Abstract
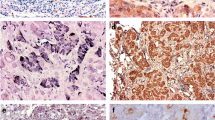
Somatostatin analogs are well established in the treatment of malignant endocrine pancreatic tumors (EPTs). Our goal is to individualize their treatment using receptor-subtype-specific analogs and, therefore, exploring the receptor expression is highly important. We have examined the expression of somatostatin receptor (sst) subtypes 1–5 on tumor cells and in intratumoral vessels in 28 tumor tissues from malignant EPTs with immunohistochemistry using sst-subtype-specific polyclonal antibodies. We found that sst2 and sst4 stained positive in 90% and sst1 in 70% of the tumor tissues, whereas sst3 and sst5 stained positive in only 50% of the tumor tissues. Sst expression in intratumoral vessels was high for sst2 and sst4 (80%), moderate for sst1 (40%), and low for sst3 and sst5 (10%). The ssts were evenly distributed among the different tumor subtypes. However, tumors belonging to the same subgroup of EPTs showed a variable expression of receptor subtypes. No differences in receptor-subtype expression could be seen between poorly and well-differentiated tumors, nor between primary tumors and metastases. Prior medical treatment did not influence sst expression pattern. In conclusion, sst2 and sst4 were expressed in most tumor tissues and intratumoral vessels from EPTs. However, sst3 and sst5 were lacking in half of the tumor tissues and in most of the intratumoral vessels. These differences indicate the importance of determining each tumor’s subset of receptors before treatment with receptor-subtype-specific analogs is initiated. The importance of sst expression in intratumoral vessels is not yet known.
Similar content being viewed by others
References
Öberg K. Neuroendocrine gastrointestinal tumors—a condensed overview of diagnosis and treatment. Ann Oncol 1999; 10: S31–38.
Welbourn RB, Wood SM, Polak JM, Bloom SR. Pancreatic endocrine tumors. In: Bloom SR, Polak JR (eds). Gut Hormones. Churchill Livingstone, London, 1981, pp. 547–554.
Eriksson B, Öberg K. An update of medical treatment of malignant endocrine pancreatic tumors. Acta Oncol 1993; 32: 203–208.
Reubi JC, et al. Detection of somatostatin receptors in surgical and percutaneous needle biopsy samples of carcinoids and islet cell carcinomas. Cancer Res 1990; 50: 5969–5977.
Patel YC. Somatostatin and its receptor family. Front Neuroendocrinol 1999; 20: 157–198.
Scarpignato C. Somatostatin analogues in the management of endocrine tumors of the pancreas. In: Mignon M, Jensen RT (eds). Endocrine Tumors of the Pancreas. Karger, Basel, 1995, pp. 385–414.
Arnold R, et al. Somatostatin analogue octreotide and inhibition of tumor growth in metastatic endocrine gastroenteropancreatic tumors. Gut 1996; 38: 430–438.
Reubi JC, Horisberger U, Laissue J. High density of somatostatin receptors in veins surrounding human cancer tissue: Role in tumor-host interaction? Int J Cancer 1994; 56: 681–688.
Watson JC, et al. Growing vascular endothelial cells express somatostatin receptor subtype 2 receptors. Br J Cancer 2001; 85: 266–272.
Albini A, et al. Somatostatin controls Kaposi’s sarcoma tumor growth through inhibition of angiogenesis. FASEB J 1999; 13: 647–655.
Yamada Y, et al. Cloning and functional characterization of a family of human and mouse somatostatin receptors expressed in brain, gastrointestinal tract and kidney. Proc Natl Acad Sci USA 1992; 89: 251–255.
Yamada Y, et al. Somatostatin receptors, an expanding gene family: cloning and functional characterization of human sstR3, a protein coupled to adenylyl cyclase. Mol Endocrinol 1992; 6: 2136–2142.
Yamada Y, et al. Cloning, functional expression and pharmacological characterization of a fourth (hsstR4) and a fifth (hsstR5) human somatostatin receptor subtype. Biochem Biophys Res Commun 1993; 95: 844–852.
Portela-Gomes GM, Stridsberg M, Grimelius L, öberg K, Janson ET. Expression of the five different somatostatin receptor subtypes in endocrine cells of the pancreas. Appl Immunohistochem Mol Morph 2000; 8: 126–132.
Kulacsiz H, et al. Identification of somatostatin receptor subtypes 1, 2A, 3 and 5 in neuroendocrine tumors with subtype specific antibodies. Gut 2002; 50: 52–60.
Papotti M, et al. Expression of somatostatin receptor subtypes 1–5 in 81 cases of gastrointestinal and pancreatic endocrine tumors. A correlative immunohistochemical and reverse-transcriptase polymerase chain reaction analysis. Wirchows Arch 2002; 440: 461–475.
Rindi G, Capella C, Solcia C. Cell biology, clinicopathological profile, and classification of gastro-enteropancreatic endocine tumors. J Mol Med 1998; 76: 413–420.
O’Shaughnessy DJ. Radioimmunoassay of gut regulatory peptides. In: Bloom SR, Long RD (eds). Antibodies. WB Saunders, London, 1982, pp. 9–20.
Vaitukaitis J, Robbins JB, Nieschlag E, Ross GT. A method for producing specific antisera with small doses of immunogen. J Clin Endocrinol 1971; 33: 988–991.
Janson ET, Gobl A, Kälkner KM, Öberg K. A comparison between the efficacy of somatostatin receptor scintigraphy and that of in situ hybridization for somatostatin receptor subtype 2 messenger RNA to predict therapeutic outcome in carcinoid patients. Cancer Res 1996; 56: 2561–2565.
Buscail L, et al. Stimulation of tyrosine phosphatase and inhibition of cell proliferation by somatostatin analogues: mediation by human somatostatin receptor subtypes sstR1 and sstR2. Proc Natl Acad Sci USA 1994; 91: 2315–2319.
Buscail L, et al. Inhibition of cell proliferation by the somatostatin analogue RC-160 is mediated by somatostatin receptor subtypes sstR2 and sstR5 through different mechanisms. Proc Natl Acad Sci USA 1995; 92: 1580–1584.
Sharma K, Patel YC, Srikant CB. Subtype-selective induction of wild-type p53 and apoptosis, but not cell cycle arrest, by human somatostatin receptor subtype 3. Mol Endocrinol 1996; 10: 1688–1696.
Sharma K, Srikant CB. Induction of wild-type p53, Bax and acidic endonuclease during somatostatin-signaled apoptosis in MCF-7 human breast cancer cells. Int J Cancer 1998; 76: 259–266.
Rohrer SP, et al. Rapid identification of subtype-selective agonists of the somatostatin receptor through combinatorial chemistry. Science 1998; 282: 737–740.
Afargan M, et al. Novel long-acting somatostatin analog with endocrine selectivity: potent suppression of growth hormone but not of insulin. Endocrinol 2001; 142: 477–486.
Eriksson B, Renstrup J, Imam H, Öberg K. High-dose treatment with lanreotide of patients with advanced neuroendocrine gastrointestinal tumors: clinical and biological effects. Ann Oncol 1997; 8: 1041–1044.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fjällskog, ML., Ludvigsen, E., Stridsberg, M. et al. Expression of somatostatin receptor subtypes 1 to 5 in tumor tissue and intratumoral vessels in malignant endocrine pancreatic tumors. Med Oncol 20, 59–67 (2003). https://doi.org/10.1385/MO:20:1:59
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/MO:20:1:59