Abstract
Purpose
To investigate the use of advanced SPECT/CT quantification in guiding surgical selection of positive sentinel lymph nodes (SLNs) in head and neck melanoma.
Methods
We retrospectively reviewed data from patients with cutaneous head and neck melanoma who underwent lymphoscintigraphy with SPECT/CT prior to SLN biopsy (SLNB). Quantification of radiotracer uptake from SPECT/CT data was performed using in-house segmentation software. SLNs identified using SPECT/CT were compared to SLNs identified surgically using an intraoperative γ-probe. A radioactivity count threshold using SPECT/CT for detecting a positive SLN was calculated.
Results
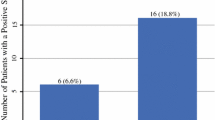
One hundred and five patients were included. Median number of SLNs detected was 3/patient with SPECT/CT and 2/patient with intraoperative γ-probe. The hottest node identified by SPECT/CT and intraoperative γ-probe were identical in 85% of patients. All 20 histologically positive SLNs were identified by SPECT/CT and γ-probe. On follow-up, all nodal recurrences occurred at lymph node levels with the hottest node identified by SPECT/CT and either the hottest or second hottest node identified by γ-probe during SLNB. Using our data, a SPECT/CT radioactivity count threshold of 20% would eliminate the unnecessary removal of 11% of SPECT/CT identified nodes and 12% of intraoperatively detected nodes.
Conclusion
Utilizing SPECT/CT quantification, we propose that a radioactivity count threshold can be developed to help guide the selective removal of lymph nodes in head and neck SLNB. Furthermore, the nodal level containing the hottest node identified by SPECT/CT quantification must be thoroughly investigated for SLNs and undergo careful follow-up and surveillance for recurrence.

Similar content being viewed by others
References
Balch CM, Soong SJ, Gershenwald JE, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol. 2001;19(16):3622–34.
Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med. 2014;370(7):599–609.
Testori A, De Salvo GL, Montesco MC, et al. Clinical considerations on sentinel node biopsy in melanoma from an Italian multicentric study on 1,313 patients (SOLISM-IMI). Ann Surg Oncol. 2009;16(7):2018–27.
Garbe C, Buttner P, Bertz J, et al. Primary cutaneous melanoma. Prognostic classification of anatomic location. Cancer. 1995;75(10):2492–8.
Lachiewicz AM, Berwick M, Wiggins CL, Thomas NE. Survival differences between patients with scalp or neck melanoma and those with melanoma of other sites in the Surveillance, Epidemiology, and End Results (SEER) program. Arch Dermatol. 2008;144(4):515–21.
Kaveh AH, Seminara NM, Barnes MA, et al. Aberrant lymphatic drainage and risk for melanoma recurrence after negative sentinel node biopsy in middle-aged and older men. Head Neck. 2016;38 Suppl 1:E754–60.
McMasters KM, Noyes RD, Reintgen DS, et al. Lessons learned from the Sunbelt Melanoma Trial. J Surg Oncol. 2004;86(4):212–23.
Miller MW, Vetto JT, Monroe MM, Weerasinghe R, Andersen PE, Gross ND. False-negative sentinel lymph node biopsy in head and neck melanoma. Otolaryngol Head Neck Surg. 2011;145(4):606–11.
Jimenez-Heffernan A, Ellmann A, Sado H, et al. Results of a prospective multicenter international Atomic Energy Agency sentinel node trial on the value of SPECT/CT over planar imaging in various malignancies. J Nucl Med. 2015;56(9):1338–44.
Wagner T, Buscombe J, Gnanasegaran G, Navalkissoor S. SPECT/CT in sentinel node imaging. Nucl Med Commun. 2013;34(3):191–202.
Chapman BC, Gleisner A, Kwak JJ, et al. SPECT/CT improves detection of metastatic sentinel lymph nodes in patients with head and neck melanoma. Ann Surg Oncol. 2016;23(8):2652–7.
McMasters KM, Reintgen DS, Ross MI, et al. Sentinel lymph node biopsy for melanoma: how many radioactive nodes should be removed? Ann Surg Oncol. 2001;8(3):192–7.
Som PM, Curtin HD, Mancuso AA. An imaging-based classification for the cervical nodes designed as an adjunct to recent clinically based nodal classifications. Arch Otolaryngol Head Neck Surg. 1999;125(4):388–96.
Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016;15(2):155–63.
McHugh ML. Interrater reliability: the kappa statistic. Biochem Med (Zagreb). 2012;22(3):276–82.
de Rosa N, Lyman GH, Silbermins D, et al. Sentinel node biopsy for head and neck melanoma: a systematic review. Otolaryngol Head Neck Surg. 2011;145(3):375–82.
Hu Y, Melmer PD, Slingluff CL, Jr. Localization of the sentinel lymph node in melanoma without blue dye. Ann Surg. 2016;263(3):588–92.
Liu LC, Parrett BM, Jenkins T, et al. Selective sentinel lymph node dissection for melanoma: importance of harvesting nodes with lower radioactive counts without the need for blue dye. Ann Surg Oncol. 2011;18(10):2919–24.
Faries MB, Thompson JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 2017;376(23):2211–22.
Leiter U, Stadler R, Mauch C, et al. Complete lymph node dissection versus no dissection in patients with sentinel lymph node biopsy positive melanoma (DeCOG-SLT): a multicentre, randomised, phase 3 trial. Lancet Oncol. 2016;17(6):757–67.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
No potential conflicts of interest to disclose. No financial or material support to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kwak, J.J., Kesner, A.L., Gleisner, A. et al. Utility of Quantitative SPECT/CT Lymphoscintigraphy in Guiding Sentinel Lymph Node Biopsy in Head and Neck Melanoma. Ann Surg Oncol 27, 1432–1438 (2020). https://doi.org/10.1245/s10434-019-08078-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-08078-0