Abstract
Background: The interplay between state- and trait-related disruptions in structural networks remains unclear in bipolar disorder (BD), but graph theory can offer insights into global and local network changes. We sought to use diffusion-tensor imaging (DTI) and graph theory approaches to analyze structural topological properties across distinct mood states and identify high-risk individuals by examining state- and trait-related impairments in BD.
Methods: We studied changes in white matter network among patients with BD and healthy controls, exploring relationships with clinical variables. Secondary analysis involved comparing patients with BD with unaffected people at high genetic risk for BD.
Results: We included 152 patients with BD, including 52 with depressive BD (DBD), 64 with euthymic BD (EBD) and 36 with manic BD (MBD); we also included 75 healthy controls. Secondary analyses involved 27 unaffected people at high genetic risk for BD. Patients with DBD and MBD exhibited significantly lower global efficiencies than those with EBD and healthy controls, with patients with DBD showing the lowest global efficiencies. In addition, patients with DBD displayed impaired local efficiency and normalized clustering coefficient (γ). At a global level, γ correlated negatively with depression and anxiety. Compared with healthy controls, and across mood states, patients with BD showed abnormal shortest path lengths in the frontolimbic circuit, a trend mirrored among those at high genetic risk for BD.
Limitations: Considerations include medication effects, absence of recorded BD episode counts and the cross-sectional nature of the study.
Conclusion: Mood-specific whole-brain network metrics could serve as potential biomarkers in BD for transitions between mood states. Moreover, these findings contribute to evidence of trait-related frontolimbic circuit irregularities, shedding light on underlying pathophysiological mechanisms in BD.
Introduction
Bipolar disorder (BD) typically features recurring cycles, with brief elevated mood periods followed by extended depressive episodes.1,2 Patients with BD display diverse patterns of progression, treatment response and clinical manifestations. Often, brief episodes of elevated mood in the early stages go unnoticed, resulting in delayed diagnosis and potentially inadequate treatment. Enhancing management requires a profound grasp of the neurobiology of BD and the development of early detection features. Certain features are linked to recurrent manic and depressive episodes, indicating their state-related presence during acute mood episodes. Conversely, some characteristics represent the trait of BD, manifested across various phases and among people at high genetic risk for BD.3–6
Conventional theories of brain function have traditionally centred on specialized domains responsible for cognitive and emotional functions,7 presuming BD emerges from aberrations within regions governing emotional regulation. Recent research, however, has expanded this understanding to include the role of large-scale circuits and networks.8–10 Anatomic wiring patterns in the brain are organized into networks, contributing to dynamic patterns of large-scale neural activity.11,12 These networks integrate sensory, associative and motor areas, supporting the complex features of human cognition and behaviour that span across different systems and modalities.13 Complex network analysis, known as graph theory, quantifies the topologies of network representations. It provides a rigorous mathematical framework for analyzing the topology of complex brain networks consisting of nodes (represented by brain regions) and edges (represented by physical connections between brain regions).10
The prevailing corpus of graph theory research related to BD lacks distinction regarding the particular mood states that define the expression of the disorder. Nonetheless, employing graph-theoretical methodologies on entire brain networks has unveiled a more intricate portrayal; BD has exhibited subtle reductions in network integration, as indicated by either the characteristic path length14 or equivalent global efficiency.3 However, the latter metric did not retain statistical significance under family-wise error correction.3 Disparities in whole-brain segregation, assessed through the clustering coefficient, have also been documented, albeit with contradictory trends (i.e., both diminished3,15 and elevated14 levels), potentially attributed to mixed emotional states. Specifically, Wang and colleagues16 found abnormal global properties in BD, including increased characteristic path length, and decreased global efficiency and local efficiency. Locally, patients with BD showed abnormal nodal parameters (e.g., nodal strength, nodal efficiency), predominantly in the parietal, orbitofrontal, occipital and cerebellar regions.16 The observed inconsistencies in global network patterns within BD research, stemming from the conflation of distinct affective states, may further underscore the potential vulnerability of BD’s global network properties to external modulatory factors.17 Some studies indicated that the subtle decreases in whole-brain integration observed in BD may reflect alterations to interhemispheric connectivity,3 as well as the disrupted connectivity of frontolimbic circuits.17,18
