Abstract
Chromium is of great concern in the vitrification of high-level nuclear waste sludges because it forms separate crystallites in the molten glass. Inadequate removal of chromium from sludges could result in the production of an unacceptably large volume of HLW glass. Alkaline oxidative leaching is considered one of the pretreatment strategies to remove chromium before the vitrification.
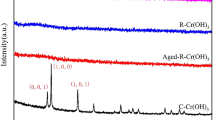
In this study, Cr(III) hydroxide solids were prepared under different conditions and characterized by EXAFS and IR. The rate of oxidation of the solids by hydrogen peroxide in alkaline solutions was studied by UV absorption spectroscopy. EXAFS and IR experiments indicate that the degree of oligomerization in the Cr(III) hydroxide solids increases with the increase in the concentration of NaOH in solution, the aging temperature and the aging time. The rate of oxidation of the solids follows the same order previously observed for Cr(III) oligomers in solution, i.e., species with higher degree of oligomerization are oxidized more slowly.
Similar content being viewed by others
References
B. M. Rapko, G. J. Lumetta, and M. J. Wagner, Washing and Caustic Leaching of Hanford Tank Sludges: Results of FY 1995 Studies. PNNL-10712, Pacific Northwest National Laboratory, Richland, WA, 1995.
J. L. Swanson, Clean Option: An Alternative Strategy for Hanford Tank Waste Remediation. Volume 2: Detailed Description of First Example Flowsheet. PNL-8288, Vol.2, Pacific Northwest National Laboratory, Richland, WA, 1993.
L. Spiccia, W. Marty, Inorg. Chem. 25, 266–271 (1987).
L. Spiccia, H. Stoeckli-Evans, W. Marty, R. Giovanoli, Inorg. Chem. 26, 474–482 (1987).
L. Rao, Z. Zhang, J. I. Friese, B. Ritherdon, S. B. Clark, N. J. Hess, D. Rai, J. Chem. Soc. Dalton Trans., 267–274 (2002).
J. I. Friese, B. Ritherdon, S. B. Clark, Z. Zhang, L. Rao, D. Rai, Anal. Chem., 74, 2977–2984 (2002).
D. Rai, N. J. Hess, L. Rao, Z. Zhang, A. R. Felmy, D. A. Moore, S. B. Clark, G. J. Lumetta, J. Sol. Chem. 31, 343–367 (2002).
G. G. Li, F. Bridges, C. W. Booth, Phys. Rev. B, 52, 6332 (1995).
S. I. Zabinsky, J. J. Rehr, A. Ankudinov, R. C. Albers, M. J. Eller, Phys. Rev. B, 52, 2995 (1995).
A. N. Christensen, P. Hansen, M. S. Lehmann, J. Solid State Chemistry, 21, 325–329 (1977); 19, 299-304 (1976).
Acknowledgments
This work was supported by the Environmental Management Science Program of the U.S. Department of Energy, Office of Science, Biological and Environmental Research, under Contract No. DE-AC03-76SF0098 at Lawrence Berkeley National Laboratory. The EXAFS experiments were conducted at the Stanford Synchrotron Radiation Laboratory, which is operated by the Department of Energy, Division of Chemical Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, L., Zhang, Z., Rai, D. et al. Characterization of Chromium(III) Hydroxide Solids and their Oxidation by Hydrogen Peroxide. MRS Online Proceedings Library 824, 290–295 (2004). https://doi.org/10.1557/PROC-824-CC6.5
Published:
Issue Date:
DOI: https://doi.org/10.1557/PROC-824-CC6.5