Abstract
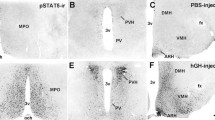
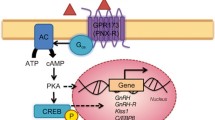
To identify new drug targets for the treatment of obesity, we employed a degenerate reverse transcriptase-polymerase chain reaction technique to isolate novel members of the G-protein coupled receptor superfamily from mouse hypothalamus. One of our clones was found to encode a protein with 90% amino acid identity to human GPR10, which was previously identified as the receptor for prolactin-releasing peptide (PrRP) and has been implicated in lactation, the regulation of food intake and other physiological functions. To investigate the role of GPR10 in food intake and body weight homeostasis, we generated mice carrying a targeted deletion of the GPR10 gene. First, using these knockout animals, we confirmed that GPR10 is the principle receptor for PrRP in the mouse hypothalamus because deletion of GPR10 completely abolished PrRP binding to isolated hypothalamic cell membranes. Second, we investigated the effect of normal and high-fat diets on energy intake, body weight, and glucose homeostasis in wild-type and GPR10 knockout mice. After fasting and refeeding, food intake in knockout animals was unchanged relative to control littermates. However, beginning at 16 wk of age on a normal diet, knockout mice became hyperphagic, obese, and showed significant increases in body fat and the levels of leptin and insulin, as well as decreased glucose tolerance. This metabolic profile was similar to the effect of a high-fat diet on wild-type animals. Our findings provide direct evidence that GPR10 is the receptor for PrRP and that it is involved in the regulation of energy balance in mice. GPR10 knockout mice will also prove useful for investigating other proposed activities for PrRP.
Similar content being viewed by others
References
Bhattacharyaa S., Luan J., Challis B., et al. (2003) Association of polymorphisms in GPR10, the gene encoding the prolactin-releasing peptide receptor with blood pressure, but not obesity, in a U.K Caucasian population. Diabetes. 52, 1296–1299.
Bray G. A. and York D. A. (1979) Hypothalamic and genetic obesity in experimental animals: an autonomic and endocrine hypothesis. Physiol. Rev. 59, 719–809.
Ellacott K. L., Lawrence C. B., Rothwell N. J., and Luckman S. M. (2002) PRL-releasing peptide interacts with leptin to reduce food intake and body weight. Endocrinology 143, 368–374.
Fujii R., Fukusumi S., Hosoya M., et al. (1999) Tissue distribution of prolactin-releasing peptide (PrRP) and its receptor. Regul. Pept. 83, 1–10.
Hinuma S., Habata Y., Fujii R., et al. (1998) A prolactin-releasing peptide in the brain. Nature 393, 272–276.
Ibata Y., Iijima N., Kataoka Y., et al. (2000) Morphological survey of prolactin-releasing peptide and its receptor with special reference to their functional roles in the brain. Neurosci. Res. 38, 223–230.
Iijima N., Kataoka Y., Kakihara K., et al. (1999) Cytochemical study of prolactin-releasing peptide (PrRP) in the rat brain. Neuroreport. 10, 1713–1716.
Jarry H., Heuer H., Schomburg L. and Bauer K. (2000) Prolactin-releasing peptides do not stimulate prolactin release in vivo. Neuroendocrinology 71, 262–267.
Langmead C. J., Szekeres P. G., Chambers J. K., et al. (2000) Characterization of the binding of [(125)I]-human prolactin releasing peptide (PrRP) to GPR10, a novel G protein coupled receptor. Br. J. Pharmacol. 131, 683–688.
Lawrence C. B., Celsi F., Brennand J., and Luckman S. M. (2000) Alternative role for prolactin-releasing peptide in the regulation of food intake. Nat. Neurosci. 3, 645–646.
Lawrence C. B., Ellacott K. L., and Luckman, S. M. (2002) PRL-releasing peptide reduces food intake and may mediate satiety signaling. Endocrinology. 143, 360–367.
Lin S. H., Arai A. C., Espana R. A., et al. (2002) Prolactin-releasing peptide (PrRP) promotes awakening and suppresses absence seizures. Neuroscience. 114, 229–238.