Given that emotional dysregulation is central to the phenomenological diagnosis of the illness, it is not surprising that contemporary neurobiological models of BD stress the importance of a frontolimbic network thought to serve affective processing.19–23 Several studies involving BD have implicated an abnormal constellation of regions in the frontolimbic circuit.24–28 Contemporary neurobiological theories propose that emotion is associated with frontolimbic regions — including the hippocampus and insula — that connect to anxiety and fear circuitry, such as the amygdala. These regions interact with cognitive control areas like the inferior frontal gyrus and anterior cingulate cortex.29,30 Supporting this notion, structural magnetic resonance imaging (sMRI) studies have identified morphological abnormalities in frontolimbic and subcortical structures.31–37 The agranular cortices proposed to embed interoceptive predictions comprise the prefrontal and limbic areas that are repeatedly implicated in BD.38–40
Studies on BD face numerous challenges, including the influence of various medication classes, comorbidity and potential secondary effects related to illness manifestation. Tackling these concerns can be accomplished through the examination of unaffected high-risk cohorts. Recent investigations of people at high genetic risk of BD report morphological differences in functionally related brain regions.4,34 Notably, studies on the structural network properties of those at high genetic risk of BD are scarce. One study found abnormal nodal properties in the global network among those at high genetic risk relative to healthy controls;41 however, this study had a small sample size. Furthermore, whether the frontolimbic circuit mediates the relationship between the underlying genetic vulnerability and the disease expression remains unknown. In addition, most studies have ignored the role of connections within the greater network by examining only a small subset of possible connections.
Few network studies have concurrently assessed diverse mood states to elucidate state- and trait-related aspects. Such clarity is pivotal for comprehending the disease’s pathophysiology, neural attributes linked to susceptibility to and recovery from acute mood episodes, and markers of treatment response. We aimed to explore how this framework can integrate diverse connectomic disruptions among both patients with BD and those at high genetic risk of BD, encompassing local and large-scale connectivity alterations. We sought to use graph theory to probe the state- and trait-related attributes of BD among patients (including those experiencing manic [MBD], depressed [DBD] or euthymic [EBD] episodes), compared with healthy controls. We also sought to conduct further evaluations among those at high genetic risk to substantiate the manifestation of disrupted connectivity in this context, while considering potential confounding effects of medication.
Methods
Participants
We recruited patients with BD from the psychiatric inpatient unit of the Department of Psychiatry of the First Affiliated Hospital of China Medical University and Shenyang Mental Health Centre in Shenyang, China. Two trained clinical psychiatrists determined diagnoses according to the Structured Clinical Interview for DSM-IV Axis I Disorders. All patients met the diagnostic criteria of the DSM-IV for BD without any other Axis I disorder. On the day of the scan, the patients were rated using the Hamilton Depression Rating Scale (HAMD), Hamilton Anxiety Rating Scale (HAMA) and Young Mania Rating Scale (YMRS), with different thresholds for patients with DBD (HAMD ≥ 20 and YMRS ≤ 5), MBD (YMRS ≥ 13 and HAMD ≤ 7) or EBD (HAMD ≤ 7 and YMRS ≤ 7).
We excluded people with BD in mixed episodes, those with substance or alcohol misuse or dependence; those with a concomitant major medical disorder; those with other Axis I disorders; those with substantial pathological changes (found via T1- and T2-weighted MRI); those with a history of head trauma with loss of consciousness for 5 minutes or longer; those with any neurologic disorder; those with any infectious disease, such as HIV/AIDS or severe acute respiratory syndrome; those with any MRI contraindications; and those with suboptimal imaging data quality.
We also recruited healthy controls without a diagnosis of BD, with the same exclusion criteria as that of the patients. We also recruited 27 first-degree relatives of patients with BD (i.e., at least 1 of their parents or children met the DSM-IV diagnostic criteria for BD). These participants must not have had previous or current DSM-IV Axis I disorders. In addition, we required both healthy controls and those at high genetic risk to have HAMD and YMRS scores of 4 or less.
