Abstract
Clustered regulatory interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 nuclease (Cas9), the third-generation genome editing tool, has been favored because of its high efficiency and clear system composition. In this technology, the introduced double-strand breaks (DSBs) are mainly repaired by non-homologous end joining (NHEJ) or homology-directed repair (HDR) pathways. The high-fidelity HDR pathway is used for genome modification, which can introduce artificially controllable insertions, deletions, or substitutions carried by the donor templates. Although high-level knock-out can be easily achieved by NHEJ, accurate HDR-mediated knock-in remains a technical challenge. In most circumstances, although both alleles are broken by endonucleases, only one can be repaired by HDR, and the other one is usually recombined by NHEJ. For gene function studies or disease model establishment, biallelic editing to generate homozygous cell lines and homozygotes is needed to ensure consistent phenotypes. Thus, there is an urgent need for an efficient biallelic editing system. Here, we developed three pairs of integrated selection systems, where each of the two selection cassettes contained one drug-screening gene and one fluorescent marker. Flanked by homologous arms containing the mutated sequences, the selection cassettes were integrated into the target site, mediated by CRISPR/Cas9-induced HDR. Positively targeted cell clones were massively enriched by fluorescent microscopy after screening for drug resistance. We tested this novel method on the amyloid precursor protein (APP) and presenilin 1 (PSEN1) loci and demonstrated up to 82.0% biallelic editing efficiency after optimization. Our results indicate that this strategy can provide a new efficient approach for biallelic editing and lay a foundation for establishment of an easier and more efficient disease model.
概要
目的
本研究拟基于同源介导修复 (HDR) 原理, 创建一种高效的双等位基因编辑系统
创新点
通过引入双药物和双荧光筛选系统, 提高双等位基因编辑效率的基础上, 进一步通过剪切肽缩短表达盒长度, 提高了 CRISPR/Cas9 双等位编辑的效率.
方法
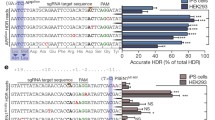
将嘌呤霉素抗性基因及博莱霉素抗性基因同绿色荧光蛋白及红色荧光蛋白分别配对组成筛选表达盒, 插入带有基因组同源臂区域和突变序列的供体载体中组成双等位编辑供体组合. 利用CRISPR/Cas9打靶系统在基因组靶位点处造成双链断裂, 经HDR方式将突变序列引入基因组中, 并经过荧光显微镜和双药物抗性筛选富集细胞克隆. 采用聚合酶链式反应 (PCR)、 限制性酶切和DNA测序对细胞基因组编辑阳性率进行鉴定和统计.
结论
成功构建了三套高效、 普适的基因组双等位敲入系统, 为基因组双等位精确编辑提供了新的思路. 此方法的建立可为通过CRISPR/Cas9双等位编辑鉴定疾病发生过程中的纯合基因功能奠定基础.
