Abstract
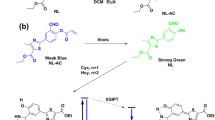
In this work, a new fluorescence probe (DC) with a donor–π–acceptor (D–π–A) structure was designed and synthesized for the detection of three kinds of biothiols (Cys, Hcy and GSH) in live cells and organisms. DC displayed an intense red-emission centered at 625 nm. In the presence of biothiols, the nucleophilic addition reaction between the C=C double bond of DC and the sulfhydryl group (–SH) of biothiols occurred, resulting in obvious fluorescence quenching responses. DC exhibited high selectivity towards biothiols over other common bioactive species with low detection limits (0.26, 0.43, and 0.44 µM for Cys, Hcy and GSH, respectively). In addition, DC displayed a rapid response to biothiols within 4 min. The applications of DC in biothiols detection and imaging were then successfully demonstrated for the real-time monitoring of endogenous and exogenous biothiols in live cells and live animals.
Graphical abstract











Similar content being viewed by others
References
B. Gao, L. Cui, Y. Pan, G. Zhang, Y. Zhou, C. Zhang, S. Shuang, C. Dong, RSC Adv. 6, 43028 (2016)
J.S. Lan, R.F. Zeng, Y. Liu, Y.W. Xiang, X.Y. Jiang, L. Liu, S.S. Xie, Y. Ding, T. Zhang, Analyst 144, 3676 (2019)
X. Chen, Y. Zhou, X. Peng, J. Yoon, Chem Soc Rev. 39, 2120 (2010)
X. Xia, Y. Qian, B. Shen, J. Mater. Chem. B 6, 3023 (2018)
Y. Cao, J. Liu, L. Zou, B. Ye, G. Li, Anal. Chim. Acta 1145, 46 (2021)
Y. Hu, C.H. Heo, G. Kim, E.J. Jun, J. Yin, H.M. Kim, J. Yoon, Anal. Chem. 87, 3308 (2015)
Z.H. Fu, X. Han, Y. Shao, J. Fang, Z.H. Zhang, Y.W. Wang, Y. Peng, Anal. Chem. 2017, 89 (1937)
L.Y. Niu, Y.Z. Chen, H.R. Zheng, L.Z. Wu, C.H. Tung, Q.Z. Yang, Chem. Soc. Rev. 44, 6143 (2015)
Y. Yue, F. Huo, P. Ning, Y. Zhang, J. Chao, X. Meng, C. Yin, J. Am. Chem. Soc. 139, 3181 (2017)
S. Feng, Y. Fang, W. Feng, Q. Xia, G. Feng, Dyes Pigm. 146, 103 (2017)
J. Zheng, J. Li, H. Luo, L. Sun, M. Sang, X. Yu, RSC Adv. 11, 4356 (2021)
W. Zhang, H. Zhang, M. Wang, P. Li, P.C. Ding, W. Zhang, H. Wang, B. Tang, Anal. Chem. 92, 16221 (2020)
Y. Yue, F. Huo, Y. Wang, K. Ma, X. Li, C. Yin, Chem. Commun. 56, 9146 (2020)
P. Hu, Y. Zhang, D. Wang, G. Qi, Y. Jin, Anal. Chem. 93, 4240 (2021)
J. Vacek, B. Klejdus, J. Petrlova, L. Lojkova, V. Kuban, Analyst 131, 1167 (2006)
T. Inoue, J.R. Kirchhoff, Anal. Chem. 74, 1349 (2002)
N. Burford, M.D. Eelman, D.E. Mahony, M. Morash, Chem. Commun. (2003). https://doi.org/10.1039/b210570e
R.F. Zeng, J.S. Lan, T. Wu, L. Liu, Y. Liu, R.J.Y. Ho, Y. Ding, T. Zhang, Food Chem. 318, 126358 (2020)
A.S.K. Kumar, W.B. Tseng, M.J. Wu, Y.Y. Huang, W.L. Tseng, Anal. Chim. Acta 1113, 43 (2020)