MRI acquisition
We performed MRI using a 3.0 T GE Sigma system (General Electric) with a standard 8-channel head coil at the First Affiliated Hospital of China Medical University. We acquired T1 images using a 3-dimensional fast-spoiled gradient echo sequence with a repetition time of 7.1 ms, echo time of 3.2 ms, image matrix of 240 × 240, field of view of 240 × 240 mm2, 176 contiguous slices of 1 mm without gap and a voxel size of 1.0 × 1.0 × 1.0 mm3. We acquired diffusion tensor imaging (DTI) using a single short–spin echo-planar imaging sequence with a repetition time of 17 000 ms, echo time of 85.4 ms, image matrix of 120 × 120, field of view of 240 × 240 mm2, 65 contiguous slices of 2 mm without gap and 25 noncollinear directions (b = 1000 s/mm2), alongside an axial acquisition without diffusion weighting (b = 0) and a voxel size of 2.0 × 2.0 × 2.0 mm3.
MRI preprocessing
The DTI data set was preprocessed using PANDA.42 Data preprocessing included brain extraction, correction for eddy-current distortion and simple head motion, correction for the B-matrix, computation of the diffusion tensor and fractional anisotropy.
Network construction
We constructed the white matter network using PANDA.42 The procedure consists of 2 basic steps, the definition of nodes and the definition of edges.
Network node definition
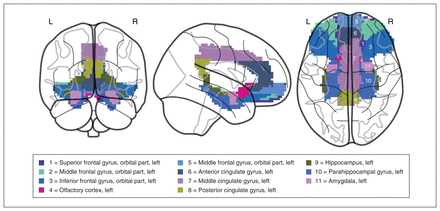
We used an automated anatomic labelling atlas to parcellate the cortical and subcortical structures of the brain into 90 regions (excluding the pons and cerebellum).43 Each region represents a node in the white matter network. First, the structural image (i.e., T1-weighted image) of each participant was coregistered to the b0 image in the native DTI space by linear transformation. Next, the coregistered image was nonlinearly normalized to the T1 template of ICBM152 in the Montreal Neurological Institute (MNI) space, resulting in a nonlinear transformation. Finally, an inverse transformation was applied to warp the automated anatomic labelling template from the MNI space to each participant’s native DTI space. The frontolimbic circuit involves key brain regions such as the superior, middle and inferior frontal gyri (orbital parts), the olfactory cortex, the cingulate gyrus (anterior, middle and posterior), the hippocampus, the parahippocampal gyrus and the amygdala. These interconnected regions play a vital role in emotional processing, cognitive control and regulation, contributing to understanding of emotional experiences and behaviours.30,44 On this basis, we used 22 brain regions (11 in each hemisphere) of the frontolimbic circuit as nodes to establish the white matter structural subnetwork according to automated anatomic labelling (Figure 1).
The schematic diagram of the frontolimbic circuit brain area.
Network edge definition
We implemented the reconstruction of whole-brain white matter tracts using a deterministic fibre assignment by continuous tracking algorithm.45 If the turn angle of a fibre tract was greater than 45° or if any voxel of a fibre’s fractional anisotropy was less than 0.2, the tracking procedure was terminated. We determined that an edge existed if at least 3 tracts had terminal points between 2 nodes. In this study, we defined the weight of the edges using the mean fractional anisotropy value (an index for evaluating fibre integrity of white matter46) of the white matter fibres between 2 nodes. We reconstructed frontolimbic white matter tracts according to the same procedure. Using these criteria, we obtained 90 × 90 and 22 × 22 fractional anisotropy–weighted metrics in the whole brain and the frontolimbic circuit network for each participant.
We used PANDA to check the quality of the registrations, such as the registration of DTI to T1, T1 to MNI space, and DTI to MNI space. We used TrackVis (www.trackvis.org) to check the quality of deterministic fibre tracking.
