Abstract
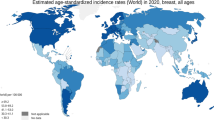
Breast cancer is globally the most common invasive cancer in women and remains one of the leading causes of cancer-related deaths. Surgery, radiotherapy, chemotherapy, immunotherapy, and endocrine therapy are currently the main treatments for this cancer type. However, some breast cancer patients are prone to drug resistance related to chemotherapy or immunotherapy, resulting in limited treatment efficacy. Consequently, traditional Chinese medicinal materials (TCMMs) as natural products have become an attractive source of novel drugs. In this review, we summarized the current knowledge on the active components of animal-derived TCMMs, including Ophiocordyceps sinensis-derived cordycepin, the aqueous and ethanolic extracts of O. sinensis, norcantharidin (NCTD), Chansu, bee venom, deer antlers, Ostrea gigas, and scorpion venom, with reference to marked anti-breast cancer effects due to regulating cell cycle arrest, proliferation, apoptosis, metastasis, and drug resistance. In future studies, the underlying mechanisms for the antitumor effects of these components need to be further investigated by utilizing multi-omics technologies. Furthermore, large-scale clinical trials are necessary to validate the efficacy of bioactive constituents alone or in combination with chemotherapeutic drugs for breast cancer treatment.
概要
乳腺癌已成为发病率最高的女性恶性肿瘤之一, 严重威胁女性健康。化疗联合手术、放射治疗几乎可用于乳腺癌治疗的各个阶段, 但化疗相关耐药性常导致治疗效果有限。传统中药材已成为极具吸引力的新型药物研发来源之一。具有抗乳腺癌效应的中药主要包括中草药类、动物类中药材, 近年来关于其抗肿瘤效应的机制研究层出不穷。目前虽有相关综述对抗乳腺癌的中草药进行了讨论分析, 而关于动物类来源中药材的抗乳腺癌机制却缺乏系统的总结和分析。本文通过系统地综述和分析, 探讨了冬虫夏草来源的虫草素及其水或乙醇提取物、蟾酥、去甲斑蝥素、蜂毒素、鹿茸、牡蛎等中药材可通过调节细胞周期、增殖、凋亡及肿瘤细胞耐药性发挥抗乳腺癌作用。在未来的研究中, 需要采用多组学技术进一步研究这些成分的抗乳腺癌作用机制。此外, 有必要进行相关临床试验以验证这些动物来源中药活性成分单独或与化疗药物联合治疗乳腺癌的疗效及安全性。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adekunle AI, Cromer D, Davenport MP, 2016. Artemisininbased treatments in pregnant women with malaria. N Engl J Med, 375(3):283–284. https://doi.org/10.1056/NEJMc1604709
Ahmadi S, Knerr JM, Argemi L, et al., 2020. Scorpion venom: detriments and benefits. Biomedicines, 8(5): 118. https://doi.org/10.3390/biomedicines8050118
Al-Asmari AK, Riyasdeen A, Islam M, 2018. Scorpion venom causes upregulation of p53 and downregulation of Bcl-xL and BID protein expression by modulating signaling proteins Erk1/2 and STAT3, and DNA damage in breast and colorectal cancer cell lines. Integr Cancer Ther, 17(2): 271–281. https://doi.org/10.1177/1534735417704949
An J, Minie M, Sasaki T, et al., 2017. Antimalarial drugs as immune modulators: new mechanisms for old drugs. Annu Rev Med, 68:317–330. https://doi.org/10.1146/annurev-med-043015-123453
Bhushan A, Gonsalves A, Menon JU, 2021. Current state of breast cancer diagnosis, treatment, and theranostics. Pharmaceutics, 13(5):723. https://doi.org/10.3390/pharmaceutics13050723
Boyman O, Arenas-Ramirez N, 2019. Development of a novel class of interleukin-2 immunotherapies for metastatic cancer. Swiss Med Wkly, 149:w14697. https://doi.org/10.4414/smw.2019.14697
Cai HW, Li J, Gu BH, et al., 2018. Extracts of Cordyceps sinensis inhibit breast cancer cell metastasis via down-regulation of metastasis-related cytokines expression. J Ethnopharmacol, 214:106–112. https://doi.org/10.1016/j.jep.2017.12.012
Callender SP, Mathews JA, Kobernyk K, et al., 2017. Microemulsion utility in pharmaceuticals: implications for multi-drug delivery. Int J Pharm, 526(1–2):425–442. https://doi.org/10.1016/j.ijpharm.2017.05.005
Cao YT, Wu JH, Pan HY, et al., 2019. Chemical profile and multicomponent quantitative analysis for the quality evaluation of toad venom from different origins. Molecules, 24(19):3595. https://doi.org/10.3390/molecules24193595
Carpena M, Nuñez-Estevez B, Soria-Lopez A, et al., 2020. Bee venom: an updating review of its bioactive molecules and its health applications. Nutrients, 12(11):3360. https://doi.org/10.3390/nu12113360
Ceremuga M, Stela M, Janik E, et al., 2020. Melittin—a natural peptide from bee venom which induces apoptosis in human leukaemia cells. Biomolecules, 10(2):247. https://doi.org/10.3390/biom10020247
Chen F, Zhu L, Hu JY, et al., 2020. Bufalin attenuates triple-negative breast cancer cell stemness by inhibiting the expression of SOX2/OCT4. Oncol Lett, 20(5):171. https://doi.org/10.3892/ol.2020.12028
Chen PX, Wang SN, Nie SP, et al., 2013. Properties of Cordyceps sinensis: a review. J Funct Foods, 5(2):550–569. https://doi.org/10.1016/j.jff.2013.01.034
