Abstract
Background
Disease burden in patients with peritoneal carcinomatosis (PC) is difficult to estimate. We evaluate whether peritoneal cell-free tumor DNA can be used as a measure of disease burden.
Patients and Methods
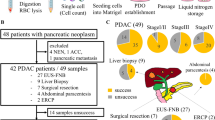
Malignant ascites or peritoneal lavage fluids were collected from patients with PC under approved IRB protocol. Cell-free DNA was extracted from peritoneal fluid. Droplet digital PCR (ddPCR) was performed using a commercially available KRAS G12/G13 screening kit. Mutant allele frequency (MAF) was calculated based on the numbers of KRAS wild-type and mutant droplets. Clinicopathological, treatment and outcome data were abstracted and correlated with MAF of cell-free KRAS mutant DNA.
Results
Cell-free KRAS mutant DNA was detected in 15/37 (40%) malignant peritoneal fluids with a MAF of 0.1% to 26.2%. While peritoneal cell-free KRAS mutant DNA was detected in all the patients with KRAS mutant tumors (N = 10), 3/16 (19%) patients with KRAS wild-type tumors also had peritoneal cell-free KRAS mutant DNA. We also found that 71% (5/7) of patients with disease amenable to cytoreductive surgery (CRS) had a MAF of < 1% (median: 0.5%, range: 0.1–4.7%), while 75% (6/8) of patients with unresectable disease had a MAF of > 1% (median: 4.4%, range: 0.1–26.2%).
Conclusions
This pilot proof-of-principle study suggests that peritoneal cell-free tumor DNA detected by ddPCR may enable prediction of disease burden and a measure of disease amenable to CRS in patients with PC.



Similar content being viewed by others
References
de Bree E, Koops W, Kroger R, van Ruth S, Witkamp AJ, Zoetmulder FA. Peritoneal carcinomatosis from colorectal or appendiceal origin: correlation of preoperative CT with intraoperative findings and evaluation of interobserver agreement. J Surg Oncol. 2004;86(2):64–73.
Pasqual EM, Bertozzi S, Bacchetti S, et al. Preoperative assessment of peritoneal carcinomatosis in patients undergoing hyperthermic intraperitoneal chemotherapy following cytoreductive surgery. Anticancer Res. 2014;34(5):2363–8.
Stroun M, Anker P, Lyautey J, Lederrey C, Maurice PA. Isolation and characterization of DNA from the plasma of cancer patients. Eur J Cancer Clin Oncol. 1987;23(6):707–12.
Diehl F, Schmidt K, Choti MA, et al. Circulating mutant DNA to assess tumor dynamics. Nat Med. 2008;14(9):985–90.
Francis G, Stein S. Circulating cell-free tumour DNA in the management of cancer. Int J Mol Sci. 2015;16(6):14122–42.
Hamakawa T, Kukita Y, Kurokawa Y, et al. Monitoring gastric cancer progression with circulating tumour DNA. Br J Cancer. 2015;112(2):352–6.
Murtaza M, Dawson SJ, Tsui DW, et al. Non-invasive analysis of acquired resistance to cancer therapy by sequencing of plasma DNA. Nature. 2013;497(7447):108–12.
Dawson SJ, Tsui DW, Murtaza M, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med. 2013;368(13):1199–209.
Buscail L, Bournet B, Cordelier P. Role of oncogenic KRAS in the diagnosis, prognosis and treatment of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2020.
Shoda K, Masuda K, Ichikawa D, et al. HER2 amplification detected in the circulating DNA of patients with gastric cancer: a retrospective pilot study. Gastric Cancer. 2015;18(4):698–710.
De Mattos-Arruda L, Olmos D, Tabernero J. Prognostic and predictive roles for circulating biomarkers in gastrointestinal cancer. Future Oncol. 2011;7(12):1385–97.
Schwaederle M, Chattopadhyay R, Kato S, et al. Genomic alterations in circulating tumor DNA from diverse cancer patients identified by next-generation sequencing. Cancer Res. 2017;77(19):5419–27.
Boysen AK, Wettergren Y, Sorensen BS, Taflin H, Gustavson B, Spindler KG. Cell-free DNA levels and correlation to stage and outcome following treatment of locally advanced rectal cancer. Tumour Biol. 2017;39(11):1010428317730976.
Spindler KL, Pallisgaard N, Andersen RF, Jakobsen A. Changes in mutational status during third-line treatment for metastatic colorectal cancer–results of consecutive measurement of cell free DNA, KRAS and BRAF in the plasma. Int J Cancer. 2014;135(9):2215–22.
Perets R, Greenberg O, Shentzer T, et al. Mutant KRAS circulating tumor DNA is an accurate tool for pancreatic cancer monitoring. Oncologist. 2018;23(5):566–72.
Hadano N, Murakami Y, Uemura K, et al. Prognostic value of circulating tumour DNA in patients undergoing curative resection for pancreatic cancer. Br J Cancer. 2016;115(1):59–65.
Baumgartner JM, Raymond VM, Lanman RB, et al. Preoperative circulating tumor DNA in patients with peritoneal carcinomatosis is an independent predictor of progression-free survival. Ann Surg Oncol. 2018;25(8):2400–2408.
