Abstract
Humans are destined to explore space, yet critical illness and injury may be catastrophically limiting for extraterrestrial travel. Humans are superorganisms living in symbiosis with their microbiomes, whose genetic diversity dwarfs that of humans. Symbiosis is critical and imbalances are associated with disease, occurring within hours of serious illness and injury. There are many characteristics of space flight that negatively influence the microbiome, especially deep space itself, with its increased radiation and absence of gravity. Prolonged weightlessness causes many physiologic changes that are detrimental; some resemble aging and will adversely affect the ability to tolerate critical illness or injury and subsequent treatment. Critical illness–induced intra-abdominal hypertension (IAH) may induce malperfusion of both the viscera and microbiome, with potentially catastrophic effects. Evidence from animal models confirms profound IAH effects on the gut, namely ischemia and disruption of barrier function, mechanistically linking IAH to resultant organ dysfunction. Therefore, a pathologic dysbiome, space-induced immune dysfunction and a diminished cardiorespiratory reserve with exacerbated susceptibility to IAH, imply that a space-deconditioned astronaut will be vulnerable to IAH-induced gut malperfusion. This sets the stage for severe gut ischemia and massive biomediator generation in an astronaut with reduced cardiorespiratory/immunological capacity. Fortunately, experiments in weightless analogue environments suggest that IAH may be ameliorated by conformational abdominal wall changes and a resetting of thoracoabdominal mechanics. Thus, review of the interactions of physiologic changes with prolonged weightlessness and IAH is required to identify appropriate questions for planning exploration class space surgical care.
Humans are a space-faring species with a destiny to explore beyond the gravity of our home planet. Fifty years ago, humans successfully left and safely returned to our home planet. In the ensuing years, humans have occupied habitats in low Earth orbit such as the International Space Station, but have not ventured beyond as plans for exploration class missions (ECMs) have not yet come to fruition. However, as there appears to be a renewed commitment by the United States to pursue a return to the moon and Mars exploration,1 as well as much progress by other space-faring counties and private industry, it is now more relevant than ever to address the challenges of medical care for critically ill or injured space explorers, other than palliation with comfort measures.2 We have long been concerned that humans are the weak link in ECM exploration, as human space explorers have a susceptibility to injury and illness in space, including a fragility that is only exaggerated by the environment itself.3–7
A recent concept with profound implications for ECM is that, in reality, humans are just a life support system for their microbiome. The genetic material and cells of the commensal microbiota within a human greatly outnumber their host.8–11 The gut represents the largest body surface in contact with the external environment and constitutes a reservoir of more than 100 trillion bacteria.12 This can be understood through the concept of the holobiont, which recognizes the centrality of microbes in the function of individual organisms, best understood as a composite of the “host organism” and the symbionts within.13
Critical illness or injury may be catastrophic for both the mission and especially this holobiont patient, with their microbial guests. Given that humans involved in ECM will be leaving Earth and entering an environment much more hostile to life, this limitation of human exploration must be addressed. Without life support systems, space is entirely unforgiving to humans. In the space vacuum that begins approximately 700 km from Earth, there are essentially no colliding air molecules, resulting in absolute cold, anoxia and pressure. In this environment, the blood of an unprotected human would immediately boil in a process known as ebullism.3,14 However, of all the challenges of space travel, the one for which a truly effective countermeasure is still lacking is the absence of gravity.
A myriad of physiologic changes occur with the prolonged absence of gravity, most of which are detrimental to human health and will adversely affect the ability to tolerate critical illness or injury and subsequent treatment. Intra-abdominal hypertension (IAH) is another factor that should not be forgotten when considering critical illness or injury in space, though it is frequently ignored in critical illness or injury on Earth.15,16 IAH is defined as abnormally raised pressure (> 12 mm Hg) within the abdominal cavity that may induce malperfusion of both the viscera and microbiome with potentially catastrophic effects.17 The potential interactions between the physiologic changes created by prolonged weightlessness coupled with increased IAH have never been fully discussed in plans of how to deliver health care during exploration class space missions.