Chen YJ, Kuo CD, Chen SH, et al., 2012. Small-molecule synthetic compound norcantharidin reverses multi-drug resistance by regulating Sonic hedgehog signaling in human breast cancer cells. PLoS ONE, 7(5):e37006. https://doi.org/10.1371/journal.pone.0037006
Chen YL, Bian XL, Guo FJ, et al., 2018. Two new 19-norbufadienolides with cardiotonic activity isolated from the venom of Bufo bufo gargarizans. Fitoterapia, 131: 215–220. https://doi.org/10.1016/j.fitote.2018.10.023
Chintalapati M, Truax R, Stout R, et al., 2009. In vitro and in vivo anti-angiogenic activities and inhibition of hormone-dependent and -independent breast cancer cells by ceramide methylaminoethylphosphonate. J Agric Food Chem, 57(12):5201–5210. https://doi.org/10.1021/jf803818y
Cho HJ, Jeong YJ, Park KK, et al., 2010. Bee venom suppresses PMA-mediated MMP-9 gene activation via JNK/p38 and NF-κB-dependent mechanisms. J Ethnopharmacol, 127(3):662–668. https://doi.org/10.1016/j.jep.2009.12.007
Choi S, Lim MH, Kim KM, et al., 2011. Cordycepin-induced apoptosis and autophagy in breast cancer cells are independent of the estrogen receptor. Toxicol Appl Pharmacol, 257(2):165–173. https://doi.org/10.1016/j.taap.2011.08.030
Chonco L, Landete-Castillejos T, Serrano-Heras G, et al., 2021. Anti-tumour activity of deer growing antlers and its potential applications in the treatment of malignant gliomas. Sci Rep, 11:42. https://doi.org/10.1038/s41598-020-79779-w
Cragg GM, Pezzuto JM, 2016. Natural products as a vital source for the discovery of cancer chemotherapeutic and chemopreventive agents. Med Princ Pract, 25(Suppl 2):41–59. https://doi.org/10.1159/000443404
Daniluk K, Kutwin M, Grodzik M, et al., 2019. Use of selected carbon nanoparticles as melittin carriers for MCF-7 and MDA-MB-231 human breast cancer cells. Materials (Basel), 13(1):90. https://doi.org/10.3390/ma13010090
Demain AL, Vaishnav P, 2011. Natural products for cancer chemotherapy. Microb Biotechnol, 4(6):687–699. https://doi.org/10.1111/j.1751-7915.2010.00221.x
DeNardo DG, Ruffell B, 2019. Macrophages as regulators of tumour immunity and immunotherapy. Nat Rev Immunol, 19(6):369–382. https://doi.org/10.1038/s41577-019-0127-6
Deng LJ, Qi M, Li N, et al., 2020. Natural products and their derivatives: promising modulators of tumor immunotherapy. J Leukoc Biol, 108(2):493–508. https://doi.org/10.1002/JLB.3MR0320-444R
DeSantis CE, Ma JM, Gaudet MM, et al., 2019. Breast cancer statistics, 2019. CA Cancer J Clin, 69(6):438–451. https://doi.org/10.3322/caac.21583
Díaz-García A, Varela D, 2020. Voltage-gated K+/Na+ channels and scorpion venom toxins in cancer. Front Pharmacol, 11:913. https://doi.org/10.3389/fphar.2020.00913
Dong CH, Guo SP, Wang WF, et al., 2015. Cordyceps industry in China. Mycology, 6(2):121–129. https://doi.org/10.1080/21501203.2015.1043967
Dong YH, Yin ST, Li JH, et al., 2011. Bufadienolide compounds sensitize human breast cancer cells to TRAIL-induced apoptosis via inhibition of STAT3/Mcl-1 pathway. Apoptosis, 16(4):394–403. https://doi.org/10.1007/s10495-011-0573-5
Dorn DC, Kou CA, Png KJ, et al., 2009. The effect of cantharidins on leukemic stem cells. Int J Cancer, 124(9): 2186–2199. https://doi.org/10.1002/ijc.24157
Duffy C, Sorolla A, Wang E, et al., 2020. Honeybee venom and melittin suppress growth factor receptor activation in HER2-enriched and triple-negative breast cancer. NPJ Precis Oncol, 4:24. https://doi.org/10.1038/s41698-020-00129-0
Dutta S, Mahalanobish S, Saha S, et al., 2019. Natural products: an upcoming therapeutic approach to cancer. Food Chem Toxicol, 128:240–255. https://doi.org/10.1016/j.fct.2019.04.012
Emens LA, 2018. Breast cancer immunotherapy: facts and hopes. Clin Cancer Res, 24(3):511–520. https://doi.org/10.1158/1078-0432.Ccr-16-3001
Fulda S, 2015. Promises and challenges of Smac mimetics as cancer therapeutics. Clin Cancer Res, 21(22):5030–5036. https://doi.org/10.1158/1078-0432.ccr-15-0365
Garbayo E, Pascual-Gil S, Rodríguez-Nogales C, et al., 2020. Nanomedicine and drug delivery systems in cancer and regenerative medicine. Wiley Interdiscip Rev Nanomed Nanobiotechnol, 12(5):e1637. https://doi.org/10.1002/wnan.1637
Garcia IJP, de Oliveira GC, de Moura Valadares JM, et al., 2019. New bufadienolides extracted from Rhinella marina inhibit Na,K-ATPase and induce apoptosis by activating caspases 3 and 9 in human breast and ovarian cancer cells. Steroids, 152:108490. https://doi.org/10.1016/j.steroids.2019.108490
