Abstract
Differential diagnosis of follicular adenoma (FA) and follicular carcinoma (FC) of the thyroid can be difficult in the routine practice of surgical pathology because the diagnosis of FC is strictly defined and determined by the presence of capsular and/or vascular invasion by the tumor. These features may be equivocal in the histologic sections. On the other hand, telomerase is expressed in many human cancers and is thought to contribute to their immortality. Human telomerase reverse transcriptase (hTERT) is the major determinant of human telomerase activity, and its expression is suggestive of capacity for unlimited replication.
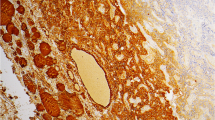
This case-control study examined the expression of hTERT using immunohistochemistry in 36 thyroid FC and 36 FA from patients who were matched by age and sex. The aim was to investigate the value of immunohistochemical staining for hTERT in the differential diagnosis of follicular neoplasms. The results revealed 23 cases of FC and 14 cases of FA that showed high expression of hTERT, with moderate to strong immunoreactivity. The remaining cases showed weak or negative staining. The difference between FA and FC was statistically significant (p<0.05).
In conclusion, immunohistochemical staining for hTERT can be considered an ancillary marker for differential diagnosis of FA and FC.
Similar content being viewed by others
References
Alvarez-Nunez F, Mora J, Matias-Guiu X. Thyroid carcinomas of the follicular epithelium: tumor markers and oncogenes. Med Clin (Barc) 121:264–269, 2003.
Liou MJ, Chan EC, Lin JD, Liu FH, Chao TC. Human telomerase reverse transcriptase (hTERT) gene expression in FNA samples from thyroid neoplasms. Cancer Lett 191:223–227, 2003.
Feng J, Funk WD, Wang SS, et al. The RNA component of human telomerase. Science 269:1236–1241, 1995.
Meyerson M, Counter CM, Eaton EN, et al. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell 90:785–795, 1997.
Nakamura TM, Morin GB, Chapman KB, et al. Telomerase catalytic subunit homologs from fission yeast and human. Science 277:955–959, 1997.
Yan P, Benhattar J, Seelentag W, Stehle JC, Bosman FT. Immunohistochemical location of hTERT protein in human tissues. Histochem Cell Biol 121:391–397, 2004.
Martin JA, DeYoung BR, Gitelis S, et al. Telomerase reverse transcriptase subunit expression is associated with chondrosarcoma malignancy. Clin Orthop 426:117–124, 2004.
Lantuejoul S, Soria JC, Moro-Sibilot D, et al. Differential expression of telomerase reverse transcriptase (hTERT) in lung tumours. Br J Cancer 90:1222–1229, 2004.
Malaska J, Kunicka Z, Borsky M, et al. Telomerase as a diagnostic and predictive marker in colorectal carcinoma. Neoplasma 51:90–96, 2004.
Hirokawa M, Carney JA, Goellner JR, et al. Observer variation of encapsulated follicular lesions of the thyroid gland. Am J Surg Pathol 26:1508–1514, 2002.
Bodnar AG, Quellette M, Frolkis M, et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279:349–352, 1998.
Hahn WC, Counter CM, Lundberg AS, Beijersbergen RL, Brooks MW, Weinberg RA. Creation of human tumour cells with defined genetic elements. Nature 400:464–468, 1999.
Jiang XR, Jimenez G, Chang E, et al. Telomerase expression in human somatic cells does not induce changes associated with a transformed phenotype. Nat Genet 21:111–114, 1999.
Kiyono T, Foster SA, Koop JI, McDougall JK, Galloway DA, Klingelhutz AJ. Both Rb of p161NK4a inactivation and telomerase activity are required to immortalize human epithelial cells. Nature 396:84–88, 1998.
Bornstein-Quevedo L, Garcia-Hernandez ML, Camacho-Arroyo I, et al. Telomerase activity in well-differentiated papillary thyroid carcinoma correlates with advanced clinical stage of the disease. Endocr Pathol 14:213–219, 2003.
