Abstract
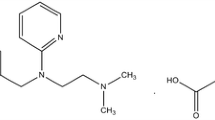
Triprolidine (Trip) ion selective electrodes of three types: the conventional polymer membrane (I), graphite coated electrode (II) and carbon paste electrode (III), have been prepared, based on the ion pair of triprolidine hydrochloride with sodium tetraphenylborate. The electrodes exhibit a linear response with a mean calibration graph slope of 56.12, 55.00 and 54.32 mV decade−1 at 25°C for I, II and III, respectively, within the concentration ranges 1.96 × 10−5–1.00 × 10−2 M for I and 3.84 × 10−5–1.00 × 10−2 M for II and III. The detection limits are 1.13 ± 0.13 × 10−5, 1.70 ± 0.06 × 10−5 and 1.78 ± 0.05 × 10−5 M for the three electrodes, respectively. The change of pH within the ranges 4.85–8.75 and 4.70–8.50 for I and III, respectively, did not affect the electrode performance. The standard electrode potentials were determined at different temperatures and were used to calculate the isothermal coefficient of the electrode. The electrodes showed a very good selectivity for Trip with respect to a large number of inorganic cations and compounds. The standard addition method was applied to the determination of TripCl in pure solution, pharmaceutical preparations, and urine samples.
Similar content being viewed by others
References
K. Parfitt (ed.), “Martindale”, 32th ed., 1999, Pharmaceutical Press, London, 420.
J. L. Chawla, R. A. Sodhi, and R. T. Sane, Indian Drugs, 1997, 34, 339.
D. D. Orsi, L. Gagliardi, A. Bolasco, and D. Tonelli, Chromatographia, 1996, 43, 496.
M. J. Akhtar, S. Khan, and M. Hafiz, J. Pharm. Biomed. Anal., 1994, 12, 379.
E. B. Hansen, T. A. Getek, and W. A. Korfmacher, J. Anal. Toxicol, 1989, 13, 185.
E. B. Hansenand H. C. Thompsen, LC Mag, 1985, 3, 986.
H. C. Thompsonand C. L. Holder, J. Chromatogr., 1984, 283, 251.
S. S. Yangand R. K. Gilpin, J. Chromatogr. Sci., 1988, 26, 416.
W. J. Bachman, J. Assoc. Off. Anal. Chem., 1981, 64, 564.
W. J. Bachman, J. Assoc. Off. Anal. Chem., 1980, 63, 91.
E. Dinc and F. Onur, S. T. P. Pharma Sciences, 1998, 8, 203.
R. Jones, M. J. Orchard, and K. Hall, J. Pharm. Biomed. Anal., 1985, 3, 335.
A. G. Davidsonand L. M. M. Mkoji, J. Pharm. Biomed. Anal., 1988, 6, 449.
C. S. P. Sastry, A. S. R. P. Tipirneni, and M. V. Suryanarayana, J. Pharm. Biomed. Anal., 1990, 8, 287.
F. H Metwally, J. Pharm. Biomed. Anal., 2001, 26, 265.
T. Aman, A. Ahmad, M. Aslam, and M. A. Kashmiri, Anal. Lett., 2002, 35, 733.
E. Bishop and W. Hussein, Analyst, 1984, 109, 913.
G. Kanoute, N. E. Guernet, and M. Guernet, J. Pharm. Biomed. Anal., 1988, 6, 977.
A. Craggs, G. J. Moody, and J. D. R. Thomas, J. Chem. Educ., 1974, 51, 541.
V. Oesch and W. Simon, Anal. Chem., 1980, 52, 692.
I. Svancara and K. Vytras, Chem. Listy, 1994, 88, 138.
A. F. Shoukry, S. S. Badawy, and Y. M. Issa, Analyst, 1986, 111, 1363.
D. G. Peter, J. M. Hayes, and G. M. Hieftje, “Chemical Separation and Measurements”, 1974, Saunders, Philadelphia, USA.
R. P. Buckand V. V. Cosofret, Pure Appl. Chem., 1993, 65, 1849.
R. P. Armstrongand G. Horvai, Electrochim. Acta, 1990, 35, 1.
U. Oesch, D. Ammann, and W. Simon, Clin. Chem., 1986, 32, 1448.
N. T. Abdel-Ghani, A. F. Shoukry, and R. M. ElNashar, Analyst, 2001, 126, 79.
R. P. Buck, Anal. Chem., 1976, 48, 23.
The United States Pharmacopeia 23, The National Formulary 18, United States Pharmacopeial Convention, INC., Twinbrook Parkway, Rockville, MD, 1995, 1607.
J. C. Millerand J. N. Miller, “Statistics for Analytical Chemistry”, 3rd ed., 1993, Ellis Horwood, Chichester, 53.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zayed, S.I.M. New Plastic Membrane and Carbon Paste Ion Selective Electrodes for the Determination of Triprolidine. ANAL. SCI. 20, 1043–1048 (2004). https://doi.org/10.2116/analsci.20.1043
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.20.1043