Brychta N, Krahn T, von Ahsen O. Detection of KRAS mutations in circulating tumor DNA by digital PCR in early stages of pancreatic cancer. Clin Chem. 2016;62(11):1482–91.
Spindler KL, Pallisgaard N, Vogelius I, Jakobsen A. Quantitative cell-free DNA, KRAS, and BRAF mutations in plasma from patients with metastatic colorectal cancer during treatment with cetuximab and irinotecan. Clin Cancer Res. 2012;18(4):1177–85.
Fan G, Zhang K, Yang X, Ding J, Wang Z, Li J. Prognostic value of circulating tumor DNA in patients with colon cancer: Systematic review. PloS one. 2017;12(2):e0171991.
Cheng H, Luo G, Jin K, et al. Kras mutation correlating with circulating regulatory T cells predicts the prognosis of advanced pancreatic cancer patients. Cancer Med. 2020.
Portilla AG, Shigeki K, Dario B, Marcello D. The intraoperative staging systems in the management of peritoneal surface malignancy. J Surg Oncol. 2008;98(4):228–31.
Gerlinger M, Rowan AJ, Horswell S, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366(10):883–92.
Normanno N, Rachiglio AM, Lambiase M, et al. Heterogeneity of KRAS, NRAS, BRAF and PIK3CA mutations in metastatic colorectal cancer and potential effects on therapy in the CAPRI GOIM trial. Ann Oncol. 2015;26(8):1710–4.
Sclafani F, Chau I, Cunningham D, et al. KRAS and BRAF mutations in circulating tumour DNA from locally advanced rectal cancer. Sci Rep. 2018;8(1):1445.
Watanabe F, Suzuki K, Tamaki S, et al. Longitudinal monitoring of KRAS-mutated circulating tumor DNA enables the prediction of prognosis and therapeutic responses in patients with pancreatic cancer. PloS one. 2019;14(12):e0227366.
Sugimori M, Sugimori K, Tsuchiya H, et al. Quantitative monitoring of circulating tumor DNA in patients with advanced pancreatic cancer undergoing chemotherapy. Cancer Sci. 2020;111(1):266–78.
Demuth C, Spindler KG, Johansen JS, et al. Measuring KRAS mutations in circulating tumor DNA by droplet digital PCR and next-generation sequencing. Transl Oncol. 2018;11(5):1220–4.
Jung K, Fleischhacker M, Rabien A. Cell-free DNA in the blood as a solid tumor biomarker–a critical appraisal of the literature. Clin Chim Acta. 2010;411(21-22):1611–24.
Ako S, Nouso K, Kinugasa H, et al. Utility of serum DNA as a marker for KRAS mutations in pancreatic cancer tissue. Pancreatology. 2017;17(2):285–90.
Swisher EM, Wollan M, Mahtani SM, et al. Tumor-specific p53 sequences in blood and peritoneal fluid of women with epithelial ovarian cancer. Am J Obstet Gynecol. 2005;193(3 Pt 1):662–667.
Haupts A, Roth W, Hartmann N. [Liquid biopsy in colorectal cancer: An overview of ctDNA analysis in tumour diagnostics]. Pathologe. 2019;40(Suppl 3):244–51.
Riva F, Bidard FC, Houy A, et al. Patient-specific circulating tumor DNA detection during neoadjuvant chemotherapy in triple-negative breast cancer. Clin Chem. 2017;63(3):691–9.
Galbiati S, Damin F, Ferraro L, et al. Microarray approach combined with ddPCR: an useful pipeline for the detection and quantification of circulating tumour DNA mutations. Cells. 2019;8(8):769.
Pishvaian MJ, Bender RJ, Halverson D, et al. Molecular profiling of patients with pancreatic cancer: initial results from the know your tumor initiative. Clin Cancer Res. 2018;24(20):5018–27.
Acknowledgements
This study was supported by the Holden Comprehensive Cancer Center through funds from the National Cancer Institute of the National Institutes of Health under award no. P30 CA086862 for supporting the Molecular Epidemiology Resource Core. A.G.K. was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under award no. T35 HL007485. A.M.M. was supported by the National Institutes of Health Free Radical and Radiation Biology T32 CA078586 training grant. H.R.S. was supported by the Iowa Center for Research by Undergraduates Fellowship Awards.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
P.M.K.: consultancy/advisory board (Taiho, Ipsen, Foundation Medicine, Natera); Honoraria (AstraZeneca, Foundation Medicine, Natera). Foundation Medicine, a company focusing on genomics and ctDNA in blood/tissue, is not directly related to the submitted work but to ctDNA testing. Natera, a company focusing on ctDNA for stage II/III colorectal cancer, is not directly related to the submitted work but to the field of liquid biopsies. All other authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leick, K.M., Kazarian, A.G., Rajput, M. et al. Peritoneal Cell-Free Tumor DNA as Biomarker for Peritoneal Surface Malignancies. Ann Surg Oncol 27, 5065–5071 (2020). https://doi.org/10.1245/s10434-020-08832-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08832-9