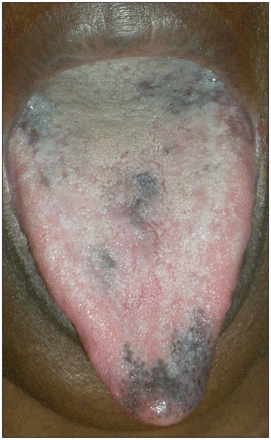
A 47-year-old black woman with chronic hepatitis C (genotype 4) was referred for hyperpigmentation of the tongue. She was a nonsmoker. Eight weeks after treatment with weekly injections of subcutaneous pegylated interferon α-2a and daily oral ribavirin was started for hepatitis C, asymptomatic dark brown patches developed on the dorsum and the apex of her tongue (Figure 1). No other sites were involved. Laboratory examination showed neutropenia with a neutrophil count of 0.88 (normal 1.5–7.0) × 109/L. Investigations including liver enzymes, cortisol and thyroid-stimulating hormone were normal. In the absence of an alternative explanation, an adverse effect of treatment was suspected. The treatment course with interferon and ribavirin was continued until its completion at 48 weeks. During this time, there was a slight increase in hyperpigmentation.
Tongue of a 47-year-old woman with hyperpigmented patches, which developed during treatment with pegylated interferon and ribavirin for chronic hepatitis C.
Hyperpigmentation of the tongue is an uncommon adverse effect of combination therapy with pegylated interferon and ribavirin for hepatitis C, described mainly in dark-skinned patients. In one series, it was observed in 5 of 171 dark-skinned patients versus none in 149 white patients.1 However, instances in white patients have also been reported.2 This adverse effect usually develops within the first few months of treatment, and improves or disappears slowly after the end of therapy. The hyperpigmentation is usually asymptomatic, but a burning sensation may be reported.2 Associated involvement of the gums and oral mucosa, and hyperpigmentation of the skin have been described.1
Histologic examination shows increased amounts of melanin in the basal layer of the epithelium.2 Upregulation of melanocyte-stimulating hormone receptors by interferon-α is suspected, which increases tyrosinase activity in melanocytes and tyrosine, the main precursor of melanin.2,3 The differential diagnosis includes Addison disease, Peutz–Jeghers syndrome, Laugier–Hunziker syndrome, amalgam tattoo, nevi, lichen planus pigmentosus, and adverse reactions to other medications, such as chloroquine and minocycline.
Footnotes
-
Competing interests: Claude Bachmeyer has received consultancy fees from Amgen for treatment of cutaneous adverse drug reactions induced by anti–epidermal growth factor receptor therapy. None declared by Jean-Charles Pellen.
-
This article has been peer reviewed.