Network analysis
We chose an array of graph metrics to examine the brain graphs in terms of global and nodal functional integration and segregation. The threshold value for individual participants’ connectivity matrices varied from 0.10 to 0.30 with steps of 0.01. We computed the topological metrics of the white matter network using graph theory. We used GRETNA (www.nitrc.org/projects/gretna/) to calculate topological metrics.47
Graph metrics
We calculated global efficiency, local network efficiency, clustering coefficient (Cp), shortest path length (Lp), normalized clustering coefficient (γ), normalized shortest path length (λ), and small worldness (σ) for all of the individual networks as global network properties. We also calculated the Cp and Lp in the frontolimbic circuit network. The characteristic path length is the average of all the Lp values between each possible pair in the network. The global efficiency is the average inverse Lp.48 The local network efficiency is described as the mean of the local efficiencies for all of the nodes in the network.
Small-world properties
To investigate the small-world topological characteristics of the networks, we contrasted the network’s Cp and Lp values, denoted as Cp(real) and Lp(real), respectively, with those from 100 randomly generated networks, denoted as Cp(rand) and Lp(rand), respectively. We established these random networks using the random rewiring approach, maintaining identical node and edge counts as the actual networks, along with the degree distribution, while redistributing the associated weights. We then computed the normalized clustering coefficient
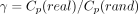
and the normalized characteristic path length.
Global efficiency
In addition to the conventional small-world parameters, a more biologically sensible property of brain networks is the network efficiency that can be described in terms of global efficiency (Eglob).48 In a complex network, the shortest path length is defined as the smallest sum of the edges throughout all possible paths from nodes i to j in the graph. The average global efficiency of the network (G) is defined as the reciprocal of the average of the reciprocals of shortest path length between pairs of nodes within the network,49
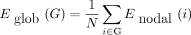
where
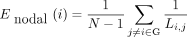
where Li<j is the shortest path length between node i and node j, and N is the number of nodes of the network G. The mean local efficiency across all nodes in the network is what defines the local network efficiency. Local network efficiency reflects the network’s overall resilience to faults and disruptions.
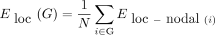
The efficiency metric offers several technical and conceptual benefits compared with the conventional Cp and Lp measures. It possesses the capability to encapsulate the efficiency of both local and global information exchange within a single measure.
Statistical analysis
We used 1-way analysis of covariance to compare demographic variables (except for sex, which was compared using the χ2 test) among the 4 groups (patients with EBD, DBD or MBD and healthy controls), followed by post hoc 2-sample t tests. We used MATLAB (Mathworks) software for statistical evaluations. We performed analysis of variance for between-group comparisons of global, regional and nodal network parameters. We further evaluated statistical differences between 2 groups using post hoc 2-sample t tests. When we compared group differences of those network parameters (global and regional nodal parameters), we considered age, sex and education years as nuisance covariates and regressed them out. We performed partial correlation analyses with age, sex and education years as covariates to investigate the relationship between white matter network metrics and clinical symptoms across all BD groups, with correction for false discovery rate (FDR). Statistical significance was set at p < 0.05 for the analysis of demographic and clinical characteristics as well as network metrics. For analyses involving nodal network metrics of the frontolimbic network, we applied an FDR correction for multiple comparisons (n = 22 tests; significance set to a corrected pFDR < 0.05). For the secondary analysis, we have described detailed information in Appendix 1, available at jpn.ca/lookup/doi/10.1503/jpn.230069/tab-related-content.
Ethics approval
This study was approved by the Medical Science Research Ethics Committee of The First Affiliated Hospital of China Medical University. After receiving a detailed description of the study, all participants provided written informed consent.
Results
We included 152 patients with BD — including 52 with DBD, 64 with EBD and 36 with MBD — and 75 healthy controls. All participants were right-handed, with ages ranging from 18 to 50 years. As shown in Table 1, most demographic variables were similar. However, there were significant differences in terms of education level and clinical measures among the 4 groups.