Physiologic and pathophysiologic effects of prolonged human exposure to weightlessness
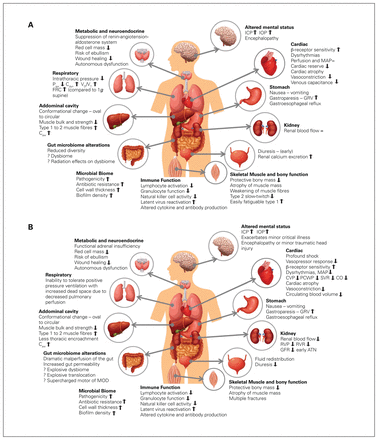
After prolonged exposure to weightlessness, the injured or ill astronaut will be at a physiologic disadvantage compared with patients on Earth.3,4,18,19 Although the physiologic changes are extensive and resemble those of aging in many regards,20 highlights of those most likely to degrade the capacity to withstand injury include reductions in circulating blood volume, severe vascular physiology changes (especially in the upper body), reduced red cell mass, loss of the protective bony mass, loss of both muscle mass and strength, immune suppression and negative effects on cognition.2,19,21–26 Fluid redistribution and diuresis result in up to a 10%–23% reduction in blood volume.3,27–29 It is reassuring that vigorous physical exercise and healthy nutrition can mitigate bone and muscle loss,2,30 but without dedicated countermeasures, cardiac atrophy, dysrhythmias, reduced cardiac output, and alterations in vascular tone and neuroendocrine function may occur after prolonged weightlessness.31 Although it is “space-appropriate,” when faced with the reintroduction of gravity, the cardiac functional reserve will be reduced and the autonomic nervous system re-adapted with greater β compared with α receptor sensitivity, presumably affecting vasoconstriction in hemorrhage.18 Basic wound healing may also be affected.32 Tensiometric and histologic findings from rat abdominal incisions in orbital space flight showed greater inflammatory responses, increased fibroplasia, abnormal collagen deposition and reduced stress loading capacity.32 Figure 1 shows the effects of weightlessness on different organ functions. Finally, it needs to be appreciated that only 4 humans have as yet spent greater than a year in low Earth orbit; no one has yet ventured beyond Earth’s orbit, certainly not for the 3 years or more that may be required to explore Mars.2
(A) Effects of prolonged weightlessness on organ function in health. (B) Postulated effects of prolonged weightlessness on organ function in critical illness or injury. Paw = airway pressures; Cdyn = Compliance-dynamic; Vd/Vt = dead space ventilation; FRC = functional residual capacity; ICP = intracerebral pressure; IOP = intraocular pressure; MAP = mean arterial pressure; CVP = central venous pressure; PCWP = pulmonary capillary wedge pressure; SVR = systemic vascular resistance; CO = cardiac output; RVP = renal vein perfusion; RVR = renal vascular resistance; GFR = glomerular filtration ratio; ATN = acute tubular necrosis; MOD = multiple organ dysfunction syndrome; Caw = central airway compliance; GRV = gastric residual volume. This is an original figure created by Dr. Manu L. N. G. Malbrain.
The abdominal wall, abdominal compartment and gut in space
Despite the large amount of life sciences research that has been done to address space physiology (e.g., ground-based simulations, parabolic flight and short- and long-duration space missions), the abdomen and its macroscopic function have not been well-addressed. Discussion of gastrointestinal function changes and intra-abdominal pressures (IAP) are not available in general reviews of human space-adapted physiology, though body composition studies have been performed.25,33 The limited data show that soft-tissue congestion of the head may affect taste and smell; upper and lower gastrointestinal motility often decreases, leading to reduced oral intakes, and astronauts may be at risk for dehydration.34 It has also been hypothesized that the absence of gravity in space may tend to increase both gastric emptying35 and the transit rate along the small intestine, potentially by decreasing the dimensionless ratio of gravitational forces to viscous forces.36 According to Smirnov and Lizko, extended space missions are associated with a hypersecretory syndrome of the stomach and a decrease in the functional capacity of the pancreas.35
As will be discussed further, the major determinant of abdominal stiffness is the anterolateral abdominal musculature. There have been no specific studies of abdominal musculature relevant to concepts involving abdominal compliance, in terms of the abdominal container. However, there has been substantial research on skeletal musculature because space-induced deconditioning of the postural muscles is a critical issue in ECM space flight. It is well-established that skeletal muscles undergo substantial atrophy in response to actual and simulated weightlessness.25,37 Animal studies show that the postural muscles, which generally contain a higher percentage of type 1 slow-twitch fibres, are more prone to atrophy than nonpostural type 2 fibres.37,38 Such changes may occur very quickly, even after only 2–5 days of space flight.39 There also appears to be a phenotypic shift in muscle fibre type, from type 1 to type 2, that would increase fatiguability over time.40 Besides total muscle atrophy of up to 30%,41 there is also a degenerative effect on neural drive and muscle activation capacity, with up to a 35%–40% reduction in electromyographic activity activity.42 On Earth, the abdominal muscles support the trunk for both posture and locomotion.43 They are predominantly (55%–58%) slow-twitch type 1 fibres.43 We assume and speculate that the abdominal musculature will decondition just as much as the anti-gravity appendicular muscles, for which extensive conditioning exercises are carried out by astronauts in space.
