Abstract
Purpose
Gastrointestinal stromal tumor (GIST) is associated with increased risk of additional cancers. In this study, synchronous GIST, and peritoneal mesothelioma (PM) were characterized to evaluate the relationship between these two cancers.
Methods
A retrospective chart review was conducted for patients diagnosed with both GIST and PM between July 2010 and June 2021. Patient demographics, past tumor history, intraoperative reports, cross-sectional imaging, peritoneal cancer index (PCI) scoring, somatic next-generation sequencing (NGS) analysis, and histology were reviewed.
Results
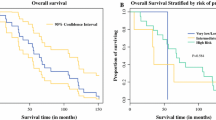
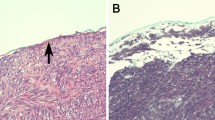
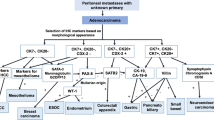
Of 137 patients who underwent primary GIST resection from July 2010 to June 2021, 8 (5.8%) were found to have synchronous PM, and 4 patients (50%) had additional cancers and/or benign tumors. Five (62.5%) were male, and the median age at GIST diagnosis was 57 years (range: 45–76). Seventy-five percent of GISTs originated from the stomach. Of the eight patients, one patient had synchronous malignant mesothelioma (MM), and the remaining had well-differentiated papillary mesothelioma (WDPM), which were primarily located in the region of the primary GIST (89%). The median PCI score was 2 in the WDPM patients. NGS of GIST revealed oncogenic KIT exon 11 (62.5%), PDGFRA D842V (25%), or SDH (12.5%) mutations, while NGS of the MM revealed BAP1 and PBRM1 alterations.
Conclusions
One in 17 GIST patients undergoing resection in this series have PM, which is significantly higher than expected if these two diseases were considered as independent events. Our results indicate that synchronous co-occurrence of GIST and PM is an underrecognized finding, suggesting a possible relationship that deserves further investigation.


Similar content being viewed by others
References
Ma GL, Murphy JD, Martinez ME, et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomark Prev. 2015;24:298–302.
Patil DT, Rubin BP. Gastrointestinal stromal tumor: advances in diagnosis and management. Arch Pathol Lab Med. 2011;135:1298–310.
Murphy JD, Ma GL, Baumgartner JM, et al. Increased risk of additional cancers among patients with gastrointestinal stromal tumors: a population-based study. Cancer. 2015;121:2960–7.
Ferreira SS, Werutsky G, Toneto MG, et al. Synchronous gastrointestinal stromal tumors (GIST) and other primary cancers: case series of a single institution experience. Int J Surg. 2010;8:314–7.
Ponti G, Luppi G, Martorana D, et al. Gastrointestinal stromal tumor and other primary metachronous or synchronous neoplasms as a suspicion criterion for syndromic setting. Oncol Rep. 2010;23:437–44.
Chen CI, Castellanos MB, Ruch MA, et al. Gastrointestinal stromal tumor with synchronous colorectal adenocarcinoma. QJM. 2017;110:591–2.
Comandini D, Damiani A, Pastorino A. Synchronous GISTs associated with multiple sporadic tumors: a case report. Drugs Context. 2017;6:212307.
Pandurengan RK, Dumont AG, Araujo DM, et al. Survival of patients with multiple primary malignancies: a study of 783 patients with gastrointestinal stromal tumor. Ann Oncol. 2010;21:2107–11.
Greenbaum A, Alexander HR. Peritoneal mesothelioma. Transl Lung Cancer Res. 2020;9:S120–32.
Sugarbaker PH, Welch LS, Mohamed F, et al. A review of peritoneal mesothelioma at the Washington cancer institute. Surg Oncol Clin N Am. 2003;12(605–21):xi.
Cheung M, Testa JR. BAP1, a tumor suppressor gene driving malignant mesothelioma. Transl Lung Cancer Res. 2017;6:270–8.
Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res. 1996;82:359–74.
Sun M, Zhao L, Yu L, et al. Well differentiated papillary mesothelioma: a 17 year single institution experience with a series of 75 cases. Ann Diagn Pathol. 2019;38:43–50.
Smith MJ, Smith HG, Mahar A, et al. The impact of additional malignancies in patients diagnosed with gastrointestinal stromal tumors. Int J Cancer. 2016;139(8):1744–51.
Stevers M, Rabban JT, Garg K, et al. Well-differentiated papillary mesothelioma of the peritoneum is genetically defined by mutually exclusive mutations in TRAF7 and CDC42. Mod Pathol. 2019;32:88–99.
Shrestha R, Nabavi N, Volik S, et al. Well-differentiated papillary mesothelioma of the peritoneum is genetically distinct from malignant mesothelioma. Cancers (Basel). 2020;12:1568.
Yu W, Chan-On W, Teo M, et al. First somatic mutation of E2F1 in a critical DNA binding residue discovered in well-differentiated papillary mesothelioma of the peritoneum. Genome Biol. 2011;12:R96.
de la Torre J, Banerjee S, Baumgartner J, et al. Tumor symbiosis: gastrointestinal stromal tumor as a host for primary peritoneal mesothelioma. J Gastrointest Surg. 2019;23:879–81.
Kim M, Kim HS. Clinicopathological characteristics of well-differentiated papillary mesothelioma of the peritoneum: a single-institutional experience of 12 cases. In vivo. 2019;33:633–42.
Malpica A, Sant’Ambrogio S, Deavers MT, et al. Well-differentiated papillary mesothelioma of the female peritoneum: a clinicopathologic study of 26 cases. Am J Surg Pathol. 2012;36:117–27.
Rathi V, Hyde S, Newman M. Well-differentiated papillary mesothelioma in association with endometrial carcinoma: a case report. Acta Cytol. 2010;54(5 Suppl):793–7.
McGinnis JM, Bloomfield V, Kazerouni H, Helpman L. Well-differentiated papillary mesothelioma with two synchronous serous gynaecologic carcinomas in a 62-year-old woman: lessons learned for the gynaecologic surgeon. J Obstet Gynaecol Can. 2020;42(10):1262–6. https://doi.org/10.1016/j.jogc.2019.12.012.
Atay S, Banskota S, Crow J, et al. Oncogenic KIT-containing exosomes increase gastrointestinal stromal tumor cell invasion. Proc Natl Acad Sci. 2014;111:711–6.
Atay S, Godwin AK. Tumor-derived exosomes: a message delivery system for tumor progression. Commun Integr Biol. 2014;7:e28231.
Atay S, Wilkey DW, Milhem M, et al. Insights into the proteome of gastrointestinal stromal tumors-derived exosomes reveals new potential diagnostic biomarkers. Mol Cell Proteomics. 2018;17:495–515.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
J.K.S. receives research funds from Foundation Medicine Inc. and Amgen; consultant fees from Deciphera and Grand Rounds; speakers fees from Deciphera, La-Hoffman Roche, Foundation Medicine, Merck, and QED; owns stock in Personalis. A.S. received financial support through the NIH T32 CA121938 Cancer Therapeutics (CT2) Training Fellowship. All other authors have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Courelli, A.S., Sharma, A.K., Madlensky, L. et al. Co-Localization of Gastrointestinal Stromal Tumors (GIST) and Peritoneal Mesothelioma: A Case Series. Ann Surg Oncol 29, 7542–7548 (2022). https://doi.org/10.1245/s10434-022-12211-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-12211-x