Participant demographic and clinical characteristics
Altered global connectome topology
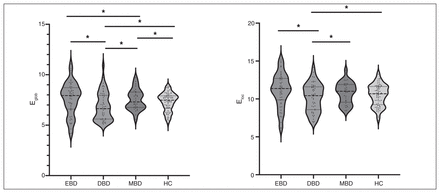
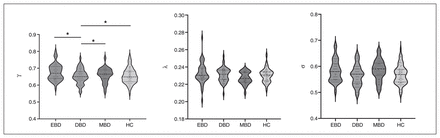
We observed statistically significant distinctions in global efficiency, local network efficiency and γ across the EBD, DBD, MBD and control groups (Figure 2 and Figure 3). Subsequent post hoc analyses showed that both the DBD and MBD groups exhibited notably reduced levels of global efficiency compared with the EBD and control groups. Moreover, the local network efficiency was significantly lower in the DBD group than the MBD group. Remarkably, the DBD group displayed significantly diminished local network efficiency and γ values compared with the other 3 groups. Conversely, no discernible differences in λ and σ were noted among the 4 cohorts.
Global (Eglob) and local (Eloc) network efficiency among patients with euthymic bipolar disorder (EBD), depressive bipolar disorder (DBD) or manic bipolar disorder (MBD), and healthy controls (HC). Each dot represents 1 participant. *p < 0.05.
Small-world properties among patients with euthymic bipolar disorder (EBD), depressive bipolar disorder (DBD) or manic bipolar disorder (MBD), and healthy controls (HC). Each dot represents 1 participant. *p < 0.05. γ = normalized clustering coefficient; λ = normalized shortest path length; σ = small worldness.
Similar frontolimbic circuit topology
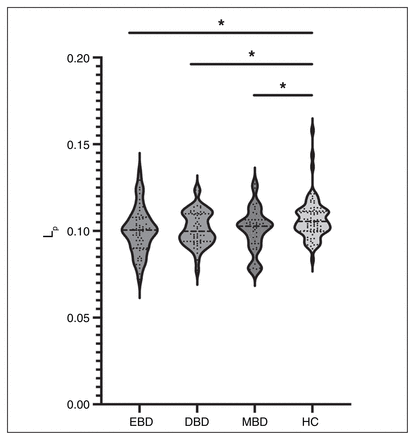
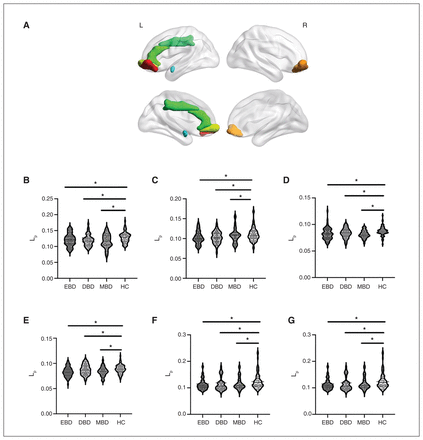
Figure 4 shows the topologies (Lp and Cp) of the frontolimbic circuit in the EBD, DBD, MBD and control groups. Post hoc comparisons showed that patients with EBD, DBD or MBD had significantly lower Lp values than healthy controls. There were no differences in Cp among the 4 groups. Figure 5 shows the brain regions with a significant group effect in the Lp among the 4 groups. We found a group effect in 6 regions, located in the bilateral orbital middle frontal gyrus, the left anterior and paracingulate gyrus, the left medial paracingulate gyrus, the left amygdala and the right lateral orbital superior frontal gyrus. Post hoc comparisons revealed that all these regions had a significantly lower Lp among patients with BD than among healthy controls.
Shortest path length (Lp) of the frontolimbic network among patients with euthymic bipolar disorder (EBD), depressive bipolar disorder (DBD) or manic bipolar disorder (MBD), and healthy controls (HC). Each dot represents 1 participant. *p < 0.05.
(A) The brain regions with significant group differences in nodal shortest path length (Lp) in the frontolimbic network among patients with euthymic bipolar disorder (EBD), depressive bipolar disorder (DBD) or manic bipolar disorder (MBD), and healthy controls (HC), including the (B) left orbital middle frontal gyrus, (C) right orbital middle frontal gyrus, (D) right medial orbital frontal gyrus, (E) left anterior cingulate and paracingulate gyrus, (F) left medial cingulate and paracingulate gyrus and (G) left amygdala. *p < 0.05. Each dot represents 1 participant.