The human microbiome
Humans, whether on Earth or in space, represent a scaffold on which diverse microbial ecosystems are established.44 The microbial world that exists within the human gut has profound implications for human health, particularly when proposing to send humans away from Earth for years. The human “microbiota” refers to the communities of microbes (including commensal and pathogenic bacteria, viruses and fungi), whereas the “microbiome” refers to microbial genes, gene products (proteins, metabolites) and community structure (distribution, diversity, evenness), as well as the particulars of the environment in which the microbes reside, constituting the full microbial ecosystem of the body.45 In health, the microbiome is characterized by a state of symbiotic homeostasis between the host, microbial commensals, as well as many potentially pathogenic bacteria.46 The composition and ecological structure of the microbiome are continuously evolving in response to environmental pressures (e.g., dietary intake, exposure to new microbes, antibiotics) as well as changes in host physiology.47 Therefore, the combination of drastic environmental changes and extreme physiologic adaptations of space travel will have pronounced effects on the human microbiome. Indeed, the recent NASA Twins Study provided the first glimpse of the impact of space travel on the human microbiome, showing marked alterations to the composition of the fecal microbiota during a year-long space flight.2
In general, it appears that a highly diverse and complex microbiome is advantageous for the host. The microbiome is critically important for regulating many aspects of human physiology, including immune function, gut mucosal barrier integrity, nutritional and metabolic functions, as well as colonization resistance against pathogens and infections.45,48 Loss of microbial diversity and community structure (called dysbiosis) results in dysregulation of these physiologic systems, and often, outgrowth and overrepresentation of potentially pathogenic organisms within the gut. Consequently, gut microbiome dysbiosis has been linked to an extensive list of disease states, ranging from gastrointestinal disorders, autoimmunity and infections to cancer, metabolic syndrome and neurologic diseases.48,49 Our understanding of the cellular and molecular mechanisms controlling host–microbiota mutualism in health and disease has evolved tremendously in recent years. Gut bacteria have been shown to actively engage with the mucosal immune system of the gut to maintain local host defence and barrier integrity. This involves a complex interplay between microbes, microbial products and metabolites, the mucosal epithelium, stromal cells and a variety of mucosal immune cells (both innate and adaptive players).47 However, the immunological influence of the microbiota extends well beyond the confines of the gut, as microbial cues have been found to regulate systemic immune responses, and even organ-specific immunity at sites distant from any direct microbial contact, such as the brain.50 During critical illness or injury, there can be a catastrophic loss of microbial diversity with the induction of a state of severe dysbiosis.51 The loss of normal microbial diversity is met with overrepresentation by potentially pathogenic organisms that, combined with loss of gut barrier integrity, yields a greater potential to translocate to extraintestinal sites.52 In general, the occurrence of severe illness or injury is accompanied by inflammation and reduction in microbial diversity.9,53 Although much remains to be learned about the impact of critical illness and extreme physiologic abnormalities on gut microbiome composition and the resulting pathological host–microbial interactions, it is believed that that this process serves as a central driver of critical illness.
Exploration class space flight, microbial passengers and the human biome
Even without the occurrence of a critical illness or injury, the ubiquitous space characteristics of prolonged weightlessness and increased exposure to cosmic radiation, together with the dietary changes that are required during space travel, will contribute to microbial dysbiosis. Changes in bacterial physiology are likely to have a profound impact on the health and well-being of astronauts.26,46,53 Although it is reassuring that space-induced microbiota changes reverted to their preflight levels within weeks of return to Earth for the NASA Twins Study astronaut,2,30 there were still profound in-flight changes that occurred despite the astronaut not having any critical illness or injury. This landmark study deserves comment: 2 identical twins, the Kellys, were studied by 10 science teams who examined the twins’ physiology, memory abilities and genetic material, among other parameters, before, during and after a year in which 1 twin was terrestrial and 1 was in weightlessness on-board the International Space Station. Although many, if not most, physiologic functions were affected by prolonged space flight, most resolved upon return to Earth and terrestrial gravity. However, it remains extremely concerning that persistent changes might include altered gene expression levels, increased DNA damage from chromosomal inversions, increased numbers of short telomeres and attenuated cognitive function.2
At the cellular level, every cell and organ system previously conditioned to experience gravity is likely affected by the absence of this normally ubiquitous force,20 and bacteria inherently change when removed from its influence. For example, thicker cell walls with higher minimal inhibitory requirements for antibiotics in common pathological bacteria have been encountered.54,55 Salmonella typhimurium cultures grown under modelled microgravity were more virulent and were recovered in higher numbers from the murine spleen and liver following oral infection, compared with organisms grown under normal gravity.56 Furthermore, microgravity-grown salmonellae were more resistant to acid stress and macrophage killing, and exhibited substantial differences in protein synthesis than did organisms grown under normal gravity.57 Space flight investigations have reported that bacterial growth appears greater in weightlessness compared with terrestrial controls.58 Biofilms protect bacteria from environmental conditions and may increase antibiotic resistance; in diminished gravitational conditions in the laboratory, bacteria showed thickened and more antibiotic-resistant growth.46,59 Postflight bacteria collected from the crew of the Apollo-Soyuz Project exhibited increased antibiotic resistance compared with preflight.60 An animal study noted increased animal death from injection of Klebsiella pneumoniae after simulated weightlessness compared to controls.61 Thus, the internal atmosphere of any space habitat will likely be populated by an increasingly virulent microbiological flora.3
