. Zhou, and C. Duan, Adv. Funct. Mater., 2010, 20, 1903.
M. Yadavi, A. Badiei, and G. M. Ziarani, Appl. Surf. Sci., 2013, 279, 121.
C. Niu, G. Zeng, L. Chen, G. Shen, and R. Yu, Analyst, 2004, 129, 20.
V. B. Bojinov, I. P. Panova, and J. Chovelon, Sens. Actuators, B, 2008, 135, 172.
C.-Y. Li, F. Xu, Y.-F. Li, K. Zhou, and Y. Zhou, Anal. Chim. Acta, 2012, 717, 122.
J. Gan, K. Chen, C.-P. Chang, and H. Tian, Dyes Pigm., 2003, 57, 21.
D. W. Cho, M. Fujitsuka, A. Sugimoto, U. C. Yoon, P. S. Mariano, and T. Majima, J. Phys. Chem. B, 2006, 110, 11062.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, YF., Wei, YF., Tan, Y. et al. Detection and Removal of Hg2+ Based on Mesoporous Silica Material Functionalized by Naphthalimide in Aqueous Solution. ANAL. SCI. 30, 257–262 (2014). https://doi.org/10.2116/analsci.30.257
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.30.257