Similar content being viewed by others
References
Anzalone AV, Randolph PB, Davis JR, et al., 2019. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature, 576(7785):149–157. https://doi.org/10.1038/s41586-019-1711-4
Bétermier M, Bertrand P, Lopez BS, 2014. Is non-homologous end-joining really an inherently error-prone process? PLoS Genet, 10(1):e1004086. https://doi.org/10.1371/journal.pgen.1004086
Bibikova M, Beumer K, Trautman JK, et al., 2003. Enhancing gene targeting with designed zinc finger nucleases. Science, 300(5620):764. https://doi.org/10.1126/science.1079512
Boch J, Scholze H, Schornack S, et al., 2009. Breaking the code of DNA binding specificity of TAL-type III effectors. Science, 326(5959):1509–1512. https://doi.org/10.1126/science.1178811
Chen XY, Janssen JM, Liu J, et al., 2017. In trans paired nicking triggers seamless genome editing without double-stranded DNA cutting. Nat Commun, 8:657. https://doi.org/10.1038/s41467-017-00687-1
Chu VT, Weber T, Wefers B, et al., 2015. Increasing the efficiency of homology-directed repair for CRISPR-Cas9-induced precise gene editing in mammalian cells. Nat Biotechnol, 33(5):543–548. https://doi.org/10.1038/nbt.3198
Codner GF, Mianné J, Caulder A, et al., 2018. Application of long single-stranded DNA donors in genome editing: generation and validation of mouse mutants. BMC Biol, 16:70. https://doi.org/10.1186/s12915-018-0530-7
Cong L, Ran FA, Cox D, et al., 2013. Multiplex genome engineering using CRISPR/Cas systems. Science, 339(6121): 819–823. https://doi.org/10.1126/science.1231143
Eggenschwiler R, Moslem M, Fráguas MS, et al., 2016. Improved bi-allelic modification of a transcriptionally silent locus in patient-derived iPSC by Cas9 nickase. Sci Rep, 6:38198. https://doi.org/10.1038/srep38198
Evan G, 2012. Taking a back door to target Myc. Science, 335(6066):293–294. https://doi.org/10.1126/science.1217819
Frangoul H, Altshuler D, Cappellini MD, et al., 2021. CRISPR-Cas9 gene editing for sickle cell disease and β-thalassemia. N Engl J Med, 384(3):252–260. https://doi.org/10.1056/NEJMoa2031054
Hsu PD, Lander ES, Zhang F, 2014. Development and applications of CRISPR-Cas9 for genome engineering. Cell, 157(6):1262–1278. https://doi.org/10.1016/j.cell.2014.05.010
Jinek M, Chylinski K, Fonfara I, et al., 2012. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science, 337(6096):816–821. https://doi.org/10.1126/science.1225829
Koch B, Nijmeijer B, Kueblbeck M, et al., 2018. Generation and validation of homozygous fluorescent knock-in cells using CRISPR-Cas9 genome editing. Nat Protoc, 13(6): 1465–1487. https://doi.org/10.1038/nprot.2018.042
Li C, Brant E, Budak H, et al., 2021. CRISPR/Cas: a Nobel Prize award-winning precise genome editing technology for gene therapy and crop improvement. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 22(4):253–284. https://doi.org/10.1631/jzus.B2100009
Li XY, Bai YC, Cheng XZ, et al., 2018. Efficient SSA-mediated precise genome editing using CRISPR/Cas9. FEBS J, 285(18):3362–3375. https://doi.org/10.1111/febs.14626
Mali P, Yang LH, Esvelt KM, et al., 2013. RNA-guided human genome engineering via Cas9. Science, 339(6121): 823–826. https://doi.org/10.1126/science.1232033
Paquet D, Kwart D, Chen A, et al., 2016. Efficient introduction of specific homozygous and heterozygous mutations using CRISPR/Cas9. Nature, 533(7601):125–129. https://doi.org/10.1038/nature17664
Ren CH, Xu K, Liu ZT, et al., 2015. Dual-reporter surrogate systems for efficient enrichment of genetically modified cells. Cell Mol Life Sci, 72(14):2763–2772. https://doi.org/10.1007/s00018-015-1874-6
Saleh-Gohari N, Helleday T, 2004. Conservative homologous recombination preferentially repairs DNA double-strand breaks in the S phase of the cell cycle in human cells. Nucleic Acids Res, 32(12):3683–3688. https://doi.org/10.1093/nar/gkh703
Savic N, Ringnalda FC, Lindsay H, et al., 2018. Covalent linkage of the DNA repair template to the CRISPR-Cas9 nuclease enhances homology-directed repair. eLife, 7: e33761. https://doi.org/10.7554/eLife.33761