Potentially even more profound than isolated bacterial changes, however, are dramatic alterations in the human microbiome related to behavioural alterations of normal commensal organisms. Stressful conditions to be expected in space flight include increased cosmic radiation, reduced intake of commensal bacteria, potential antibiotic usage and the absence of gravity.10,19,35,46,62 The genetic diversity of microbes that astronauts will be exposed to during a multiyear ECM will be greatly altered even before the mission begins. Isolation of the crew, purification of food, restricted diets that are high in iron and weightlessness itself may be associated with the induction of a dysbiosis and shift away from defensive organisms (i.e., bifidobacteria and lactobacilli) toward a greater dominance of opportunistic pathogens such as Bacteriodes, Pseudomonas and Clostridia species.10,19,35,46,62 Such changes in the health and diversity of the microbiome have been linked to immune dysregulation and dysfunction, and increased susceptibility to infection.46 Dysbiotic conditions associated with weightlessness have also been associated with the increased elaboration of proinflammatory cytokines and the decreased secretion of barrier immunoglobins.10 Li and colleagues found that weightless analogue control animals had a fourfold expansion of pathogenic colonizing bacteria, a halving of regulatory T cell numbers and anti-inflammatory interleukin (IL)-10 production, as well as a twofold increase in colonic IL-1β expression, increased circulating neutrophils and colonic neutrophil infiltration.63 After launch and attainment of a deep space mission profile, the microbiome may be even more profoundly affected, with potentially apocalyptic harm as there is no reliable prediction of how the vastly increased (and rapidly replicating) number of microbiota genes will respond to adverse extraterrestrial conditions, including increased radiation fluxes.10 Finally, the gut lining cells, in intimate physical and physiologic proximity with the microbiome, are some of the most actively replicating human cells and thus potentially susceptible to radiation-induced genetic replication errors.19
The motor of multisystem organ dysfunction within a potential pressure cooker
The gut may be considered a clinically silent organ system that may be a “ticking time bomb” in critically ill or injured patients. On Earth, multiple organ dysfunction syndrome (MODS) is legitimately feared as a leading cause of morbidity, death and resource use in critical care units.12,64,65 In space, with potentially increased microbial virulence, immune dysfunction, relative hypovolemia, potentially reduced cardiorespiratory compensation and a dysbiome, MODS might be catastrophic. For more than half a century, it has been hypothesized that the gut is the motor of a catastrophic sequence of events that ultimately results in MODS.12,66–68 The theory is that translocation of aerobic gram-negative bacilli and/or absorption of their resultant endotoxin contribute to a systemic inflammatory state resulting in widespread organ dysfunction, tissue damage and subsequent MODS.67
It is commonly assumed that shock is the precipitating factor that results in gut hypoperfusion and initiation of MODS. MODS is assumed to occur as a result of splanchnic hypoperfusion and injury to the gut at the expense of other organs such as the heart and brain.9,69 However, this poorly explains the later development of MODS in patients where shock resuscitation has been prompt. Despite even adequate resuscitation, gut dysfunction promotes distant organ injury.9,69 This dysfunction is manifested in a number of related but distinct pathologies including mucosal ischemia, altered intestinal transit, luminal nutrient transportation and disuse-associated villus atrophy, resulting in overall reduction in mucosal surface area, loss of barrier function and increased permeability.9 MODS also likely involves the rapid transformation of a healthy microbiome into a dysbiome or pathobiome.70 This specific sensitivity of the gut to ischemic injury in shock also correlates with a secondary sensitivity of the lungs to gut-mediated lung injury. The gut–lymph hypothesis suggests that translocating and dead bacteria, cytokines and chemokines actually travel through the mesenteric lymphatics to induce acute lung injury and propagate further distant organ dysfunction.9,71 A remarkable, but potentially chilling, finding was studied in simulated weightlessness and showed nearly identical findings in rat guts, simply due to the environment itself.19 Compared with controls, simulated weightlessness damaged the intestinal villi and structural integrity of tight junctions, increased the intestinal permeability, upregulated the expression of proapoptotic proteins and downregulated the expression antiapoptotic proteins.19 Space flight may be inherently proinflammatory, with microgravity exposure being associated with high levels of macrophage inflammatory receptors and inflammatory mediators and cytokine expression.10,72 This was confirmed during prolonged space flight during the NASA Twins study, whereby the space explorer had increased levels of IL-1α, IL-1β, IL-2 and prostaglandin F2α compared with the Earth-bound control twin.30 Crucian and colleagues hypothesized that such persistent low-level inflammation during space flight could derive from either environmental radiation or from space-associated alterations in the gut microbiome.72
Once a severe insult with progressive dysoxia has been sustained by the gut, sustained progressive organ failure may occur despite subsequent normalization of cardiac output. We have previously speculated that IAH also potentiates this progressive organ failure, despite being a potentially treatable factor that is often ignored.73–75 This can be conceptualized as placing the “motor” of MODS within the abdominal cavity “pressure cooker,” with the capability to squeeze out perfusing blood and thus inducing ischemia to supercharge the developing injury to the gut. In experimental animal models, what might erroneously be considered “mild” grade 1 IAH (15 mm Hg) had profound effects on mucosal blood flow, which was reduced by 50% after only 4 hours.76 When IAH was even more severe (in the range commensurate with abdominal compartment syndrome, defined as IAP > 20, associated with new organ failure),69,77 there was profound injury to the gut.
IAH and abdominal compartment syndrome have been conceptualized as having a largely mechanical pathogenesis, occurring when the physical capacity of the abdominal container is exceeded by the addition of surgical packing, abdominal viscera edema, hematomata and/or ascites all of which increases the intra-abdominal volume (IAV). In addition to this mechanical schema, Malbrain and colleagues have added the concept of 2 physiologic “hits,” that sequentially produce a self-perpetuating process of abdominal pathology.78,79 In the first hit, resuscitation of shock induces bowel ischemia reperfusion injury.80,81 This “acute bowel injury” results in the release of proinflammatory mediators into the peritoneum and systemic circulation. This in turn leads to neutrophil priming, increased intestinal wall permeability, extravasation of fluid into the bowel wall and mesentery, translocation of intestinal bacteria and absorption of bacterial endotoxin.82–85 With a pathogenic dysbiome, such a hit might prove itself to be catastrophic. It is remarkable how the effects of simulated weightlessness on gut permeability19 mirror the same severe gut end-organ pathology with mild IAH, recognizing severe systemic downstream effects.73,76,86 With contemporary resuscitation practices, a second hit is almost inevitable. In the second hit, the resultant abdominal visceral edema leads to IAH, which compresses intra-abdominal lymphatics and results in a progressive spiral of decreased gut perfusion, mucosa-to-serosa intestinal necrosis, a further increase in bowel wall permeability, heightened bacterial translocation and endotoxin absorption and release of proinflammatory mediators.81,82 During an ECM, with limited critical care support, such an occurrence would be catastrophic.