Guo ZR, 2016. Artemisinin anti-malarial drugs in China. Acta Pharm Sin B, 6(2):115–124. https://doi.org/10.1016/j.apsb.2016.01.008
Hao Y, Baker D, Ten Dijke P, 2019. TGF-β-mediated epithelial-mesenchymal transition and cancer metastasis. Int J Mol Sci, 20(11):2767. https://doi.org/10.3390/ijms20112767
Hassan MSU, Ansari J, Spooner D, et al., 2010. Chemotherapy for breast cancer (Review). Oncol Rep, 24(5): 1121–1131. https://doi.org/10.3892/or_00000963
He Q, Xue SY, Tan YQ, et al., 2019. Dual inhibition of Akt and ERK signaling induces cell senescence in triple-negative breast cancer. Cancer Lett, 448:94–104. https://doi.org/10.1016/j.canlet.2019.02.004
Hematyar M, Soleimani M, Es-Haghi A, et al., 2018. Synergistic co-delivery of doxorubicin and melittin using functionalized magnetic nanoparticles for cancer treatment: loading and in vitro release study by LC-MS/MS. Artif Cells Nanomed Biotechnol, 46(sup 3):S1226–S1235. https://doi.org/10.1080/21691401.2018.1536063
Hsia TC, Yu CC, Hsu SC, et al., 2014. Cantharidin induces apoptosis of H460 human lung cancer cells through mitochondria-dependent pathways. Int J Oncol, 45(1): 245–254. https://doi.org/10.3892/ijo.2014.2428
Hu W, Qi L, Tian YH, et al., 2015. Studies on the purification of polypeptide from sika antler plate and activities of antitumor. BMC Complement Altern Med, 15:328. https://doi.org/10.1186/s12906-015-0845-7
Huang PS, Wang XL, Liang XY, et al., 2019. Nano-, micro-, and macroscale drug delivery systems for cancer immunotherapy. Acta Biomater, 85:1–26. https://doi.org/10.1016/j.actbio.2018.12.028
Huang SY, Yao YD, Chen LL, et al., 2010. Anti-invasive and anti-metastasis effect of norcantharidin on high-metastatic human breast cancer cell lines. J Trop Med, 10(9):1034–1038 (in Chinese).
Huang Y, Liu Q, Liu K, et al., 2009. Suppression of growth of highly-metastatic human breast cancer cells by norcantharidin and its mechanisms of action. Cytotechnology, 59(3):201–208. https://doi.org/10.1007/s10616-009-9210-3
Huo YS, Huo H, Zhang J, 2014. The contribution of deer velvet antler research to the modern biological medicine. Chin J Integr Med, 20(10):723–728. https://doi.org/10.1007/s11655-014-1827-1
Jeong MH, Lee CM, Lee SW, et al., 2013. Cordycepin-enriched Cordyceps militaris induces immunomodulation and tumor growth delay in mouse-derived breast cancer. Oncol Rep, 30(4):1996–2002. https://doi.org/10.3892/or.2013.2660
Jeong YJ, Choi Y, Shin JM, et al., 2014. Melittin suppresses EGF-induced cell motility and invasion by inhibiting PI3K/Akt/mTOR signaling pathway in breast cancer cells. Food Chem Toxicol, 68:218–225. https://doi.org/10.1016/j.fct.2014.03.022
Jiang ZW, Chi JH, Han BQ, et al., 2017. Preparation and pharmacological evaluation of norcantharidin-conjugated carboxymethyl chitosan in mice bearing hepatocellular carcinoma. Carbohydr Polym, 174:282–290. https://doi.org/10.1016/j.carbpol.2017.06.072
Jin L, Jin LS, Wu RJ, et al., 2021. Hirsutella sinensis fungus regulates CD8+ T cell exhaustion through involvement of T-bet/Eomes in the tumor microenvironment. Front Pharmacol, 11:612620. https://doi.org/10.3389/fphar.2020.612620
Jin XH, Wang JB, Zou S, et al., 2020. Cinobufagin triggers defects in spindle formation and cap-dependent translation in liver cancer cells by inhibiting the AURKA-mTOR-eIF4E axis. Am J Chin Med, 48(3):651–678. https://doi.org/10.1142/s0192415x20500330
Kampo S, Ahmmed B, Zhou TT, et al., 2019. Scorpion venom analgesic peptide, BmK AGAP inhibits stemness, and epithelial-mesenchymal transition by down-regulating PTX3 in breast cancer. Front Oncol, 9:21. https://doi.org/10.3389/fonc.2019.00021
Kato T, Nishimura K, Suparmin A, et al., 2021. Effects of cordycepin in Cordyceps militaris during its infection to silkworm larvae. Microorganisms, 9(4):681. https://doi.org/10.3390/microorganisms9040681
Kim KH, Seo HS, Choi HS, et al., 2011. Induction of apoptotic cell death by ursolic acid through mitochondrial death pathway and extrinsic death receptor pathway in MDA-MB-231 cells. Arch Pharm Res, 34(8):1363–1372. https://doi.org/10.1007/s12272-011-0817-5
Lan YL, Lou JC, Jiang XW, et al., 2019. A research update on the anticancer effects of bufalin and its derivatives (Review). Oncol Lett, 17(4):3635–3640. https://doi.org/10.3892/ol.2019.10062
Lee CT, Huang KS, Shaw JF, et al., 2020. Trends in the immunomodulatory effects of Cordyceps militaris: total extracts, polysaccharides and cordycepin. Front Pharmacol, 11:575704. https://doi.org/10.3389/fphar.2020.575704
