Abstract
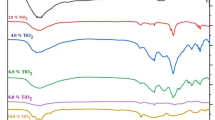
The infrared transmission spectra of four silicate glasses were investigated. By using blown glass films, 1–2 μm thick, detailed infrared transmission spectra were generated over the 4000–180 cm−1 range, both before and after the films were exposed to water. The water had little effect on the spectra of the 70SiO2–20Na2O–10Al2O3 (mol %) and Pyrex compositions, but had a large effect on the spectra of the 70SiO2–30Na2O (mol %) and Corning 015 compositions. The Si–O nonbridged stretching band at ∼950 cm−1 and a largely overlooked bending band at ∼600 cm−1 were the bands most sensitive to hydration in the 70/30 and 015 compositions. Changes were also seen in the Si–O–Si bridged stretching bands at ∼1050 cm−1 and ∼770 cm−1. The water, however, had no effect on the dominant Si–O–Si bending band at 460 cm−1. It was also discovered that the 70/30 and 015 films reacted with the atmosphere to form a carbonate layer on their surface. This carbonate accounted for the 1450 cm−1 and 230 cm−1 bands seen in their infrared transmission spectra.
Similar content being viewed by others
References
H. Tomozawa and M. Tomozawa, J. Non-Cryst. Solids 109, 311 (1989).
B. H. Olbert and R. H. Doremus, J. Am. Ceram. Soc. 66 (3), 163 (1983).
H. Dunken and R. H. Doremus, J. Non-Cryst. Solids 92, 61 (1987).
R. F. Bartholomew, B. L. Butler, H. L. Hoover, and C. K. Wu, J. Am. Ceram. Soc. 63 (9–10), 481 (1980).
L. L. Hench, J. Non-Cryst. Solids 19, 27 (1975).
D.E. Clark, M.F. Dilmore, E.C. Ethridge, and L.L. Hench, J. Am. Ceram. Soc. 59 (1–2), 62 (1976).
D.E. Clark, M.F. Dilmore, E.C. Ethridge, and L.L. Hench, Glass Technol. 18 (4), 121 (1977).
M. F. Dilmore, D. E. Clark, and L. L. Hench, J. Am. Ceram. Soc. 61 (9–10), 439 (1978).
L. A. Zellmer and W. B. White, in Scientific Basis for Nuclear Waste Management VIII, edited by C. M. Jantzen, J. A. Stone, and R. C. Ewing (Mater. Res. Soc. Symp. Proc. 44, Pittsburgh, PA, 1985, p. 73.
J.R. Sweet and W.B. White, Phys. Chem. Glasses 10 (6), 246 (1969).
R. Hanna and G-J. Su, J. Am. Ceram. Soc. 47 (12), 597 (1964).
V. A. Florinskaya, in The Structure of Glass (Consultants Bureau, New York, 1960), Vol. 2, p. 154.
H. M. Heaton and H. Moore, J. Soc. Glass Technol. 41, 28T (1957).
P. E. Jellyman and J. P. Procter, J. Soc. Glass Technol. 39, 173T (1955).
J.W. Park and H. Chen, J. Non-Cryst. Solids 40, 515 (1980).
J. R. Ferraro and M. H. Manghnani, J. Appl. Phys. 43 (11), 4595 (1972).
J. R. Ferraro, M. H. Manghnani, and A. Quattrochi, Phys. Chem. Glasses 13 (4), 116 (1972).
D. E. Day and G. E. Rindone, J. Am. Ceram. Soc. 45 (10), 489 (1962).
V. A. Kolesova, in The Structure of Glass (Consultants Bureau, New York, 1960), Vol. 2, p. 177.
N. J. Harrick, Appl. Optics 10 (10), 2344 (1971).
N. P. Bansal and R. H. Doremus, Handbook of Glass Properties (Academic Press, New York, 1986).
M. Prassas, J. Phalippou, L.L. Hench, and J. Zarzycki, J. Non-Cryst. Solids 48, 79 (1982).
Sadtler Research Laboratories, Sadder High Resolution Spectra of Inorganic and Related Compounds (Sadtler Research Laboratories Inc., Philadelphia, PA, 1965).
R. Hanna, J. Phys. Chem. 69 (11), 3846 (1965).
Joint Committee on Powder Diffraction Standards, Powder Diffraction File (JC on PDS, Philadelphia, PA, 1974).
R. H. Doremus, in Materials Stability and Environmental Degradation, edited by A. Barkatt, E.D. Verink Jr., and L.R. Smith (Mater. Res. Soc. Symp. Proc. 125, Pittsburgh, PA, 1988), p. 177.
R.H. Doremus, J. Non-Cryst. Solids 41, 145 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Husung, R.D., Doremus, R.H. The infrared transmission spectra of four silicate glasses before and after exposure to water. Journal of Materials Research 5, 2209–2217 (1990). https://doi.org/10.1557/JMR.1990.2209
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1990.2209