Abdominal compliance, the abdominal wall, and abdominal components
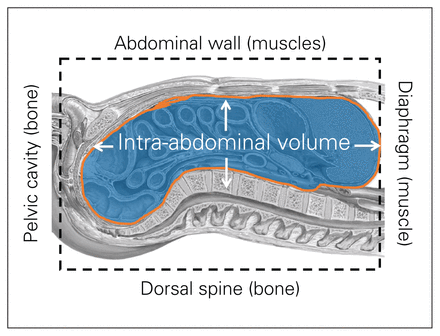
Abdominal compliance (Cab) is defined as a measure of the ease of abdominal cavity expansion, expressed as a change in IAV per change in IAP,69 and has been largely neglected in the past.87 The Cab is a dynamic variable dependent on baseline IAV and IAP, and is critically dependent on the abdominal reshaping and stretching capacity.87–90 Patients with decreased Cab are at risk of fulminant IAH, with modest increases in IAV, whereas those with an increased or compliant Cab, are relatively “protected” from IAV increases.87,89 Of all the body cavities, the abdominal cavity is the most susceptible to changes in container compliance, which is referred to as abdominal compliance for further discussion. The anatomy of the abdominal cavity determines the potential for volume expansion (Fig. 2). The posterior wall is rigid due to the spine and the retroperitoneal organs; the lower abdominal wall is restricted by the pelvic girdle. The upper abdominal border is constituted by the diaphragm, which can potentially expand into the chest with negative respiratory effects relative to the gravitational environment.87,91
Anatomy of the abdominal cavity. Figure reproduced and adapted from Malbrain ML, Roberts DJ, De Laet I, et al. The role of abdominal compliance, the neglected parameter in critically ill patients - a consensus review of 16. Part 1: definitions and pathophysiology. Anaesthesiol Intensive Ther 2014;46:392–405 (licensed under CC by 4.0).89
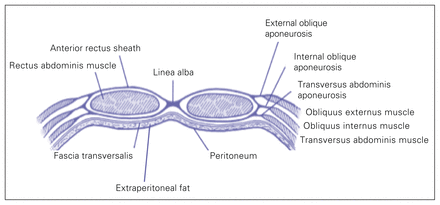
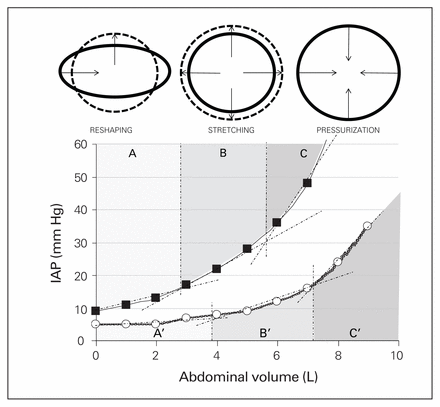
From a practical standpoint, the anterolateral abdominal wall is the most relevant determinant of functional Cab, with a small contribution from the diaphragm, all other structures being relatively fixed.87,89 The anterolateral abdominal wall consists of skin, superficial fascia, fat, muscles with their aponeuroses, transverse fascia and the parietal peritoneum (Fig. 3). The abdominal muscles have a composite–laminate structure, with extracellular matrix largely determining the nonlinear stretch characteristics.92 Forces generated by the 3 flat muscles, passed between one another through connective tissue linkages, and the greater stiffness and stress generated by the abdominal wall muscles, will have the added effect of enhancing intra-abdominal pressure.92 When examining a pressure–volume93 graph of abdominal Cab, there are 3 distinct phases of mechanical behaviour: a reshaping phase of unstressed volume, with a very flat slope on the pressure–volume graph and minimal increase in IAP, if any; a stretching phase with nearly linear matching of pressure increases and added volume; and an exponential phase of overpressurization that is very sensitive to any volume increases, with often disastrous results.87 However, given this exponential relationship, removal of only a modest IAV in a patient with severe IAH may dramatically improve the condition.87
Relevant anatomy of the anterolateral abdominal wall. Figure reproduced and adapted from Malbrain ML, Roberts DJ, De Laet I, et al. The role of abdominal compliance, the neglected parameter in critically ill patients - a consensus review of 16. Part 1: definitions and pathophysiology. Anaesthesiol Intensive Ther 2014;46:392–405 (licensed under CC by 4.0).89
A critical determinant of tolerance to increased IAV, without development of severe IAH, appears to be the reshaping capacity of the abdominal wall, which relates to the inherent mechanical health of the abdominal wall. Reshaping capacity may be increased with multiparty previous laparoscopies, but is more likely decreased with previous laparotomy (variably), external binding or dressings, obesity, well-developed healthy athletic musculature, abdominal edema and inflammation, whether localized or as a generalized whole-body condition.89
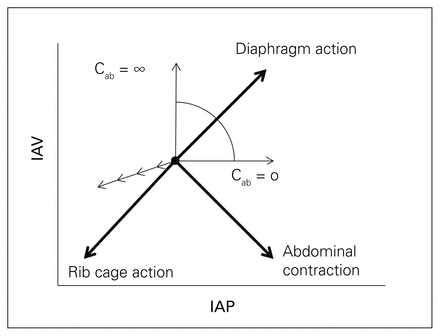
The abdominal volume generally behaves as a hydraulic system.88,89 Actual intra-abdominal pressure measurements of canines in various positions and ventilatory conditions indicated that the abdomen does, at times, behave like a hydraulic system.94 However, abdominal volume can deviate from simple hydrostatic behaviour to the extent that shape-stable abdominal viscera can be deformed.94 Loring and colleagues concluded that there are 3 factors affecting IAP, including uniform hydraulic compression, gravitational compression, and shear deformation.95 Thus, compression of the abdominal volume, as may be induced by anterior abdominal muscle activity, diaphragmatic activity, mechanical ventilation with positive pressure or torso binding, results in spatially homogeneous IAP changes that augment a hydrostatic gravitation gradient in terrestrial environments (Fig. 4). Malbrain has summarized and hypothesized that, based on the available data, the impact of shear deformation on the measurement of IAP is probably not significant in a critically ill or injured patient who is fully sedated and mechanically ventilated in a completely supine position.89