Nakayama J, Tahara H, Tahara E, et al. Telomerase activation by hTRT in human normal fibroblasts and hepatocellular carcinomas. Nat Genet 18:65–68, 1998.
Yashima K, Milchgrub S, Gollahon LS, et al. Telomerase enzyme activity and RNA expression during the multistage pathogenesis of breast carcinoma. Cancer Res 4:229–234, 1998.
Ramakrishnan S, Eppenberger U, Mueller H, Shinkai Y, Narayanan R. Expression profile of the putative catalytic subunit of the telomerase gene. Cancer Res 58:622–625, 1998.
Trulsson LM, Velin AK, Herder A, Soderkvist P, Ruter A, Smeds S. Telomerase activity in surgical specimens and fine-needle aspiration biopsies from hyperplastic and neoplastic human thyroid tissues. Am J Surg 186:83–88, 2003.
Matthews P, Jones CJ, Skinner J, Haughton M, de Micco C, Wynford-Thomas D. Telomerase activity and telomere length in thyroid neoplasia: biological and clinical implications. J Pathol 194:183–193, 2001.
Zeiger MA, Smallridge RC, Clark DP, et al. Human telomerase reverse transcriptase (hTERT) gene expression in FNA samples from thyroid neoplasms. Surgery 126:1195–1198, 1999.
Umbricht CB, Saji M, Westra WH, Udelsman R, Zeiger MA, Sukumar S. Telomerase activity: a marker to distinguish follicular thyroid adenoma from carcinoma. Cancer Res 57:2144–2147, 1997.
Kawakami Y, Kitamoto M, Nakanishi T, et al. Immunohistochemical detection of human telomerase reverse transcriptase in human liver tissues. Oncogene 19:3888–3893, 2000.
Kumaki F, Kawai T, Hiroi S, et al. Telomerase activity and expression of human telomerase RNA component and human telomerase reverse transcriptase in lung carcinomas. Hum Pathol 32:188–195, 2001.
Kumaki F, Kawai T, Churg A, et al. Expression of telomerase reverse transcriptase (TERT) in malignant mesotheliomas. Am J Surg Pathol 26:365–370, 2002.
Tahara H, Yasui W, Tahara E, et al. Immunohistochemical detection of human telomerase catalytic component hTERT in human colorectal tumour and non tumour tissue sections. Oncogene 18:1561–1567.
Iczkowski KA, Pantazis CG, McGregor DH, Wu Y, Tawfik Ossama W. Telomerase reverse transcriptase subunit immunoreactivity—a marker for high-grade prostate carcinoma. Cancer 95:2487–2493, 2002.
Isobe K, Yashiro T, Omura S, et al. Expression of the human telomerase reverse transcriptase in pheochromocytoma and neuroblastoma tissues. Endocr J 51:47–52, 2004.
Hruban RH, Huvos AG, Traganos F, Reuter V, Lieberman PH, Melamed MR. Follicular neoplasms of the thyroid in men older than 50 years of age. A DNA flow cytometric study. Am J Clin Pathol 94:527–532, 1990.
Brennan MD, Bergstralh EJ, van Heerden JA, McConahey WM. Follicular thyroid cancer treated at the Mayo clinic, 1946 through 1970: initial manifestations, pathologic findings, therapy and outcome. Mayo Clin Proc 66:11–22, 1991.
Shaha AR, Loree TR, Shah JP. Prognostic factors and risk group analysis in follicular carcinoma of the thyroid. Surgery 18:1136–1138, 1995.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, SL., Chen, WT., Wu, MT. et al. Expression of human telomerase reverse transcriptase in thyroid follicular neoplasms: An immunohistochemical study. Endocr Pathol 16, 211–217 (2005). https://doi.org/10.1385/EP:16:3:211
Issue Date:
DOI: https://doi.org/10.1385/EP:16:3:211