Lee D, Lee WY, Jung K, et al., 2019. The inhibitory effect of cordycepin on the proliferation of MCF-7 breast cancer cells, and its mechanism: an investigation using network pharmacology-based analysis. Biomolecules, 9(9):414. https://doi.org/10.3390/biom9090414
Lee HJ, Burger P, Vogel M, et al., 2012. The nucleoside antagonist cordycepin causes DNA double strand breaks in breast cancer cells. Invest New Drugs, 30(5):1917–1925. https://doi.org/10.1007/s10637-012-9859-x
Lee S, Lee Y, Choi YJ, et al., 2014. Cyto-/genotoxic effects of the ethanol extract of Chan Su, a traditional Chinese medicine, in human cancer cell lines. J Ethnopharmacol, 152(2):372–376. https://doi.org/10.1016/j.jep.2014.01.023
Lei SY, Zheng RS, Zhang SW, et al., 2021. Global patterns of breast cancer incidence and mortality: a population-based cancer registry data analysis from 2000 to 2020. Cancer Commun (Lond), 41(11):1183–1194. https://doi.org/10.1002/cac2.12207
Li CY, 2012. Deer antler regeneration: a stem cell-based epimorphic process. Birth Defects Res C Embryo Today, 96(1):51–62. https://doi.org/10.1002/bdrc.21000
Li FJ, Hu JH, Ren X, et al., 2021. Toad venom: a comprehensive review of chemical constituents, anticancer activities, and mechanisms. Arch Pharm (Weinheim), 354(7): 2100060. https://doi.org/10.1002/ardp.202100060
Li J, Cai HW, Sun HH, et al., 2020. Extracts of Cordyceps sinensis inhibit breast cancer growth through promoting M1 macrophage polarization via NF-κB pathway activation. J Ethnopharmacol, 260:112969. https://doi.org/10.1016/j.jep.2020.112969
Li M, Wang XJ, Zhao Q, et al., 2020. Bufalin-induced cardiotoxicity: new findings into mechanisms. Chin J Nat Med, 18(7):550–560. https://doi.org/10.1016/s1875-5364(20)30065-0
Li SP, Li P, Dong TTX, et al., 2001. Anti-oxidation activity of different types of natural Cordyceps sinensis and cultured Cordyceps mycelia. Phytomedicine, 8(3):207–212. https://doi.org/10.1078/0944-7113-00030
Li W, Xie L, Chen Z, et al., 2010. Cantharidin, a potent and selective PP2A inhibitor, induces an oxidative stress-independent growth inhibition of pancreatic cancer cells through G2/M cell-cycle arrest and apoptosis. Cancer Sci, 101(5):1226–1233. https://doi.org/10.1111/j.1349-7006.2010.01523.x
Li XT, Li HC, Li CB, et al., 2010. Protective effects on mitochondria and anti-aging activity of polysaccharides from cultivated fruiting bodies of Cordyceps militaris. Am J Chin Med, 38(6):1093–1106. https://doi.org/10.1142/s0192415x10008494
Li XW, Chen CH, Dai Y, et al., 2019. Cinobufagin suppresses colorectal cancer angiogenesis by disrupting the endothelial mammalian target of rapamycin/hypoxia-inducible factor 1α axis. Cancer Sci, 110(5): 1724–1734. https://doi.org/10.1111/cas.13988
Li YF, Xiao YJ, Lin HP, et al., 2019. In vivo β-catenin attenuation by the integrin α5-targeting nano-delivery strategy suppresses triple negative breast cancer stemness and metastasis. Biomaterials, 188:160–172. https://doi.org/10.1016/j.biomaterials.2018.10.019
Li YL, Tian X, Liu XD, et al., 2018. Bufalin inhibits human breast cancer tumorigenesis by inducing cell death through the ROS-mediated RIP1/RIP3/PARP-1 pathways. Carcinogenesis, 39(5):700–707. https://doi.org/10.1093/carcin/bgy039
Li YL, Gong PC, Kong CC, et al., 2019. Bufalin engages in RIP1-dependent and ROS-dependent programmed necroptosis in breast cancer cells by targeting the RIP1/RIP3/PGAM5 pathway. Anticancer Drugs, 30(7):706–713. https://doi.org/10.1097/cad.0000000000000770
Liao LX, Song XM, Wang LC, et al., 2017. Highly selective inhibition of IMPDH2 provides the basis of antineuroinflammation therapy. Proc Natl Acad Sci USA, 114(29): E5986–E5994. https://doi.org/10.1073/pnas.1706778114
Lin SS, Lyu XD, Yu J, et al., 2016. MHP-1 inhibits cancer metastasis and restores topotecan sensitivity via regulating epithelial-mesenchymal transition and TGF-β signaling in human breast cancer cells. Phytomedicine, 23(10):1053–1063. https://doi.org/10.1016/j.phymed.2016.06.013
Lin YE, Chen YC, Lu KH, et al., 2021. Antidepressant-like effects of water extract of Cordyceps militaris (Linn.) Link by modulation of ROCK2/PTEN/Akt signaling in an unpredictable chronic mild stress-induced animal model. J Ethnopharmacol, 276:114194. https://doi.org/10.1016/j.jep.2021.114194
Liu DW, Chen ZW, 2009. The effects of cantharidin and cantharidin derivates on tumour cells. Anticancer Agents Med Chem, 9(4):392–396. https://doi.org/10.2174/1871520610909040392