Relationship between intra-abdominal volume (IAV), abdominal wall compliance (Cab) and intra-abdominal pressure (IAP). The direction of the movement associated with the sole action of the rib cage inspiratory muscles, the abdominal expiratory muscles and the diaphragm are shown. The direction of the latter depends on abdominal compliance (Cab), but is constrained within the sector shown. When the diaphragm contracts, it moves downwards into the abdominal cavity, and this displacement will increase IAV with a resulting increase in IAP (depending on Cab). Reductions in IAV will result in a decrease in IAP (small nonbolded arrows). Figure reproduced and adapted from Malbrain ML, Roberts DJ, De Laet I, et al. The role of abdominal compliance, the neglected parameter in critically ill patients - a consensus review of 16. Part 1: definitions and pathophysiology. Anaesthesiol Intensive Ther 2014;46:392–405 (licensed under CC by 4.0).89
Polycompartment physiology and intra-abdominal hypertension in weightlessness
Polycompartment syndrome is a condition where 2 or more anatomic compartments have elevated compartmental pressures.69 Compartment pathology in one of the 4 major body cavities will affect the physically contiguous cavities through direct pressure, as well as hemodynamic, humoural and biomediator interactions.96–98 In particular, the chest and abdomen are inexorably linked and must be considered as a single physiologic couple acting in series and in parallel.90 On Earth, the abdominal cavity is typically the enemy of the chest cavity in critical illness and injury, especially with obesity or IAH.87–89,99 The abdominal cavity can be considered an incompressible volume held between the diaphragm and abdominal muscles, with contraction of either causing a reciprocal passive displacement of the other.100,101
We and others have long hypothesized that the respiratory system of the ill or injured astronaut could benefit from weightlessness, specifically compared with the supine bed rest position typical of most critically ill terrestrial patients.3,5,91 This perspective, however, requires an understanding of the patient’s health and positioning. The lung is exquisitely sensitive to normal gravity, which induces gradients in blood flow, alveolar size, ventilation and gas exchange.102,103 Studies in weightlessness have shown that topographic differences in ventilation, perfusion, and lung expansion are reduced, but not abolished.104,105 Multiple principles are relevant. With inflammatory mediator-induced “low-pressure” pulmonary edema, as seen in acute respiratory distress syndrome, pulmonary function might paradoxically worsen with the transition to weightless conditions, even from reduced gravity, as all regions of the lung become equally susceptible to endothelial dysfunction.106 In septic conditions, however, vasogenic factors will compete with anatomic ones. In the standing adult, gravity normally unloads the lower lung fields, increasing the functional residual capacity (FRC). Compared with upright posture on Earth, weightlessness reduces the FRC, expiratory reserve volume, total lung capacity, residual volume and inspiratory vital capacity. These reductions are largely induced by a cranial shift of the diaphragm, which is no longer pulled caudally by gravity.103,107,108 This cranial diaphragmatic shift, however, is associated with a conformational change in the pericardium to a more spherical shape, thus reducing cardiac wall tension.109
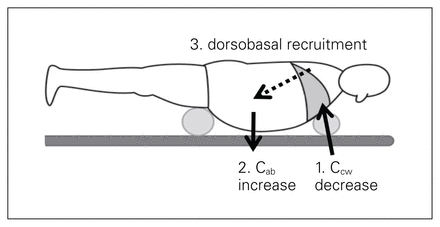
When considering critical illness, however, upright 1g lung performance is not the pertinent comparison because the supine bed rest position is ubiquitous for most critical care delivery. In terrestrial gravity, the supine, head-of-bed elevation and sitting positions are all associated with increased IAP and reduced FRC because the weight of the abdominal contents is transmitted across the diaphragm to the thoracic contents.110 In weightlessness, however, compression of the lower thoracic cavity by the abdominal contents is obviated. This might be expected from previous studies in parabolic flight and true space.103,107,108 Therefore, although the FRC decreases in weightlessness compared to the upright posture on Earth, it actually increases compared to the supine terrestrial (1g) subject.102,105 On-board the Space Life Science-1 module, the average FRC and expiratory reserve volume were 650 and 850 mL higher, respectively, over those measured in the 1 g supine posture.105 Weightlessness has an impact on the physiologic effects exerted by body position, and being in a prone position will not have the same effects as on Earth. On the other hand, weightlessness and gravitational forces also explain why the prone position with abdominal suspension in patients with increased IAP may have beneficial effects on dorsobasal recruitment on Earth (Fig. 5).