Clinical correlates
We observed significant negative correlations between γ and the total score on the HAMD total (r = −0.269, p < 0.001) and HAMA (r = −0.237, p < 0.001). We also identified a positive correlation between γ and the severity of mania (r = 0.236, p = 0.025; Table 2); however, this association did not remain statistically significant after the FDR correction.
Correlations between the normalized clustering coefficient (γ) and clinical variables
Cross-validation among people at high genetic risk of BD
In our primary analyses, all patients with BD across the 3 emotional states exhibited lower Lp values within the frontolimbic circuit than healthy controls. Our secondary objective involved detecting the diminished Lp levels among people at high genetic risk of BD to eliminate the drug influence. The comprehensive list of variables and clinical details for this group, patients with EBD and healthy controls can be found in Appendix 1.
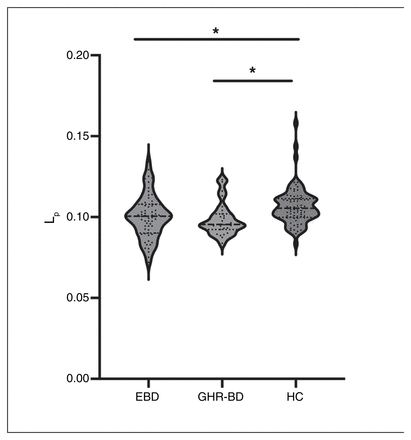
We did not observe significant disparities in global topological characteristics among the 3 groups. Intriguingly, both people at high genetic risk of BD and patients with EBD displayed notably lower Lp values within the frontolimbic network than healthy controls (Figure 6). Particularly noteworthy were the significantly lower Lp values in the lateral orbital middle frontal gyrus, the right olfactory cortex, and the right medial and paracingulate gyrus compared with the control group. In addition, the Lp value of the left hippocampus was lower among people at high genetic risk of BD than among participants in both the EBD and healthy control groups (Appendix 1, Figure S1).
Shortest path length (Lp) of the frontolimbic network among people at high genetic risk for bipolar disorder (GHR-BD), patients with euthymic bipolar disorder (EBD) and healthy controls (HC). Each dot represents 1 participant. *p < 0.05.
Discussion
Bipolar disorder is characterized by state-associated alterations in the overall structural connectomes, particularly trait-related irregularities within the frontolimbic circuit, across manic, depressed and euthymic states. Notably, the global efficiency among patients with DBD and MBD was significantly lower than among patients with EBD and healthy controls, with patients with DBD exhibiting the most pronounced reduction. In addition, patients with DBD demonstrated compromised local network efficiency and γ values compared with other groups. At a global level, γ was negatively correlated with the severity of depression and anxiety. However, the Lp differences between healthy controls and patients with BD, across the mood states, were notably prominent within the frontolimbic circuit, reflecting variations in network parameters. The presence of these consistent findings across diverse mood states, including patients with euthymic BD and people at high genetic risk of BD, underscores the trait-related abnormalities inherent to the disorder.