Liu DW, Shi PG, Yin X, et al., 2012. Effect of norcantharidin on the human breast cancer Bcap-37 cells. Connect Tissue Res, 53(6):508–512. https://doi.org/10.3109/03008207.2012.694928
Liu JY, Geng XF, Hou JX, et al., 2021. New insights into M1/M2 macrophages: key modulators in cancer progression. Cancer Cell Int, 21:389. https://doi.org/10.1186/s12935-021-02089-2
Liu MJ, Wang HT, Liu LJ, et al., 2016. Melittin-MIL-2 fusion protein as a candidate for cancer immunotherapy. J Transl Med, 14:155. https://doi.org/10.1186/s12967-016-0910-0
Ma LN, Song B, Jin H, et al., 2012. Cinobufacini induced MDA-MB-231 cell apoptosis-associated cell cycle arrest and cytoskeleton function. Bioorg Med Chem Lett, 22(3): 1459–1463. https://doi.org/10.1016/j.bmcl.2011.11.095
Mediratta K, El-Sahli S, D’Costa V, et al., 2020. Current progresses and challenges of immunotherapy in triple-negative breast cancer. Cancers (Basel), 12(12):3529. https://doi.org/10.3390/cancers12123529
Mehta AK, Kadel S, Townsend MG, et al., 2021. Macrophage biology and mechanisms of immune suppression in breast cancer. Front Immunol, 12:643771. https://doi.org/10.3389/fimmu.2021.643771
Mir Hassani Z, Nabiuni M, Parivar K, et al., 2021. Melittin inhibits the expression of key genes involved in tumor microenvironment formation by suppressing HIF-1α signaling in breast cancer cells. Med Oncol, 38(7):77. https://doi.org/10.1007/s12032-021-01526-6
Mittal V, 2018. Epithelial mesenchymal transition in tumor metastasis. Annu Rev Pathol, 13:395–412. https://doi.org/10.1146/annurev-pathol-020117-043854
Moghaddam FD, Mortazavi P, Hamedi S, et al., 2020. Apoptotic effects of melittin on 4T1 breast cancer cell line is associated with up regulation of Mfn1 and Drp1 mRNA expression. Anticancer Agents Med Chem, 20(7):790–799. https://doi.org/10.2174/1871520620666200211091451
Morrish E, Brumatti G, Silke J, 2020. Future therapeutic directions for Smac-mimetics. Cells, 9(2):406. https://doi.org/10.3390/cells9020406
Mu XZ, Wang CC, 2018. Artemisinins—a promising new treatment for systemic lupus erythematosus: a descriptive review. Curr Rheumatol Rep, 20(9):55. https://doi.org/10.1007/s11926-018-0764-y
Nazim UM, Yin HH, Park SY, 2020. Downregulation of c-FLIP and upregulation of DR-5 by cantharidin sensitizes TRAIL-mediated apoptosis in prostate cancer cells via autophagy flux. Int J Mol Med, 46(1):280–288. https://doi.org/10.3892/ijmm.2020.4566
Nedeljković M, Damjanović A, 2019. Mechanisms of chemotherapy resistance in triple-negative breast cancer—how we can rise to the challenge. Cells, 8(9):957. https://doi.org/10.3390/cells8090957
Ni LQ, Chen LL, Huang X, et al., 2020. Combating COVID-19 with integrated traditional Chinese and Western medicine in China. Acta Pharm Sin B, 10(7):1149–1162. https://doi.org/10.1016/j.apsb.2020.06.009
Noonan AM, Bunch KP, Chen JQ, et al., 2016. Pharmacodynamic markers and clinical results from the phase 2 study of the SMAC mimetic birinapant in women with relapsed platinum-resistant or -refractory epithelial ovarian cancer. Cancer, 122(4):588–597. https://doi.org/10.1002/cncr.29783
Oršolić N, 2012. Bee venom in cancer therapy. Cancer Metastasis Rev, 31(1–2):173–194. https://doi.org/10.1007/s10555-011-9339-3
Ortiz E, Gurrola GB, Schwartz EF, et al., 2015. Scorpion venom components as potential candidates for drug development. Toxicon, 93:125–135. https://doi.org/10.1016/j.toxicon.2014.11.233
Osborne CK, Bardou V, Hopp TA, et al., 2003. Role of the estrogen receptor coactivator AIB1 (SRC-3) and HER-2/neu in tamoxifen resistance in breast cancer. J Natl Cancer Inst, 95(5):353–361. https://doi.org/10.1093/jnci/95.5.353
Pan MS, Cao J, Fan YZ, 2020. Insight into norcantharidin, a small-molecule synthetic compound with potential multitarget anticancer activities. Chin Med, 15:55. https://doi.org/10.1186/s13020-020-00338-6
Pan YH, Zheng Q, Ni WT, et al., 2019. Breaking glucose transporter 1/pyruvate kinase M2 glycolytic loop is required for cantharidin inhibition of metastasis in highly metastatic breast cancer. Front Pharmacol, 10:590. https://doi.org/10.3389/fphar.2019.00590
Paterson RRM, 2008. Cordyceps—a traditional Chinese medicine and another fungal therapeutic biofactory? Phytochemistry, 69(7):1469–1495. https://doi.org/10.1016/j.phytochem.2008.01.027
Qi FH, Li AY, Inagaki Y, et al., 2011. Antitumor activity of extracts and compounds from the skin of the toad Bufo bufo gargarizans Cantor. Int Immunopharmacol, 11(3):342–349. https://doi.org/10.1016/j.intimp.2010.12.007