Marchese A., Docherty J. M., Nguyen T., et al. (1995) Cloning of human genes encoding novel G protein-coupled receptors. Genomics. 23, 609–618.
Maruyama M., Matsumoto H., Fujiwara K., et al. (2001) Prolactin-releasing peptide as a novel stress mediator in the central nervous system. Endocrinology. 142, 2032–2038.
Maruyama M., Matsumoto H., Fujiwara K., et al. (1999) Central administration of prolactin-releasing peptide stimulates oxytocin release in rats. Neurosci. Lett. 276, 193–196.
Matalon R., Rady P. L., Platt K. A., et al. (2000) Knock-out mouse for Canavan disease: a model for gene transfer to the central nervous system. J. Gene Med. 2, 165–175.
Matsumoto H., Noguchi J., Horikoshi Y., et al. (1999) Stimulation of prolactin release by prolactin-releasing peptide in rats. Biochem. Biophys. Res. Commun. 259, 321–324.
Minami S., Nakata T., Tokita R., Onodera H., and Imaki, J. (1999) Cellular localization of prolactin-releasing peptide messenger RNA in the rat brain. Neurosci. Lett. 266, 73–75.
Morales T., Hinuma S., and Sawchenko P. E. (2000) Prolactin-releasing peptide is expressed in afferents to the endocrine hypothalamus, but not in neurosecretory neurons. J. Neuroendocrinol. 12, 131–140.
Roland B. L., Sutton S. W., Wilson S. J., et al. (1999) Anatomical distribution of prolactin-releasing peptide and its receptor suggests additional functions in the central nervous system and periphery. Endocrinology. 140, 5736–5745.
Samson W. K., Resch Z. T., and Murphy, T. C. (2000) A novel action of the newly described prolactin-releasing peptides: cardiovascular regulation. Brain Res. 858, 19–25.
Seal L. J., Small C. J., Dhillo W. S., Kennedy A. R., Ghatei, M. A., and Bloom, S. R. (2002) Prolactin-releasing peptide releases corticotropin-releasing hormone and increases plasma adrenocorticotropin via the paraventricular nucleus of the hypothalamus. Neuroendocrinology. 76, 70–78.
Seal L. J., Small C. J., Kim M. S., et al. (2000) Prolactin releasing peptide (PrRP) stimulates luteinizing hormone (LH) and follicle stimulating hormone (FSH) via a hypothalamic mechanism in male rats. Endocrinology. 141, 1909–1912.
Thomas K. R. and Capecchi M. R. (1987) Site-directed mutagenesis by gene targeting in mouse embryoderived stem cells. Cell 51, 503–512.
Tokita R., Nakata T., Katsumata H., et al. (1999) Prolactin secretion in response to prolactin-releasing peptide and the expression of the prolactin-releasing peptide gene in the medulla oblongata are estrogen dependent in rats. Neurosci. Lett. 276, 103–106.
Vergoni A. V., Watanobe H., Guidetti G., Savino G., Bertolini A., and Schioth, H. B. (2002) Effect of repeated administration of prolactin releasing peptide on feeding behavior in rats. Brain Res. 955, 207–213.
Wattler S., Kelly M., and Nehls M. (1999) Construction of gene targeting vectors from lambda KOS genomic libraries. Biotechniques 26, 1150–1160.
Welch S. K., O’Hara B. F., Kilduff T. S., and Heller H. C. (1995) Sequence and tissue distribution of a candidate G-coupled receptor cloned from rat hypothalamus. Biochem. Biophys. Res. Commun. 209, 606–613.
Yamakawa K., Kudo K., Kanba S., and Arita J. (1999) Distribution of prolactin-releasing peptide-immunoreactive neurons in the rat hypothalamus. Neurosci. Lett. 267, 113–116.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gu, W., Geddes, B.J., Zhang, C. et al. The prolactin-releasing peptide receptor (GPR10) regulates body weight homeostasis in mice. J Mol Neurosci 22, 93–103 (2004). https://doi.org/10.1385/JMN:22:1-2:93
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:22:1-2:93