Only global efficiency was low among patients with MBD and DBD, which suggests, in part, that most global indices such as λ and σ remain intact in BD, aligning with previous literature.17 The disruption of extensive structural networks in BD reveals subtle attributes, primarily localized to specific regions and subnetworks within the frontal and limbic areas.17 The subtle decrease in comprehensive whole-brain integration potentially reflects perturbations in interactions within the prefrontal-limbic circuitry. Compared with the conspicuous disruptions observed in schizophrenia,50,51 the fundamental structural framework of the connectome remains largely preserved in BD. This is consistent with the cardinal differences in the phenotypes, specifically the relative interepisode preservation of cognition in BD.52
Within the conventional neuroscientific understanding of brain network architecture, there is often an emphasis on the inherent stability of white matter structural networks, positing their resilience to external perturbations.53 However, our empirical investigation, which delved into the intricate interplay of brain networks across varying emotional states, found discernible variations in the overall network properties. Beyond the conventional small-world parameters (Cp and Lp), an even more biologically relevant feature of brain networks is the sensitivity of the network efficiency, which encompasses global efficiency;54 although sensitivity maintains a relationship with Lp, this association is not strictly linear, which could be why global efficiency decreased in DBD and MBD. On the other hand, Lavagnino and colleagues55 found a reduced posterior corpus callosum volume among females with late-stage type I BD, compared with early-stage type I BD and healthy controls, after controlling for other confounding factors except episode frequency. Substantial evidence shows that white matter changes seem to be altered over the course of BD, and are especially associated with the number of episodes and severity.56–58 It is conceivable that participants in depressive or manic states may exhibit a higher propensity for mood cycling, or a distinct ratio of manic-to-depressive episodes, in contrast to participants who are in a euthymic state during the scan. In such a scenario, one could postulate that the observed variations in global efficiency may be attributed to the cumulative impact arising from the number of depressive or manic episodes throughout the course of the illness. Regrettably, we lacked data regarding the number of mood episodes, but this could be a prospective avenue for future investigation. Moreover, in this study, γ was negatively correlated with the symptoms of depression, which suggests that the impairment of global brain network segregation may be associated with the severity of depressive symptoms in BD, further corroborating the aforementioned hypothesis. This departure from the conventional supposition prompts a reconsideration of the intricate dynamics governing the relationship between emotional states and the structural underpinnings of brain global connectivity.
The human brain functions as a dynamically interconnected system, governed by 2 key organizational principles, namely segregation, denoted by Cp, and integration, evident through reduced Lp.49,59 Striking a harmonious balance between these attributes enhances the efficiency of higher-level cognitive processes.60 The decreased Lp of frontolimbic regions in mood-dysregulated BD indicates reductions in segregation and integration of the brain structure. This reduction in the length of morphological network pathways could potentially restrict long-range functional integration within brain networks. Disrupting the equilibrium between segregation and integration in the brain’s connectome results in diminished modularized information processing (i.e., reduced capacity to integrate distinct modules in the network) and decreased resilience of brain networks.61 Imbalanced functional separation and integration capabilities can result in modular information processing and fault tolerance. Compared with healthy controls, patients with BD in different mood states showed a decrease in Lp in the frontolimbic circuit. Similar to our results, Yang and colleagues61 reported a significant decrease in Lp among patients, suggesting that this decrease limits the integration of long-distance connections in the brain network. In line with our observations, Zhang and colleagues62 documented reduced Lp among patients with major depressive disorder, compared with controls. A recent study also reported that Lp was significantly lower among patients with BD with mania and depression; however, Cp did not change significantly and was not related to disease severity.63 In addition, some studies have shown that patients with BD have abnormal white matter structures in the limbic system under different emotional states, further supporting our findings.64–66 Given that emotional disorders are at the core of the phenomenological diagnosis of BD, the importance of frontolimbic neural networks is self-evident. The loss of the balance of separation and integration abilities of the frontolimbic network in BD is reflected in the non-optimization of the brain network topology and is unrelated to the emotional state of the disease.
Traditionally, the amygdala has held a pivotal role in conceptual frameworks concerning emotion regulation within the brain, primarily owing to its involvement in appraising threatening and other emotionally important stimuli.30,67–69 Nevertheless, responses to emotional stimuli are contingent on higher-order neural systems that govern affective regulation, characterized by prefrontal cortex regions.70,71 As extensively documented, the prefrontal cortex underpins an array of cognitive control and executive functions.72–74 In BD, functional underactivation has been observed within several prefrontal cortex regions — including the dorsolateral, ventrolateral, ventromedial, inferior frontal, and subgenual areas — during both emotional and cognitive control tasks.75 Conversely, heightened activation has been noted in the anterior cingulate cortex,29 although this may be contingent on task and mood dynamics, with instances of underactivation observed during cognitive control tasks among euthymic patients with BD.29 Parallel models of BD suggest that malfunctions within these frontolimbic neural circuits are at the core of the emotional and cognitive dysregulation that defines the disorder.