Effects of the prone positioning with abdominal suspension on chest (CCW) and abdominal wall compliance (Cab). The suspension placed under the chest will reduce chest wall compliance (1) while the abdominal suspension placed at the level of the symphysis will exert a gravitational effect that will increase abdominal wall compliance (2). This will result in recruitment of dorsobasal lung regions (3). Figure reproduced and adapted with permission from Regli A, Pelosi P, Malbrain MLNG. Ventilation in patients with intra-abdominal hypertension: what every critical care physician needs to know. Ann Intensive Care 2019;9:52 (original publisher: Springer; licensed under CC by 4.0).99
Thoracoabdominal interactions in variable gravity
Although the space-adapted astronaut may have diminished cardiorespiratory reserves to perfuse a theoretically tense, high-pressure abdominal cavity, there may be fundamental changes in the cardiorespiratory interactions that might be predicted to be protective regarding IAH in ECM situations.91 Although we are not aware of reports of IAP ever being measured in humans in weightlessness or space, there have been thoracic compliance measurements on anesthetized swine during parabolic flights with the Falcon 20 Aircraft. Performing such surgery required the creation of aeromobile modular critical care, resuscitation and surgical suites for operational medicine and was developed to support surgery in variable gravity on-board the aircraft (Fig. 6).5,111,112 Thereafter, while in flight, different abdominal conditions were standardized to involve no manipulation, abdominal wall retraction and standard gas insufflation, creating an IAP of 15 mm Hg (equating to grade II IAH).5,91,112,113 As swine were anesthetized, paralyzed and mechanically ventilated with a pressure control mode, the breath-by-breath tidal volumes were used to calculate the relative functional thoracoabdominal compliance in each combination of gravity (normal 1g, hypergravity 2g and weightlessness 0g) and abdominal condition (control, retraction or the IAH required for laparoscopic surgery). The expected physiologic decreased tidal volumes and compliance with the induction of IAH was confirmed in all gravitational conditions, and was especially exacerbated in hypergravity conditions. In contrast to hypergravity, weightlessness fundamentally unloaded the abdominal cavity from the thorax such that, even without any manipulation the ventilatory volumes, the thoracic compliance increased spontaneously with transition between hypergravity, normal gravity and weightlessness. Thus, weightlessness appears to be a fundamentally beneficial state in protecting the lungs from increased IAP.91
Porcine life support and surgical work station before loading. An aeromobile modular critical care, resuscitation and surgical suite for operational medicine was developed to support surgery in variable gravity on-board the Falcon 20 Aircraft. Photograph taken by Andrew W. Kirkpatrick.
The actual mechanism of this protection may be a physical reconfiguration of the abdominal geometry, with the abdomen spontaneously assuming a more round than oval shape that maximizes the stress relaxation of the cavity, an abolition of hydrostatic pressures within the cavity or a combination of both effects. When the abdominal cross-sectional width and height of anesthetized swine was measured upon transitioning between gravitational states, the anterior–posterior height increased an average of 9 mm in controls and 12 mm with IAH, while the width of the abdomen narrowed by 27 mm in controls, and 8.3 mm in those with IAH.113 This configurational change in the abdominal wall may equate to a natural reshaping versus stretching (Fig. 7).87,89 On Earth, the high compliance reshaping phase of abdominal behaviour typically continues until the abdominal wall develops a circular shape.
Evolution of the internal abdominal cavity perimeter during an increase in volume. In a case of gynoid obesity, the internal abdominal perimeter is shaped as an ellipse. Patients with an ellipse-shaped internal perimeter have a huge stretching capacity (and thus very good abdominal compliance); this is illustrated with the progression of the shape from ellipse (full line) at baseline to a sphere (dotted line) with increasing intraabdominal pressures (IAP), as obtained during laparoscopy. The arrows show the centripetal movement of the lateral edges of the ellipse and the centrifugal movement of the craniocaudal edges. During an increase in IAV from baseline to maximal stretch, the difference between the long and short axis of the ellipse decreases, while the internal perimeter and surface area increase. At maximal stretch, the external and internal abdominal perimeters are equal. Patients with android obesity don’t have this reshaping and stretching capability. A, B, and C show the reshaping, stretching and pressurization phases, respectively, in poor compliance conditions. A’, B’ and C’ show the same phases with normal abdominal compliance. Figure Reproduced from Malbrain et al, with permission Anesthesiology Intensive Therapy 2014;46:392–405 (licensed under CC by 4.0).89
We also speculate that abdominal muscle tone may be decreased in prolonged weightlessness, related to a decrease in the overall bulk of the abdominal musculature. Further, if the muscle fibre type shifts from type 1, capable of maintain prolonged tension at low metabolic cost, to type 2 fibres, which fatigue easily, then this might functionally decrease muscle tone, increasing compliance. This is conceptually similar to how botulinum toxin is used for this purpose in abdominal wall reconstruction on Earth.114,115 With decreased muscle function, the compliance of the abdominal cavity will presumably shift to a curve with greater reshaping capacity.