Qi WC, Zhou XT, Wang JQ, et al., 2020. Cordyceps sinensis polysaccharide inhibits colon cancer cells growth by inducing apoptosis and autophagy flux blockage via mTOR signaling. Carbohydr Polym, 237:116113. https://doi.org/10.1016/j.carbpol.2020.116113
Quan XG, Kwak BS, Lee JY, et al., 2020. Cordyceps militaris induces immunogenic cell death and enhances antitumor immunogenic response in breast cancer. Evid Based Complement Alternat Med, 2020:9053274. https://doi.org/10.1155/2020/9053274
Rady I, Siddiqui IA, Rady M, et al., 2017. Melittin, a major peptide component of bee venom, and its conjugates in cancer therapy. Cancer Lett, 402:16–31. https://doi.org/10.1016/j.canlet.2017.05.010
Raveendran R, Chen F, Kent B, et al., 2020. Estronedecorated polyion complex micelles for targeted melittin delivery to hormone-responsive breast cancer cells. Biomacromolecules, 21(3): 1222–1233. https://doi.org/10.1021/acs.biomac.9b01681
Rowinsky EK, 2005. Targeted induction of apoptosis in cancer management: the emerging role of tumor necrosis factor-related apoptosis-inducing ligand receptor activating agents. J Clin Oncol, 23(36):9394–9407. https://doi.org/10.1200/jco.2005.02.2889
Sarfo-Poku C, Eshun O, Lee KH, 2016. Medical application of scorpion venom to breast cancer: a mini-review. Toxicon, 122:109–112. https://doi.org/10.1016/j.toxicon.2016.09.005
Seshacharyulu P, Pandey P, Datta K, et al., 2013. Phosphatase: PP2A structural importance, regulation and its aberrant expression in cancer. Cancer Lett, 335(1):9–18. https://doi.org/10.1016/j.canlet.2013.02.036
Shou LM, Zhang QY, Li W, et al., 2013. Cantharidin and norcantharidin inhibit the ability of MCF-7 cells to adhere to platelets via protein kinase C pathway-dependent downregulation of α2 integrin. Oncol Rep, 30(3):1059–1066. https://doi.org/10.3892/or.2013.2601
Singh SK, Singh S, Lillard JW, et al., 2017. Drug delivery approaches for breast cancer. Int J Nanomedicine, 12:6205–6218. https://doi.org/10.2147/IJN.S140325
Soman NR, Baldwin SL, Hu G, et al., 2009. Molecularly targeted nanocarriers deliver the cytolytic peptide melittin specifically to tumor cells in mice, reducing tumor growth. J Clin Invest, 119(9):2830–2842. https://doi.org/10.1172/JCI38842
Son DJ, Lee JW, Lee YH, et al., 2007. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharmacol Ther, 115(2):246–270. https://doi.org/10.1016/j.pharmthera.2007.04.004
Song JJ, Wang YW, Teng MY, et al., 2016. Cordyceps militaris induces tumor cell death via the caspase-dependent mitochondrial pathway in HepG2 and MCF-7 cells. Mol Med Rep, 13(6):5132–5140. https://doi.org/10.3892/mmr.2016.5175
Song MY, Wang XF, Luo YJ, et al., 2020. Cantharidin suppresses gastric cancer cell migration/invasion by inhibiting the PI3K/Akt signaling pathway via CCAT1. Chem Biol Interact, 317:108939. https://doi.org/10.1016/j.cbi.2020.108939
Song XF, Zhang GJ, Sun AP, et al., 2012. Scorpion venom component III inhibits cell proliferation by modulating NF-κB activation in human leukemia cells. Exp Ther Med, 4(1):146–150. https://doi.org/10.3892/etm.2012.548
Song XZ, Zhang CW, Zhao MK, et al., 2015. Steroid receptor coactivator-3 (SRC-3/AIB1) as a novel therapeutic target in triple negative breast cancer and its inhibition with a phospho-bufalin prodrug. PLoS ONE, 10(10):e0140011. https://doi.org/10.1371/journal.pone.0140011
Sui ZG, Zhang LH, Huo YS, et al., 2014. Bioactive components of velvet antlers and their pharmacological properties. J Pharm Biomed Anal, 87:229–240. https://doi.org/10.1016/j.jpba.2013.07.044
Sung H, Ferlay J, Siegel RL, et al., 2021. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin, 71(3):209–249. https://doi.org/10.3322/caac.21660
Tariq M, Zhang JQ, Liang GK, et al., 2017. Macrophage polarization: anti-cancer strategies to target tumor-associated macrophage in breast cancer. J Cell Biochem, 118(9):2484–2501. https://doi.org/10.1002/jcb.25895
Tian X, Yin HZ, Zhang SC, et al., 2014. Bufalin loaded biotinylated chitosan nanoparticles: an efficient drug delivery system for targeted chemotherapy against breast carcinoma. Eur J Pharm Biopharm, 87(3):445–453. https://doi.org/10.1016/j.ejpb.2014.05.010
Tu GG, Zhan JF, Lv QL, et al., 2014. Synthesis and antiproliferative assay of norcantharidin derivatives in cancer cells. Med Chem, 10(4):376–381. https://doi.org/10.2174/15734064113099990037
Tuli HS, Sharma AK, Sandhu SS, et al., 2013. Cordycepin: a bioactive metabolite with therapeutic potential. Life Sci, 93(23):863–869. https://doi.org/10.1016/j.lfs.2013.09.030