Interestingly, the frontolimbic circuit of patients with EBD and people at high genetic risk of BD showed similar trends in network parameters, suggesting that abnormalities in the frontolimbic structural network serve as biomarkers that mediate the relationship between underlying genetic susceptibility and disease expression. Patients with BD and adolescents at high genetic risk have been reported to have impaired structural networks.76,77 In recent years, studies of candidate genes and genome-wide association studies have received extensive attention. Notably, the insights provided by a meta-analysis support the heritability of neuroimaging abnormalities found in first-degree relatives of patients with BD.78 Specifically, genetic imaging studies on single-risk variants of BD consistently found that loci near the CACNA1Z, ANK3, 5-HTTlPR, NGR1 and BDNF genes had significant effects on the frontolimbic structure.79 In summary, the low Lp in the frontolimbic circuit of patients with BD and people at high genetic risk of BD, compared with healthy controls, suggests that the imbalance of the separation and integration ability of the frontolimbic circuit is an important factor in the occurrence of disease and is a relatively stable characteristic related to disease.
Limitations
Most of the patients in this study were taking medication. We did not collect the precise dose of medication taken by patients with BD on the day of the scan, although we conducted secondary analyses for people at high genetic risk of BD to account for the potential influence of medication. The effects of medication on brain white matter networks in BD are minimal. Interestingly, Hafeman and colleagues80 found that medication appeared to influence many sMRI studies, but had limited impact on DTI findings. Most studies show no significant effect of medication status (e.g., class, dose, number) on fractional anisotropy.81–84 Future research should also include patients with nonmedicated BD. We lacked sufficient data on the frequency of mood episodes. Addressing this gap presents an opportunity for future research to delve into a crucial aspect of the disorder and its potential implications. We conducted a cross-sectional study without a longitudinal observation. Longitudinal follow-ups of patients with BD and people at high genetic risk of BD should be conducted in the future.
Conclusion
In summary, both the DBD and MBD groups displayed notably reduced global efficiency compared with the EBD and healthy control groups, with the DBD group exhibiting the lowest global efficiency. Furthermore, patients with DBD exhibited compromised local efficiency and γ, and γ exhibited a negative correlation with depression and anxiety severity. Patients with BD across various mood states exhibited abnormal Lp within the frontolimbic circuit compared with healthy controls, a pattern also observed among people at high genetic risk of BD, underscoring the intrinsic trait-related anomalies inherent in the disorder. Identifying mood-specific changes throughout the entire brain in BD could potentially provide biomarkers for transitioning between emotional states. In addition, these findings add to the accumulating evidence of enduring frontolimbic circuit disruptions linked to traits, possibly indicating fundamental pathophysiological mechanisms in BD.
Footnotes
↵* These authors contributed equally to this work.
Competing interests: None declared.
Contributors: Yifan Chen, Xizhe Zhang, Yanqing Tang and Fei Wang contributed to the conception and design of the work. Pengfei Zhao, Chunyu Pan, Miao Chang, Jia Duan and Yange Wei contributed to data acquisition, analysis and interpretation. Yifan Chen and Fei Wang drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Data sharing: The authors are committed to the principles of transparency and openness within the scientific community. To that end, they pledge to provide all data and materials related to this study to enable replication and further analysis by peers.
Funding: The authors were supported by research grants from the National Science Fund for Distinguished Young Scholars (no. 81725005 to Fei Wang), National Natural Science Foundation of China (no. 81571311 to Yanqing Tang, no. 81571331 to Fei Wang), National Key Research and Development Program (no. 2018YFC1311604 to Yanqing Tang, no. 2016YFC1306900 to Yanqing Tang, no. 2016YFC0904300 to Fei Wang), National High Tech Development Plan (no. 863; 2015AA020513 to Fei Wang), Liaoning Science and Technology Project (no. 2015225018 to Yanqing Tang), Liaoning Education Foundation (Pandeng Scholar to Fei Wang), Innovation Team Support Plan of Higher Education of Liaoning Province (no. LT2017007 to Fei Wang), Major Special Construction Plan of China Medical University (no. 3110117059 to Fei Wang).
- Received May 9, 2023.
- Revision received August 10, 2023.
- Revision received September 12, 2023.
- Accepted September 14, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/