Y. Zou, M. Li, Y. Xing, T. Duan, X. Zhou, F. Yu, ACS Sens. 5, 242 (2020)
S. Lee, J. Li, X. Zhou, J. Yin, J. Yoon, Coord. Chem. Rev. 366, 29 (2018)
Q. Hu, C. Yu, X. Xia, F. Zeng, S. Wu, Biosens. Bioelectron. 81, 341 (2016)
H. Feng, Z. Zhang, Q. Meng, H. Jia, Y. Wang, R. Zhang, Adv. Sci. 5, 1800397 (2018)
W. Wan, Y. Huang, Q. Xia, Y. Bai, Y. Chen, W. Jin, M. Wang, D. Shen, H. Lyu, Y. Tang, X. Dong, Z. Gao, Q. Zhao, L. Zhang, Y. Liu, Angew. Chem. Int. Ed. 60, 11335 (2021)
W. Wan, W. Jin, Y. Huang, Q. Xia, Y. Bai, H. Lyu, D. Liu, X. Dong, W. Li, Y. Liu, Anal. Chem. 93, 1717 (2021)
Z. Xu, T. Qin, X. Zhou, L. Wang, B. Liu, Trends Anal. Chem. 121, 115672 (2019)
W. Liu, J. Chen, Z. Xu, Coord. Chem. Rev. 429, 213638 (2021)
Y.F. Kang, L.Y. Niu, Q.Z. Yang, Chin. Chem. Lett. 30, 1791 (2019)
N. Kaur, S. Chopra, G. Singh, P. Raj, A. Bhasin, S.K. Sahoo, A. Kuwar, N. Sing, J. Mater. Chem. B 6, 4872 (2018)
M.-Y. Wu, K. Li, C.-Y. Li, J.-T. Hou, X.-Q. Yu, Chem. Commun. 50, 183 (2014)
H. Feng, Y. Wang, J. Liu, Z. Zhang, X. Yang, R. Chen, Q. Meng, R. Zhang, J. Mater. Chem. B 7, 3909 (2019)
K. Xiong, F. Huo, J. Chao, Y. Zhang, C. Yin, Anal. Chem. 91, 1472 (2019)
C. Liu, R. Zhang, W. Zhang, J. Liu, Y.L. Wang, Z. Du, B. Song, Z.P. Xu, J.L. Yuan, J. Am. Chem. Soc. 141, 8462 (2019)
F. Liu, S. Zang, J. Jing, X. Zhang, Anal. Methods 12, 3797 (2020)
Y.L. Fu, X.G. Chen, H. Li, W. Feng, Q.H. Song, New J. Chem. 44, 13781 (2020)
S. Chakraborty, M.M. Joseph, S. Varughese, S. Ghosh, K.K. Maiti, A. Samanta, A. Ajayaghosh, Chem. Sci. 11, 12695 (2020)
H. Zhang, L. Xu, W. Chen, J. Huang, C. Huang, J. Sheng, X. Song, ACS Sens. 3, 2513 (2018)
L. Zhang, D. Duan, Y. Liu, C. Ge, X. Cui, J. Sun, J. Fang, J. Am. Chem. Soc. 136, 226 (2014)
Q. Miao, Q. Li, Q. Yuan, L. Li, Z. Hai, S. Liu, G. Liang, Anal. Chem. 87, 3460 (2015)
Acknowledgements
This work was supported by the Talent Program-Outstanding Youth Science Project, Liaoning (no. XLYC1807199), the Distinguished Professor Program of Liaoning (XLYC1802074), the “Seeding Raising” Project of Young Scientific and Technological Talents of Liaoning Provincial Department of Education (2019LNQN03; 2019LNJC18), and the Youth Fund of University of Science and Technology Liaoning (2018QN02).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Advanced Publication Released Online by J-STAGE July 30, 2021.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, Y., Shang, Z., Gu, P. et al. A red-emission fluorescence probe based on 1,4-addition reaction mechanism for the detection of biothiols in vitro and in vivo. ANAL. SCI. 38, 505–514 (2022). https://doi.org/10.2116/analsci.21P177
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.21P177