Vahed SZ, Salehi R, Davaran S, et al., 2017. Liposome-based drug co-delivery systems in cancer cells. Mater Sci Eng C Mater Biol Appl, 71:1327–1341. https://doi.org/10.1016/j.msec.2016.11.073
van Vaerenbergh M, Cardoen D, Formesyn EM, et al., 2013. Extending the honey bee venome with the antimicrobial peptide apidaecin and a protein resembling wasp antigen 5. Insect Mol Biol, 22(2):199–210. https://doi.org/10.1111/imb.12013
Waks AG, Winer EP, 2019. Breast cancer treatment: a review. JAMA, 321(3):288–300. https://doi.org/10.1001/jama.2018.19323
Wang DT, Berg D, Ba HX, et al., 2019. Deer antler stem cells are a novel type of cells that sustain full regeneration of a mammalian organ—deer antler. Cell Death Dis, 10(6):443. https://doi.org/10.1038/s41419-019-1686-y
Wang KL, Chen Q, Shao YY, et al., 2021. Anticancer activities of TCM and their active components against tumor metastasis. Biomed Pharmacother, 133:111044. https://doi.org/10.1016/j.biopha.2020.111044
Wang RP, Huang SR, Zhou JY, et al., 2014. Synergistic interaction between melittin and chemotherapeutic agents and their possible mechanisms: an experimental research. Chin J Integr Tradit Western Med, 34(2):224–229 (in Chinese). https://doi.org/10.7661/CJIM.2014.02.0224
Wang S, Fu JL, Hao HF, et al., 2021. Metabolic reprogramming by traditional Chinese medicine and its role in effective cancer therapy. Pharmacol Res, 170:105728. https://doi.org/10.1016/j.phrs.2021.105728
Wang Y, Lonard DM, Yu Y, et al., 2014. Bufalin is a potent small-molecule inhibitor of the steroid receptor coactivators SRC-3 and SRC-1. Cancer Res, 74(5):1506–1517. https://doi.org/10.1158/0008-5472.CAN-13-2939
Wehbe R, Frangieh J, Rima M, et al., 2019. Bee venom: overview of main compounds and bioactivities for therapeutic interests. Molecules, 24(16):2997. https://doi.org/10.3390/molecules24162997
Wong SHM, Kong WY, Fang CM, et al., 2019. The TRAIL to cancer therapy: hindrances and potential solutions. Crit Rev Oncol Hematol, 143:81–94. https://doi.org/10.1016/j.critrevonc.2019.08.008
Xiao MZ, Tian JX, Zhou YN, et al., 2020. Efficacy of Huoxiang Zhengqi dropping pills and Lianhua Qingwen granules in treatment of COVID-19: a randomized controlled trial. Pharmacol Res, 161:105126. https://doi.org/10.1016/j.phrs.2020.105126
Xie SS, Spelmink L, Codemo M, et al., 2016. Cinobufagin modulates human innate immune responses and triggers antibacterial activity. PLoS ONE, 11 (8):e0160734. https://doi.org/10.1371/journal.pone.0160734
Xie X, Wu MY, Shou LM, et al., 2015. Tamoxifen enhances the anticancer effect of cantharidin and norcantharidin in pancreatic cancer cell lines through inhibition of the protein kinase C signaling pathway. Oncol Lett, 9(2):837–844. https://doi.org/10.3892/ol.2014.2711
Xu GG, Zhao HP, Xu JD, et al., 2021. Hard antler extract inhibits invasion and epithelial-mesenchymal transition of triple-negative and Her-2+ breast cancer cells by attenuating nuclear factor-κB signaling. J Ethnopharmacol, 269: 113705. https://doi.org/10.1016/j.jep.2020.113705
Xu J, Tan ZC, Shen ZY, et al., 2021. Cordyceps cicadae polysaccharides inhibit human cervical cancer hela cells proliferation via apoptosis and cell cycle arrest. Food Chem Toxicol, 148:111971. https://doi.org/10.1016/j.fct.2021.111971
Yan SC, Qu XJ, Xu CA, et al., 2012. Down-regulation of Cbl-b by bufalin results in up-regulation of DR4/DR5 and sensitization of TRAIL-induced apoptosis in breast cancer cells. J Cancer Res Clin Oncol, 138(8):1279–1289. https://doi.org/10.1007/s00432-012-1204-4
Yan SC, Liu YP, Qu XJ, et al., 2014a. Bufalin induced apoptosis of MCF-7 breast cancer cells by down-regulating the activity of AKT. J China Med Univ, 43(7): 598–601, 611 (in Chinese). https://doi.org/10.3969/j.issn.0258-4646.2014.07.006
Yan SC, Qu XJ, Xu L, et al., 2014b. Bufalin enhances TRAIL-induced apoptosis by redistributing death receptors in lipid rafts in breast cancer cells. Anticancer Drugs, 25(6):683–689. https://doi.org/10.1097/cad.0000000000000095
Yan SC, Jiao X, Li K, et al., 2016a. The mechanism of bufalin and paclitaxel synergistically suppress the proliferation of breast cancer cells. J Mod Oncol, 24(20): 3173–3176 (in Chinese). https://doi.org/10.3969/j.issn.1672-4992.2016.20.001
Yan SC, Jiao X, Li K, et al., 2016b. The mechanism of bufalin and cisplatin synergistically suppress the proliferation of breast cancer MCF-7 cells. Chin Clin Oncol, 21(12): 1057–1062 (in Chinese).