Another profound concept is that vascular, and potentially any physiologic, compliance of the human body may be reset once gravity is removed. For instance, it was long predicted that central venous pressure would rise once an astronaut transitioned from terrestrial (1g) to weightlessness (0g) due to the release of the gravitational capacitance volume usually residing in the lower limbs. However, when actually measured in space flight, despite echocardiographic confirmation of increased cardiac preloads and increased cardiac outputs, the central venous pressure decreased.116–118 Thus, authors have previously postulated a hydrostatic resetting of great vessel pressures, such that the same blood volume is contained at a lower pressure.116 Alternatively, Hamilton feels that this phenomenon is more appropriately explained by the cranial shift of the diaphragm, with a simple reshaping of the pericardium to a spherical maximally unstressed volume that fills at lower pressures.109 These findings suggest that, in space, either more fluid may be contained within any fixed container at a reduced pressure,116,117 or that containers spontaneously achieve the optimal unstressed volume, inviting speculation that the abdominal compartment might also be more “permissible” to increased contents. We further speculate that the totality of these adaptations and responses to prolonged weightlessness may be a shift to a more favourable pressure–volume relationship that is better able to resist IAH.
Conclusion
The long-duration space traveller will be a vulnerable component, if not the most fragile component, of human exploration class missions. Adaptations to long-duration space exploration will likely leave the astronaut with reduced physiologic reserves to combat the stresses of critical illness and injury. These adaptations may potentially be associated with increased bacterial virulence and a dysbiotic microbiome, primarily residing within the gut, primed to drive sepsis and septic shock. Although this should portend grave concerns regarding the vulnerability of the gut, a potential ameliorating effect of prolonged weightlessness may be improved thoracoabdominal mechanics, mitigating the physiologic hit of intra-abdominal hypertension that likely complicates any shock state. Given the potential accelerated plans to leave low-Earth orbit, further study and corroboration of these opinions is urgently required to plan for the medical care required to support human-crewed exploration class missions.
Footnotes
The material discussed in this manuscript was previously presented at the 9th Scientific Meeting of the Abdominal Compartment Society, in Campinas, Brazil, May 31, 2019.
Competing interests: Major Andrew W. Kirkpatrick discloses having consulted for the Innovative Trauma Care and Acelity Corporations, and to having a personal relationship with J.L. McKee. Douglas R. Hamilton reports no disclosures. Jessica L. McKee discloses being employed by the Innovative Trauma Care Corporation. She has consulted for the Asceso, Sam Medical, and the Acelity Corporations. She has also done project work for the Canadian Forces and the COOL study. Her current position with the University of Calgary is funded through a grant with Purdue University. Braedon MacDonald reports no disclosures. Paolo Pelosi reports no disclosures. Chad G. Ball is coeditor-in-chief of CJS. He was not involved in the review or decision to accept this manuscript for publication. Michael Sugrue discloses payment for educational activity with Smith and Nephew, Acelity and NovusScientific that are not relevant to this paper Edward J. Kimball discloses that he is the current president of the World Society of the Abdominal Compartment. Manu L.N.G. Malbrain discloses that he is a member of the medical advisory boards of Pulsion Medical Systems (now fully integrated in Getinge, Solna, Sweden) and Serenno Medical (Tel Aviv, Israel), and that he consults for Baxter, Maltron, ConvaTec, Acelity, Spiegelberg and Holtech Medical. Derek Roberts, Paul B. McBeth, Federico Cocolini, Luca Ansaloni, Bruno Peireira and Mark R. Campbell report no disclosures.
Disclaimer: The opinions, interpretations, conclusions and recommendations are those of the authors only and are not specifically endorsed by Department of National Défense of Canada, or any other public or governmental agencies.
Funding: This work was partially supported by a Near Earth Space Technologies Grant from the University of Calgary.
Contributors: Andrew Kirkpatrick, Jessica McKee, Paolo Pelosi, Chad Ball, Derek Roberts, Paul McBeth, Luca Ansaloni and Manu Malbrain conceived and designed the study. Andrew Kirkpatrick and Chad Ball acquired the data and analyzed/interpreted it with Douglas Hamilton, Braedon MacDonald, Paolo Pelosi, Derek Roberts, Paul McBeth and Michael Sugrue. Andrew Kirkpatrick, Chad Ball, Paolo Pelosi, Derek Roberts, Paul McBeth, Edward Kimball and Manu Malbrain wrote the article. All authors critically revised the manuscript and gave final approval of the version of the article to be published.
- Accepted January 23, 2020.