Scully R, Panday A, Elango R, et al., 2019. DNA doublestrand break repair-pathway choice in somatic mammalian cells. Nat Rev Mol Cell Biol, 20(11):698–714. https://doi.org/10.1038/s41580-019-0152-0
Shao SM, Ren CH, Liu ZT, et al., 2017. Enhancing CRISPR/Cas9-mediated homology-directed repair in mammalian cells by expressing Saccharomyces cerevisiae Rad52. Int J Biochem Cell Biol, 92:43–52. https://doi.org/10.1016/j.biocel.2017.09.012
Sur IK, Hallikas O, Vähärautio A, et al., 2012. Mice lacking a Myc enhancer that includes human SNP rs6983267 are resistant to intestinal tumors. Science, 338(6112):1360–1363. https://doi.org/10.1126/science.1228606
Wang G, Yang LH, Grishin D, et al., 2017. Efficient, footprint-free human iPSC genome editing by consolidation of Cas9/CRISPR and piggyBac technologies. Nat Protoc, 12(1):88–103. https://doi.org/10.1038/nprot.2016.152
Wu Y, Xu K, Ren CH, et al., 2017. Enhanced CRISPR/Cas9-mediated biallelic genome targeting with dual surrogate reporter-integrated donors. FEBS Lett, 591(6):903–913. https://doi.org/10.1002/1873-3468.12599
Xi LH, Schmidt JC, Zaug AJ, et al., 2015. A novel two-step genome editing strategy with CRISPR-Cas9 provides new insights into telomerase action and TERT gene expression. Genome Biol, 16:231. https://doi.org/10.1186/s13059-015-0791-1
Yan NN, Sun YS, Fang YY, et al., 2020. A universal surrogate reporter for efficient enrichment of CRISPR/Cas9-mediated homology-directed repair in mammalian cells. Mol Ther Nucleic Acids, 19:775–789. https://doi.org/10.1016/j.omtn.2019.12.021
Yang LH, Guell M, Byrne S, et al., 2013. Optimization of scarless human stem cell genome editing. Nucleic Acids Res, 41(19):9049–9061. https://doi.org/10.1093/nar/gkt555
Ye L, Wang JM, Tan YT, et al., 2016. Genome editing using CRISPR-Cas9 to create the HPFH genotype in HSPCs: an approach for treating sickle cell disease and β-thalassemia. Proc Natl Acad Sci USA, 113(38):10661–10665. https://doi.org/10.1073/pnas.1612075113
Zhang BH, 2021. CRISPR/Cas gene therapy. J Cell Physiol, 236(4):2459–2481. https://doi.org/10.1002/jcp.30064
Zhang JP, Li XL, Li GH, et al., 2017. Efficient precise knockin with a double cut HDR donor after CRISPR/Cas9-mediated double-stranded DNA cleavage. Genome Biol, 18:35. https://doi.org/10.1186/s13059-017-1164-8
Zhao X, Wei CW, Li JJ, et al., 2017. Cell cycle-dependent control of homologous recombination. Acta Biochim Biophys Sin, 49(8):655–668. https://doi.org/10.1093/abbs/gmx055
Zuo EW, Sun YD, Wei W, et al., 2019. Cytosine base editor generates substantial off-target single-nucleotide variants in mouse embryos. Science, 364(6437):289–292. https://doi.org/10.1126/science.aav9973
Acknowledgments
This work was supported by the National Science and Technology Major Project of China (No. 2018ZX08010-09B), the Swedish Research Council (No. NE 2016-04458), and the Ragnar Söderberg Foundation (No. M21/17), Sweden. We would like to thank all the members of Professor ZHANG’s lab for their excellent technical assistance and helpful discussions.
Author information
Authors and Affiliations
Contributions
Xinyi LI and Zhiying ZHANG conceived and supervised the project. Xinyi LI, Bing SUN, Hongrun QIAN, and Jinrong MA conducted experiments and data analysis. Xinyi LI wrote the manuscript and edited it in collaboration with Magdalena PAOLINO. All authors have read and approved the final manuscript, and therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Corresponding author
Ethics declarations
Xinyi LI, Bing SUN, Hongrun QIAN, Jinrong MA, Magdalena PAOLINO, and Zhiying ZHANG declare that they have no conflict of interests.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Supplementary information
Figs. S1 and S2
Supplementary information
Rights and permissions
About this article
Cite this article
Li, X., Sun, B., Qian, H. et al. A high-efficiency and versatile CRISPR/Cas9-mediated HDR-based biallelic editing system. J. Zhejiang Univ. Sci. B 23, 141–152 (2022). https://doi.org/10.1631/jzus.B2100196
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2100196