Yang Q, Yin YL, Yu GJ, et al., 2015. A novel protein with anti-metastasis activity on 4T1 carcinoma from medicinal fungus Cordyceps militaris. Int J Biol Macromol, 80: 385–391. https://doi.org/10.1016/j.ijbiomac.2015.06.050
Yang XQ, Lin P, Wang J, et al., 2021. Purification, characterization and anti-atherosclerotic effects of the polysaccharides from the fruiting body of Cordyceps militaris. Int J Biol Macromol, 181:890–904. https://doi.org/10.1016/j.ijbiomac.2021.04.083
Yin SL, Zhang H, Jia LN, et al., 2019. Apoptosis in breast cancer cells MCF-7 induced by norcantharidin through the Wnt/β-catenin signaling pathway. J Shenyang Med Coll, 21(6):500–504 (in Chinese). https://doi.org/10.16753/j.cnki.1008-2344.2019.06.003
Yong TQ, Zhang ML, Chen DL, et al., 2016. Actions of water extract from Cordyceps militaris in hyperuricemic mice induced by potassium oxonate combined with hypoxanthine. J Ethnopharmacol, 194:403–411. https://doi.org/10.1016/j.jep.2016.10.001
Yu CH, Kan SF, Pu HF, et al., 2008. Apoptotic signaling in bufalin- and cinobufagin-treated androgen-dependent and -independent human prostate cancer cells. Cancer Sci, 99(12):2467–2476. https://doi.org/10.1111/j.1349-7006.2008.00966.x
Yuan X, Gajan A, Chu Q, et al., 2018. Developing TRAIL/TRAIL death receptor-based cancer therapies. Cancer Metastasis Rev, 37(4):733–748. https://doi.org/10.1007/s10555-018-9728-y
Zhang C, Ma K, Li WY, 2019. Cinobufagin suppresses the characteristics of osteosarcoma cancer cells by inhibiting the IL-6-OPN-STAT3 pathway. Drug Des Devel Ther, 13:4075–4090. https://doi.org/10.2147/dddt.s224312
Zhang ED, Li B, 2004. Resources of Chinese Materia Medica and Conservation of Endangered Wild Animals and Plants. Shanghai Pujiang Education Press, Shanghai, China (in Chinese).
Zhang JH, Hong YJ, Xie PS, et al., 2020. Spatial lipidomics reveals anticancer mechanisms of bufalin in combination with cinobufagin in tumor-bearing mice. Front Pharmacol, 11:593815. https://doi.org/10.3389/fphar.2020.593815
Zhang P, Cui Z, Liu YS, et al., 2005. Quality evaluation of traditional Chinese drug toad venom from different origins through a simultaneous determination of bufogenins and indole alkaloids by HPLC. Chem Pharm Bull (Tokyo), 53(12):1582–1586. https://doi.org/10.1248/cpb.53.1582
Zhang S, Liu Y, Ye Y, et al., 2018. Bee venom therapy: potential mechanisms and therapeutic applications. Toxicon, 148:64–73. https://doi.org/10.1016/j.toxicon.2018.04.012
Zhang XM, Zhang BF, Zhang PH, et al., 2019. Norcantharidin regulates ERα signaling and tamoxifen resistance via targeting miR-873/CDK3 in breast cancer cells. PLoS ONE, 14(5):e0217181. https://doi.org/10.1371/journal.pone.0217181
Zhang Y, Lou YN, Wang JB, et al., 2020. Research status and molecular mechanism of the traditional Chinese medicine and antitumor therapy combined strategy based on tumor microenvironment. Front Immunol, 11:609705. https://doi.org/10.3389/fimmu.2020.609705
Zhao L, Yang GS, Bai H, et al., 2017. NCTD promotes Birinapant-mediated anticancer activity in breast cancer cells by downregulation of c-FLIP. Oncotarget, 8(16): 26886–26895. https://doi.org/10.18632/oncotarget.15848
Zhao XY, Wang XY, Wei QY, et al., 2020. Potency and selectivity of SMAC/DIABLO mimetics in solid tumor therapy. Cells, 9(4):1012. https://doi.org/10.3390/cells9041012
Zhao ZY, Li YD, Zhou LY, et al., 2021. Prevention and treatment of COVID-19 using traditional Chinese medicine: a review. Phytomedicine, 85:153308. https://doi.org/10.1016/j.phymed.2020.153308
Zheng KX, Li QL, Lin DD, et al., 2020. Peptidomic analysis of pilose antler and its inhibitory effect on triple-negative breast cancer at multiple sites. Food Funct, 11(9):7481–7494. https://doi.org/10.1039/D0FO01531H
Zhu L, Chen YX, Wei C, et al., 2018. Anti-proliferative and pro-apoptotic effects of cinobufagin on human breast cancer MCF-7 cells and its molecular mechanism. Nat Prod Res, 32(4):493–497. https://doi.org/10.1080/14786419.2017.1315575
Zhu YX, Zhu XD, Tang CJ, et al., 2021. Progress and challenges of immunotherapy in triple-negative breast cancer. Biochim Biophys Acta Rev Cancer, 1876(2): 188593. https://doi.org/10.1016/j.bbcan.2021.188593
Zou ZZ, Luo XY, Nie PP, et al., 2016. Inhibition of SRC-3 enhances sensitivity of human cancer cells to histone deacetylase inhibitors. Biochem Biophys Res Commun, 478(1):227–233. https://doi.org/10.1016/j.bbrc.2016.07.063
Acknowledgments
This work was supported by the Integrated Chinese and Western Medicine Key Research Project of the Health Commission of Hubei Province (No. 6, 2017) and the Key Research & Development Project of the Department of Science and Technology of Hubei (No. 2020BCB006), China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Chaochao YU conceived the main ideas and wrote this paper. Yi LI and Guopeng CHEN helped search the literature.
Chaoyan WU and Xiuping WANG illustrated figures. Yingwen ZHANG revised the manuscript. All authors have read and approved the final manuscript, and therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Chaochao YU, Yi LI, Guopeng CHEN, Chaoyan WU, Xiuping WANG, and Yingwen ZHANG declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Yu, C., Li, Y., Chen, G. et al. Bioactive constituents of animal-derived traditional Chinese medicinal materials for breast cancer: opportunities and challenges. J. Zhejiang Univ. Sci. B 23, 547–563 (2022). https://doi.org/10.1631/jzus.B2101019
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2101019